Chemical Biology 03 BLOOD Biomolecular Structure Myoglobin and
Chemical Biology 03 BLOOD Biomolecular Structure Myoglobin and Hemoglobin 9/28 -30/09 www. optics. rochester. edu/. . . /image 007. gif

Chemical Biology 03 BLOOD Biomolecular Structure Myoglobin and Hemoglobin Lecture 9 and 10: 9/28 -30/09
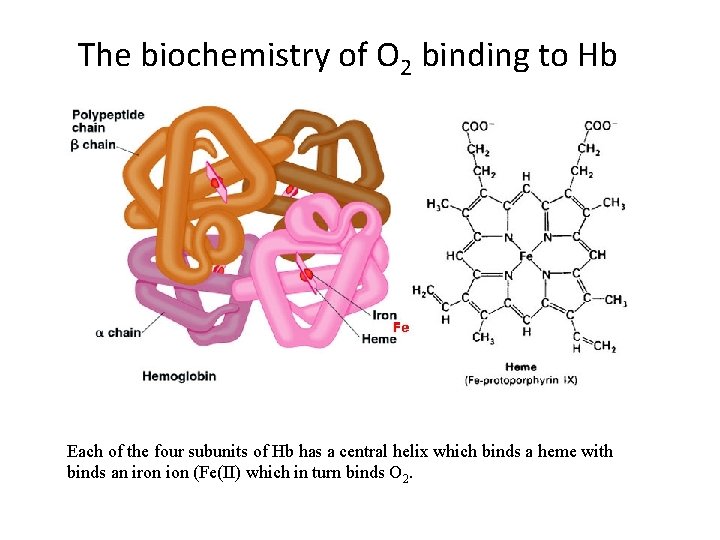
The biochemistry of O 2 binding to Hb Each of the four subunits of Hb has a central helix which binds a heme with binds an iron ion (Fe(II) which in turn binds O 2.
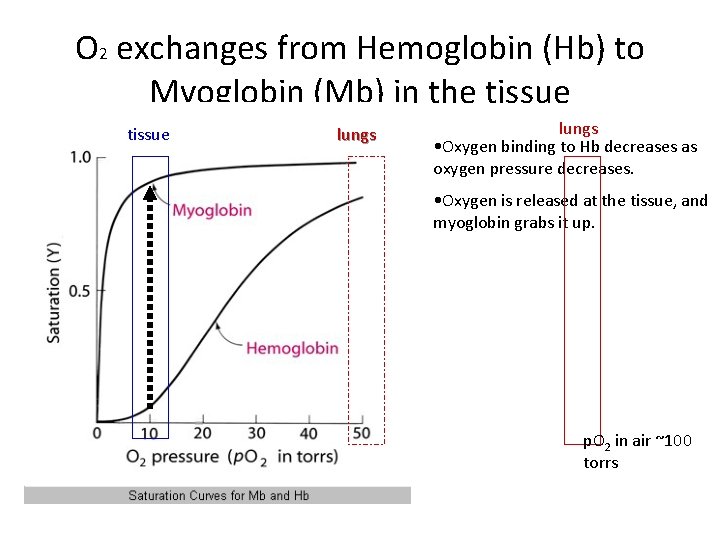
O 2 exchanges from Hemoglobin (Hb) to Myoglobin (Mb) in the tissue lungs • Oxygen binding to Hb decreases as oxygen pressure decreases. • Oxygen is released at the tissue, and myoglobin grabs it up. p. O 2 in air ~100 torrs

O 2 Exchanges from Hb to Mb • HEMOGLOBIN • MYOGLOBIN • Crystal structure is very complicated. • Crystal structure is very simple • Hb protein is four subunits, four heme groups, and seems to behave differently when all together as compared with monomers. • Oxygen saturation curve is “sigmoidal” complicated mathematical formula. O 2 • Mb protein is one subunit, one heme, and behaves simply • Oxygen saturation curve is hyperbolic, which mathematically is quite simple y= x/(a+x) So, let’s first try to understand Mb!!!
