Chemical and Physical Features of Sea Water and


































- Slides: 37

Chemical and Physical Features of Sea Water and the World Ocean Chapter 3

The Waters of the Ocean • Earth is the only planet with “liquid” water. • Marine organisms cannot control the physical and chemical nature of their environment so they have to “grin and bear it. ” • Organisms must adapt, live somewhere else, or die. • Marine organisms consist of mostly water. Most are 80% water. Jellyfish are 95% water!

The Unique Nature of Pure Water • • • Let’s take a minute to review: Atoms Molecules Hydrogen bonds Water is made of two hydrogen atoms bound to an oxygen atom by two hydrogen bonds.

The Three States of Water • Any substance can exist in three different states/phases: • Solid • Liquid • Gas • Water is the only substance that “naturally” occurs in all three states on earth.

Liquid Water • Liquid water is held together by Hydrogen bonds. These bonds move, so they break and reform constantly. • Temperature reflects the average speed of water. The higher the temperature the faster the water molecules are moving. • When water molecules move fast enough and break free of hydrogen bonds it escapes to the gas/vapor phase. This process is called “evaporation. ”

Water Vapor • Water vapor molecules are not held together by hydrogen bonds. They are separate and much farther apart. • As temperature increases the rate of evaporation increases. • Water boils at 100 C (212 F) when nearly all the hydrogen bonds are broken and the molecules enter the vapor state.

Solid Water • When liquid water cools, molecules move slower and pack closer together to take up less space. • When the temperature of water decreases the volume of water decreases without changing the mass, so the water gets denser. • Remember= D=m/V

Seawater • As seawater gets colder, it gets more dense and sinks.

Freshwater • As freshwater gets colder, it becomes less dense and floats. • That is why a sheet of ice can cover the top of a lake or pond allowing aquatic life to remain alive below.

Water Crystals • Crystals form when water molecules move slowly and the hydrogen bonds lock in a 3 -D pattern. • Ice crystals have molecules that are farther apart than liquid water. • Ice expands as it freezes.

Heat and Water • Ice is held with hydrogen bonds. • These bonds must be broken for ice to melt. • Ice melts at a much higher temperature than similar substances. • If not for the hydrogen bonds ice would melt at -90 C (-130 F) instead of 0 C (32 F)!

• Water absorbs a lot of heat when it melts. • The Latent Heat of Melting- is the amount of heat to melt a substance. • Water has a higher latent heat of melting than any other common substance. • A GREAT deal of heat must be removed to freeze water. It takes a long time before complete body of water will freeze solid!

Drinks stay cold because any heat goes to melting the ICE, not raising the temperature of the drink.

Heat Capacity • Is the amount of heat need to raise a substances temperature by a given amount. • Water has one of the highest heat capacities. • It can absorb a lot of heat and raise the temperature very little. • This provides for utility as a coolant for automobiles and allows marine life not to be subject to rapid changes in heating and cooling of their habitats.

Latent Heat of Evaporation • Ability of water to absorb a great deal of heat when it evaporates. • Evaporative cooling- when the fastest molecules leave the liquid phase take heat with them and those left behind on the organism, such as human skin, lower our body temperature.


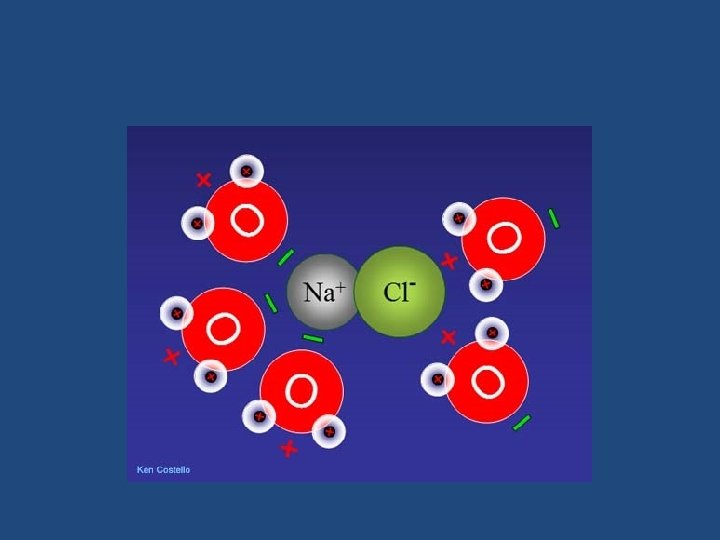
Water as a Solvent • Water can dissolve many more things than any other natural substance. • Ex: Salts- which are made of ions. Na+ Cl. Salt crystals in water attract water molecules with their charges like magnets. The ions dissociate (pull apart) and dissolve.


Seawater Composition • Seawater is composed of particles that are dissolved in the water by different means. • Weathering- produces some of the solids that are found in seawater. • Hydrothermal vents- release sediments into seawater from underwater geothermal activity.

Salt Composition- made of mostly 6 ions in 99% of seawater.

• Sodium chloride accounts for 85% of the solids in seawater. • When seawater evaporates salts are left behind. • Salinity- is the total amount of salt dissolved in seawater. • Ex: If 35 g of salt are left from 1000 g of seawater evaporation the seawater’s salinity is 35%. • Salinity is measured today with electrodes because salts are wonderful conductors.

Practical Salinity Units (psu) • Units of salinity from conductivity measurements. • Why is this important? • The degree of salinity affects organisms. • Not just the total amount of salt is important, but also the composition of the salt. • Some organisms may tolerate Na. Cl, while withstanding KCl.

• Chemist William Dittmar analyzed water samples from the Challenger and found that the major ions in seawater remained constant even though the total amount varied from place to place. • For example, Cl was always at 55. 03%. • This finding led to The Rule of Constant Proportion.

