Chemical and Physical Compatibility of Continuous Intravenous Drug

Chemical and Physical Compatibility of Continuous Intravenous Drug Infusion Combinations used in Paediatric Intensive Care Andy Fox and Marisa van der Merwe Pharmacy department

What’s the Problem? • Multiple Infusions • Lack of data – Two or more drugs – Diluents – Concentration – Non standard – Environment • Line Insertion Pharmacy department

Aims and Objectives • Provide evidence for the safety of current practice • Clinical Relevance by replicating PICU conditions • Identify combinations not suitable for single lumen administration • Satisfy legislation in relation of mixing of meds to enable informed decisions to be made. Pharmacy department

Study Design • 5 IV administered drugs: – Morphine, clonidine, ketamine, midazolam, furosemide • 3 Environmental conditions: – Diluent, temperature, blue light phototherapy • Drug mixtures: Maximum mass interaction within clinical dosage range and rate range – Clonidine was used at maximum available concentration of 150 mcg/ml Pharmacy department

Study Design • SCHOTT Type I glass vials plus® • Incubation time was either 4 or 8 hrs depending on drug combination. – Based on flow rate and distal volume after yconnector • Physical and chemical compatibility assessed – 4608 individual analyses Pharmacy department

Physical Compatibility • p. H measurement: – Upon mixing – After incubation • Precipitation: – Sympatec Helos particle sizer (cuvette module) • Colour change: – UV/Vis absorbance – Above 400 nm Pharmacy department
Chemical Compatibility Assessed with HPLC using a DAD Absorption at 200 nm Fu Mi Ke Cl Mo Time (min) Pharmacy department

Chemical Compatibility (No Precipitation) a) 90 Room temp High temp No phototherapy Pharmacy department Room temp Phototherapy High temp
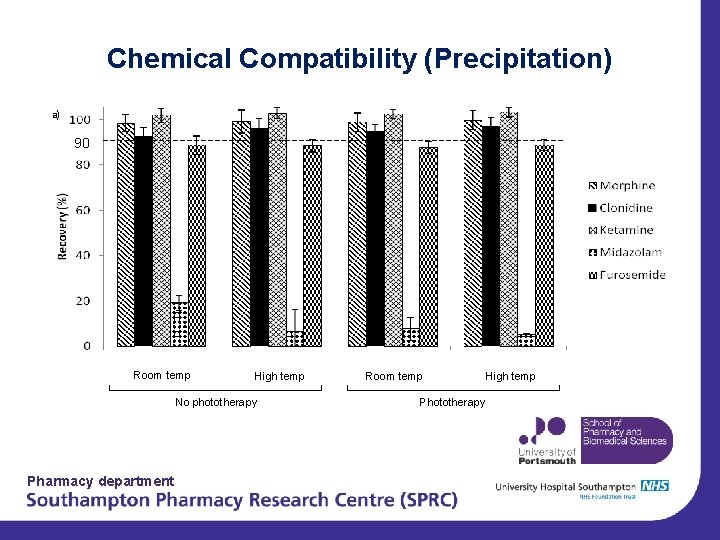
Chemical Compatibility (Precipitation) a) 90 Room temp High temp No phototherapy Pharmacy department Room temp Phototherapy High temp

Summary of Initial Results • No noticable effect on stability from the environmental conditions tested • All combinations of morphine, clonidine, ketamine and midazolam are fully compatible. • Some treatment combinations containing furosemide are compatible and some not. – Mo + Fu(9), Cl + Fu(12) and Mo, Cl and Fu(18) • Further investigation into effect of change of p. H after mixing of drugs – Depends on initial formulation p. H Pharmacy department

IV Formulation p. H Range of Drugs Min Max Drug allowed p. H Morphine 2. 5 a 6. 5 a Clonidine 4 b 7 b Ketamine 3. 5 b 5. 5 b Midazolam 2. 9 b 3. 7 b Furosemide 8 b 9. 2 b a) Value for Astramorph from Astrazeneca b) BP specified limit Pharmacy department

p. H Threshold Compatibility Study • p. H of each supplied formulation was adjusted to the extreme formulation p. H allowed. • Drugs were mixed in various combinations covering entire study range – 10 µL of another drug at extreme p. H added to “sensitive” drug – Solutions were observed for precipitation. Pharmacy department

p. H Threshold Compatibility Study Results • All combinations are compatible apart from: – Combination 9: • Morphine and furosemide INCOMPATIBLE at extreme formulation p. H – Combination 12: • Clonidine and furosemide INCOMPATIBLE at extreme formulation p. H – Combination 18: • Mixture of morphine, clonidine and furosemide compatible, however clinically this is very risky therefore INCOMPATIBLE. Pharmacy department

The Ketamine “Mystery” • Ketamine in short supply – Initial experiments: 10 mg/m. L Ketalar® precipitation – Later experiments: 100 mg/m. L Ketalar® diluted to 10 mg/m. L no precipitation • Benzethonium chloride Pharmacy department

Summary • No noticable influence from the environmental factors • All drug combinations which do not contain furosemide are compatible • Furosemide should not be mixed with any of these drugs – – p. H effect Presence of preservative Visible precipitation Reduction of intial concentration of one or more drugs in combination below 90 % Pharmacy department

Conclusions • Avoidance of unnecessary additional line insertion • Balance of Risk vs Benefit is better informed • Caution with furosemide is justified • Beware extrapolation between products and effect of excipients Pharmacy department

Future Work • Analysis of survey to select next drug combinations of interest • Numerous potential combinations • Inotropes next • Funding applications to support further work • University of Portsmouth invested in more HPLC equipment to support work Pharmacy department

Thank You • NPPG • Respondents to online survey Karen Ball, Luke Dickson, Marissa van der Merwe, Caroline Cole, John Pappachan, Adam Hocking, Andy Fox, Amanda Bevan Pharmacy department
- Slides: 18