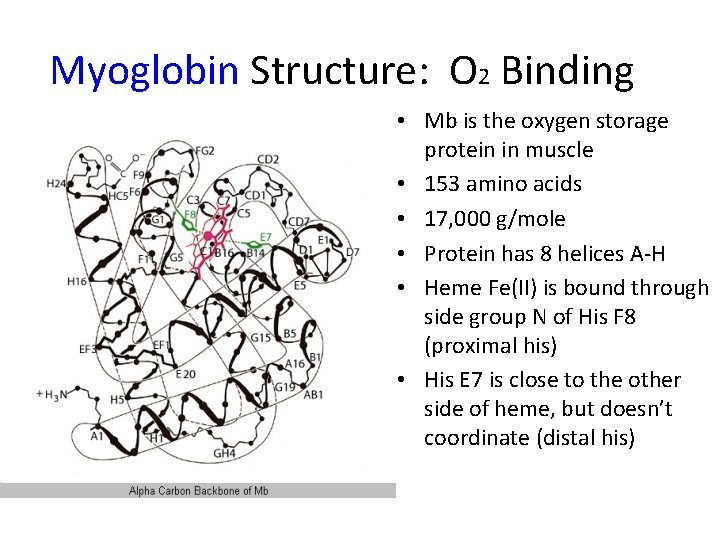
Myoglobin Structure: O 2 Binding • Mb is the oxygen storage protein in muscle • 153 amino acids • 17, 000 g/mole • Protein has 8 helices A-H • Heme Fe(II) is bound through side group N of His F 8 (proximal his) • His E 7 is close to the other side of heme, but doesn’t coordinate (distal his)
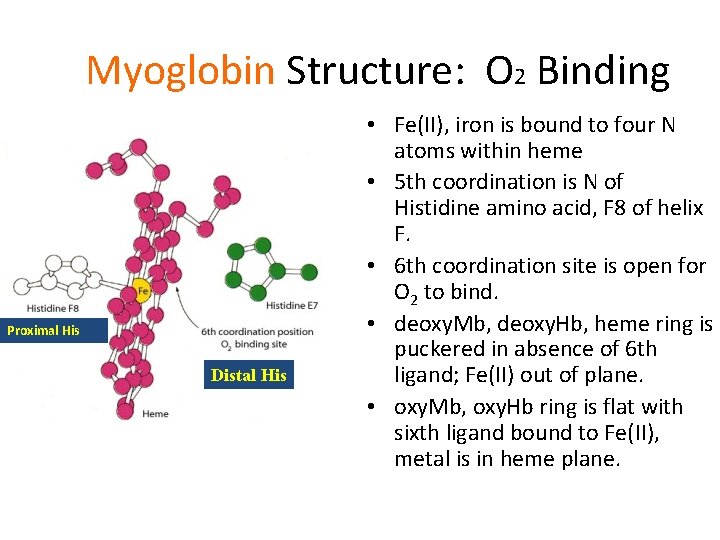
Myoglobin Structure: O 2 Binding Proximal His Distal His • Fe(II), iron is bound to four N atoms within heme • 5 th coordination is N of Histidine amino acid, F 8 of helix F. • 6 th coordination site is open for O 2 to bind. • deoxy. Mb, deoxy. Hb, heme ring is puckered in absence of 6 th ligand; Fe(II) out of plane. • oxy. Mb, oxy. Hb ring is flat with sixth ligand bound to Fe(II), metal is in heme plane.