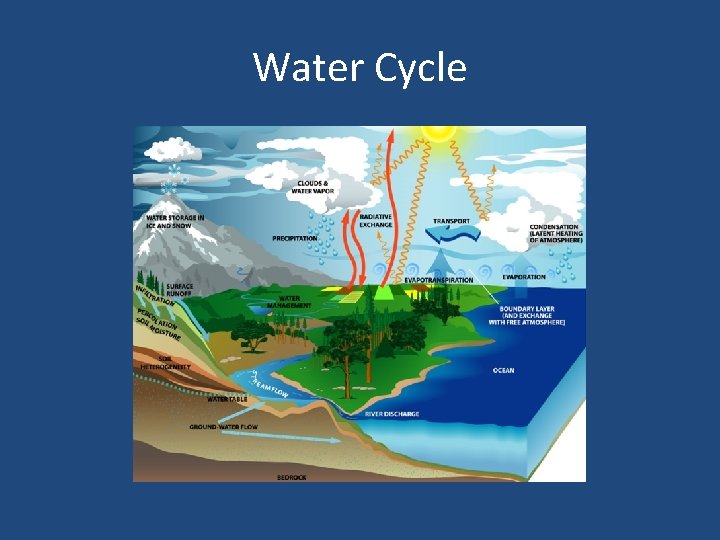
• Water is removed from the ocean primarily by evaporation. • When the water freezes the ions in the water do not freeze so frozen seawater becomes fresh! • This frozen water makes up the polar ice caps. • The salinity of the water surrounding the ice caps is very saline because of these remaining ions in the seawater.

• Water is added to the seas by precipitation and on a small scale glacial melting. • The average ocean salinity is 35%. Most seawater varies between 33%-37%. • The Red Sea has high evaporation and is 40% saline. • The Baltic Sea has a great deal of runoff from fresh water sources and is 7% saline.

Salinity/Temperature/Density • Temperature affects density. • The greater the temperature of the seawater the more dense is so it? • Salinity affects density. • The saltier the seawater the more dense the water is so it?

• Temperature in the open water varies from -2 C to 30 C (28 F-86 F). • In seawater temperatures can occur below 0 C (32 F) because seawater freezes at a cooler temperature. • This makes the ocean less prone to freeze then lakes and rivers.

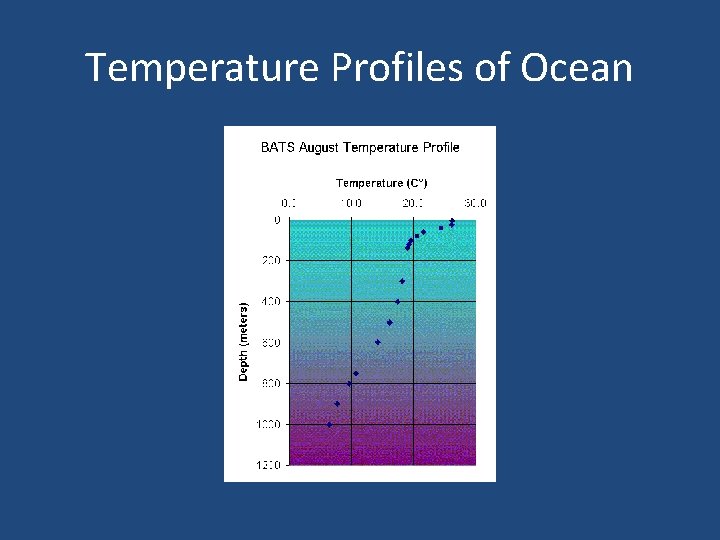
• Temperature in the ocean is measured with specially designed bottles and thermometers lowered to a desired depth. • Temperature Profiles- graphs generated from temperatures taken at different depths. • They reflect a water column- vertical column of temperatures of water at their respective depth.

Temperature Profiles of Ocean

Tools for data collection. • Researchers can also plot salinity or density at different depths using different probes. • Today CIDs are used to measure- Conductivity, Temperature, and Depth of ocean water. • Bathythermographs (XBTs) are used to measure temperature and are disposable. • Niksin bottles collect individual water sample from different depths. • Satellites measure conditions near the surface instantaneously.

Dissolved Gases in Water • • • The most important gases in seawater are: Oxygen Carbon Dioxide Nitrogen These gases are dissolved in the water from the atmosphere into the surface seawater and are released back into the atmosphere through evaporation (gas exchange. )

Water Cycle

• Gases dissolve better in cold water. • There are more dissolved gases found in polar waters. • Oxygen gas is not very soluble only 0 -8 ml of oxygen will dissolve in 1 L of seawater. Usually about 4 -6 ml. • In the atmosphere there is about 210 ml of oxygen in 1 L air.

Photosynthesis/Respiration • Photosynthesis and respiration affect the amount of dissolved oxygen in seawater. • Most of the oxygen produced in the ocean is by photosynthesizing organisms that release oxygen into the atmosphere as a product of this process. • Seawater has little dissolved oxygen.

• Carbon dioxide is much more soluble. • It makes up 80% of the dissolved gas in the ocean and stores 50 times as much CO 2 as the atmosphere! • This makes the ocean a critical component to controlling the greenhouse effect by absorbing a great deal of greenhouse gases that could be in the atmosphere increasing global

Transparency • Water is transparent • This is the ability for light to penetrate the water and allows photosynthetic organisms to grow. • Remember, sunlight has all the colors of the rainbow, and not all colors are going to penetrate equally. • Clear ocean water is most transparent to blue. • Transparency can be affected by large quantities of plankton (algae). They reduce transmittance of light from the surface.

Color Transparency of Ocean Water