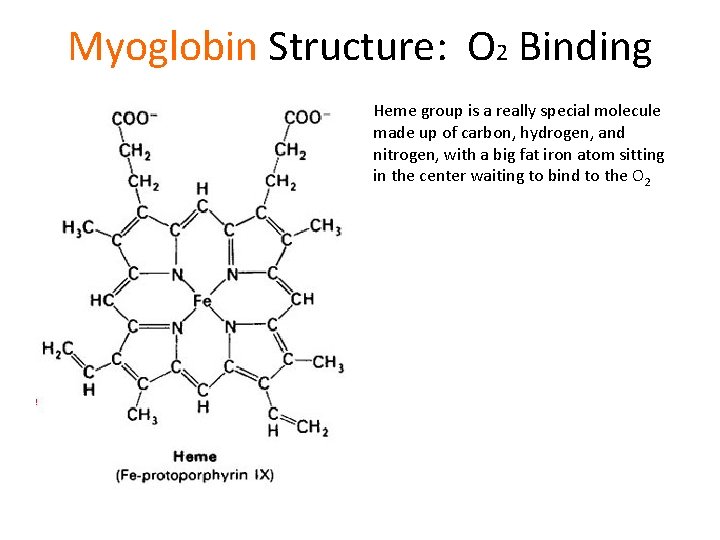
Myoglobin Structure: O 2 Binding Heme group is a really special molecule made up of carbon, hydrogen, and nitrogen, with a big fat iron atom sitting in the center waiting to bind to the O 2
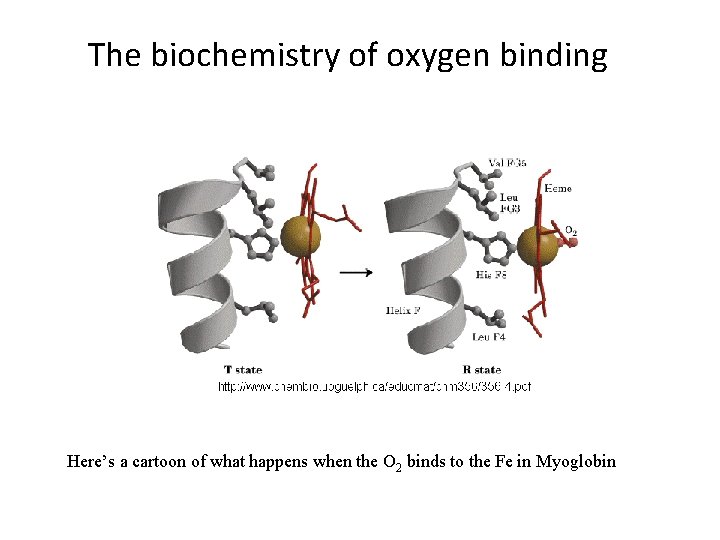
The biochemistry of oxygen binding Here’s a cartoon of what happens when the O 2 binds to the Fe in Myoglobin

Myoglobin Structure: O 2 Binding N from distal His N from proximal His – Effect of sixth site coordination on the color of myoglobin. – Oxygen does not bind straight on, the N from the distal His amino acid side group in the way.
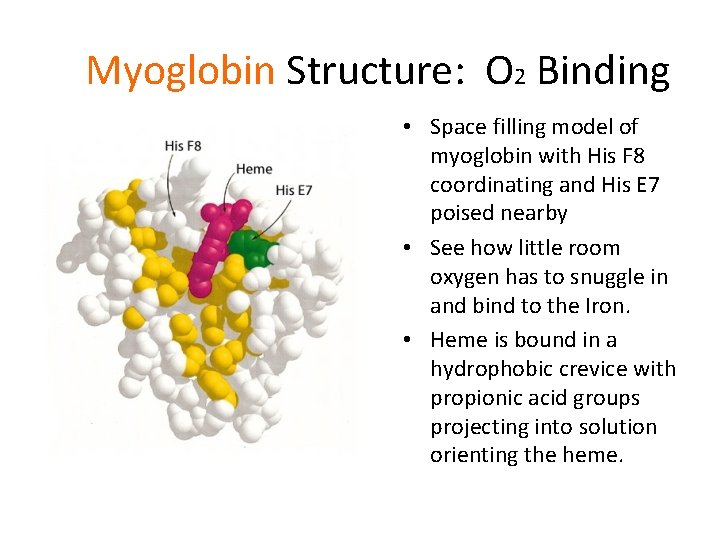
Myoglobin Structure: O 2 Binding • Space filling model of myoglobin with His F 8 coordinating and His E 7 poised nearby • See how little room oxygen has to snuggle in and bind to the Iron. • Heme is bound in a hydrophobic crevice with propionic acid groups projecting into solution orienting the heme.
A Tale of Two Binding Curves Myoglobin • getting the dissociation constant from the saturation curve Hemoglobin • sigmoidal saturation curve • two state model (T and R) • O 2 binding is cooperative
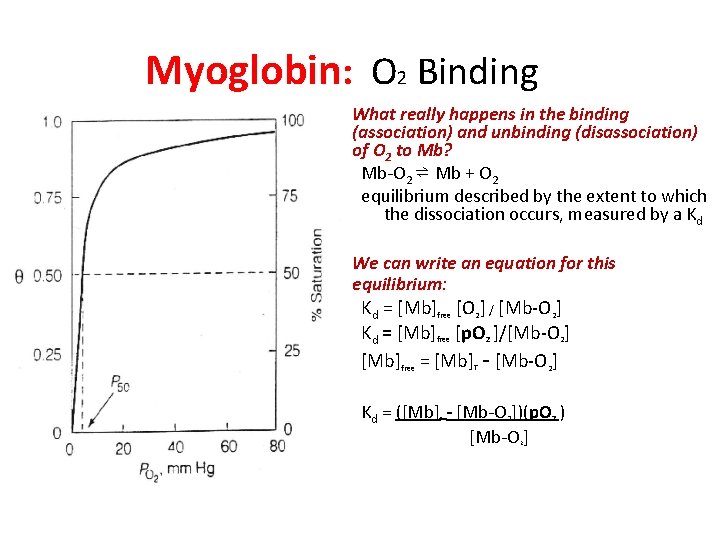
Myoglobin: O 2 Binding • What really happens in the binding (association) and unbinding (disassociation) of O 2 to Mb? Mb-O 2 ⇌ Mb + O 2 equilibrium described by the extent to which the dissociation occurs, measured by a Kd • We can write an equation for this equilibrium: Kd = [Mb]free [O 2] / [Mb-O 2] Kd = [Mb]free [p. O 2 ]/[Mb-O 2] [Mb]free = [Mb] - [Mb-O 2] T Kd = ([Mb] - [Mb-O 2])(p. O 2 ) [Mb-O ] T 2
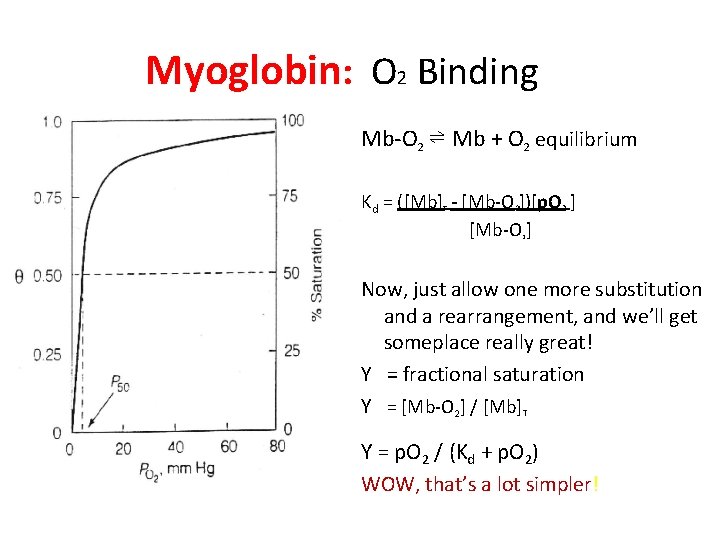
Myoglobin: O 2 Binding Mb-O 2 ⇌ Mb + O 2 equilibrium Kd = ([Mb] - [Mb-O 2])[p. O 2 ] [Mb-O ] T 2 Now, just allow one more substitution and a rearrangement, and we’ll get someplace really great! Y = fractional saturation Y = [Mb-O 2] / [Mb]T Y = p. O 2 / (Kd + p. O 2) WOW, that’s a lot simpler!

Let’s EXCEL together

Conclusions from excel about O 2 binding to Mb
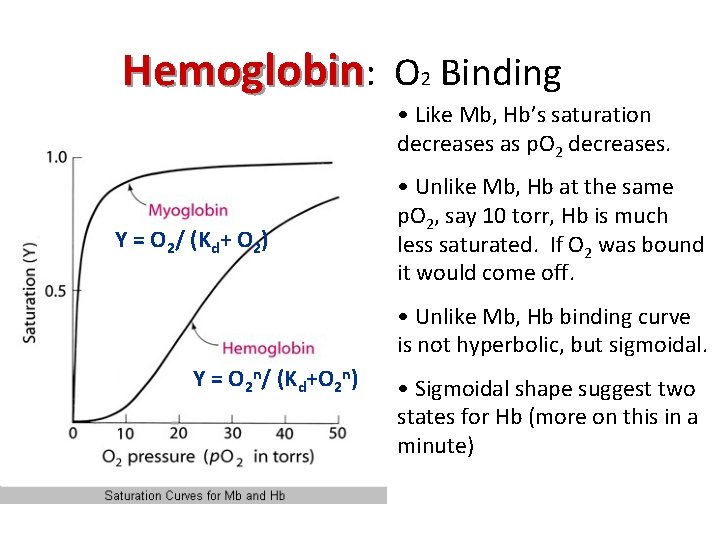
Hemoglobin: O 2 Binding • Like Mb, Hb’s saturation decreases as p. O 2 decreases. Y = O 2/ (Kd+ O 2) • Unlike Mb, Hb at the same p. O 2, say 10 torr, Hb is much less saturated. If O 2 was bound it would come off. • Unlike Mb, Hb binding curve is not hyperbolic, but sigmoidal. Y = O 2 n/ (Kd+O 2 n) • Sigmoidal shape suggest two states for Hb (more on this in a minute)

Let’s EXCEL together

Conclusions from excel about O 2 binding to Hb
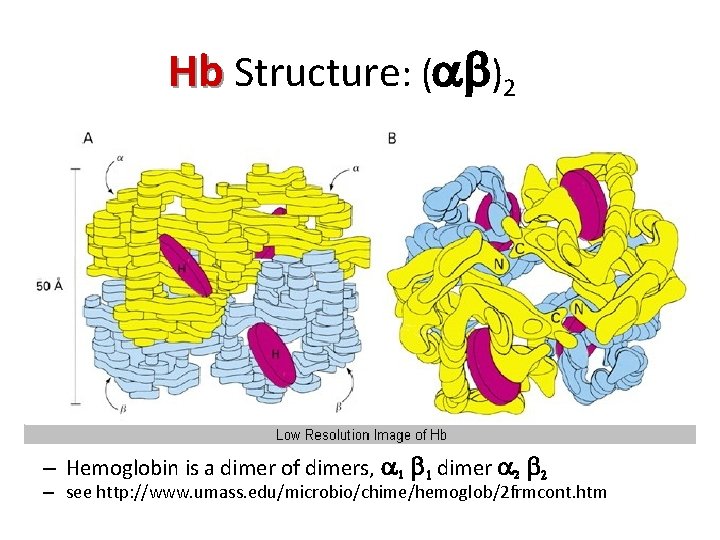
Hb Structure: (ab)2 – Hemoglobin is a dimer of dimers, a 1 b 1 dimer a 2 b 2 – see http: //www. umass. edu/microbio/chime/hemoglob/2 frmcont. htm
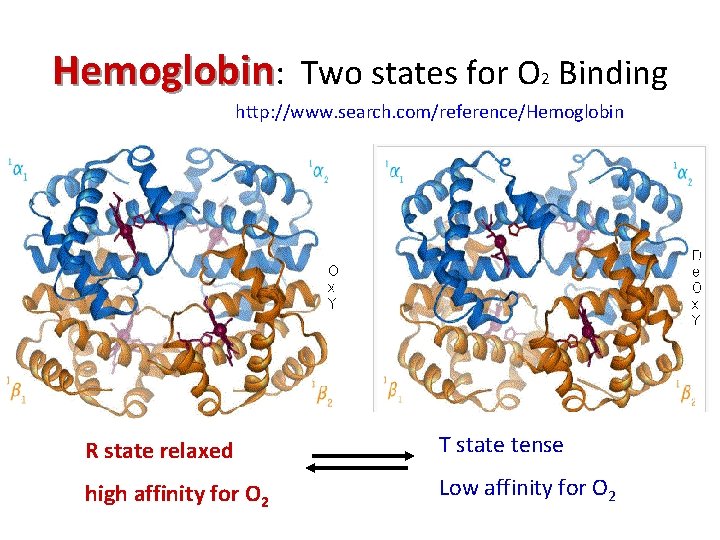
Hemoglobin: Two states for O 2 Binding http: //www. search. com/reference/Hemoglobin R state relaxed T state tense high affinity for O 2 Low affinity for O 2

Hemoglobin: Two states for O 2 Binding http: //www. search. com/reference/Hemoglobin R state relaxed T state tense high affinity for O 2 Low affinity for O 2
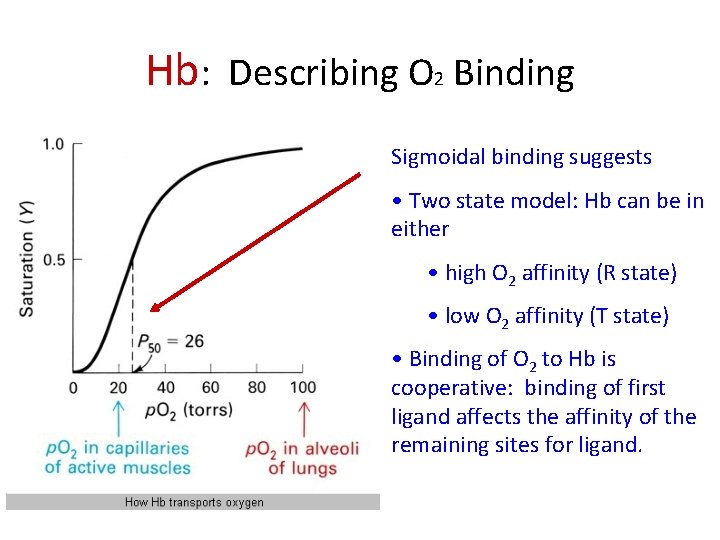
Hb: Describing O 2 Binding Sigmoidal binding suggests • Two state model: Hb can be in either • high O 2 affinity (R state) • low O 2 affinity (T state) • Binding of O 2 to Hb is cooperative: binding of first ligand affects the affinity of the remaining sites for ligand.
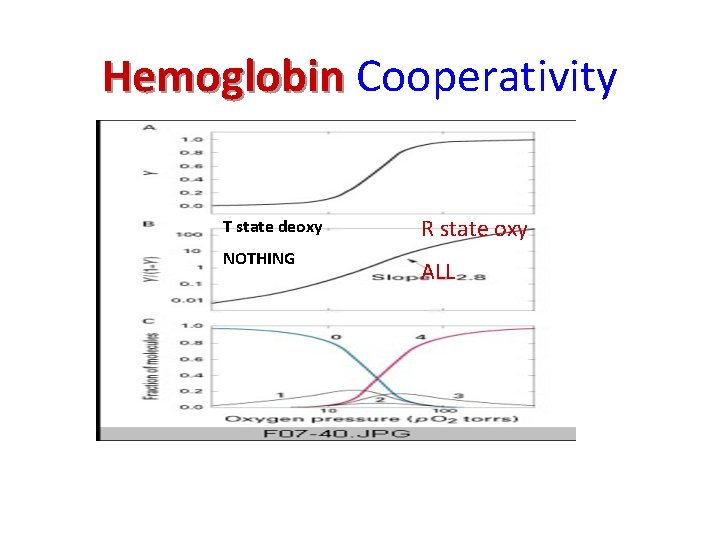
Hemoglobin Cooperativity T state deoxy NOTHING R state oxy ALL
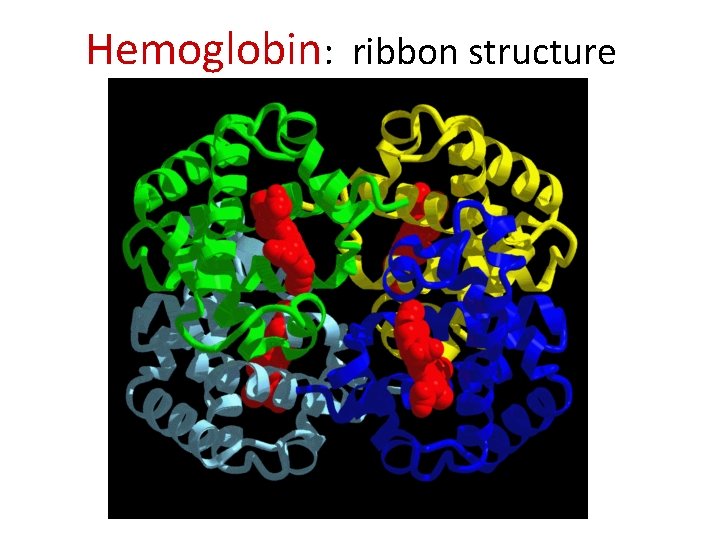
Hemoglobin: ribbon structure
- Slides: 25