Chemical Analysis Lesson Aims To be able to

Chemical Analysis

Lesson Aims Ø To be able to test for ammonium salts (and ammonia) Ø To recap the tests for carbon dioxide, hydrogen, and oxygen from Y 10 Ø To learn how to test for sulphate ions Ø To recall the method of flame testing and use it to identify ions (Ca 2+, Cu 2+, K+, Na+) Ø To recall the test for halide ions and carbonate ions

Testing for ammonium ions/ammonia • Put some of the solid in the base of the test tube • Pipette some Na. OH to the bottom of the test tube (NB It must not touch the sides of the test tube or you will get a false positive) • Wet some red litmus paper and hang it over the end of the test tube • Gently heat the test tube over a Bunsen burner

Gas tests • React some calcium carbonate with acid, bubble the gas through lime water • React magnesium with hydrochloric acid, test the gas with a lighted splint • React some hydrogen peroxide with some manganese dioxide, test the gas with a glowing splint

Testing for sulphate ions • Dissolve a tiny amount of your solid in a small amount of water (shake using a bung) • Add a few drops of barium chloride • If sulphate is present a white precipitate forms

Testing for Positive Ions - Flame tests 1. Clean your loop in a blue flame until no colour is seen 2. Dip your loop in acid to wet it 3. Pick up a few crystals on the loop 4. Put into the blue Bunsen flame and observe the colour NB Look at the first colour you see

Testing for Negative Ions – Silver Nitrate • Carefully dissolve a small amount of solid in a millilitre of water. • Add a few drops of dilute nitric acid followed by a few drops of silver nitrate solution.
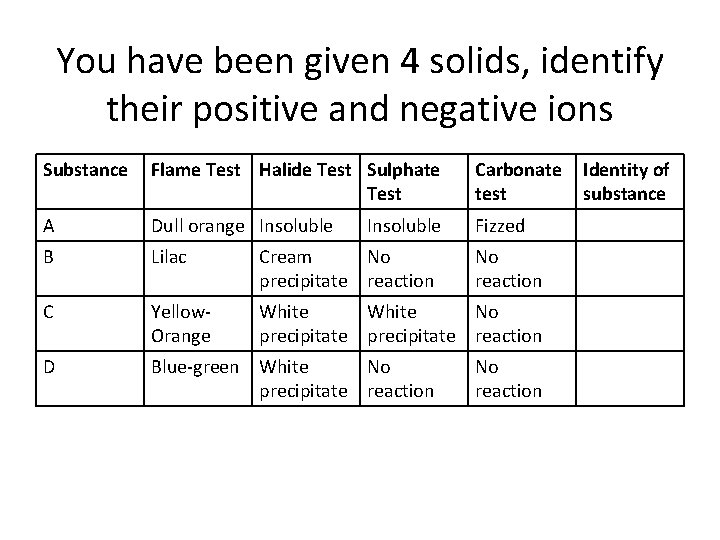
You have been given 4 solids, identify their positive and negative ions Substance Flame Test Halide Test Sulphate Test Carbonate test A Dull orange Insoluble Fizzed B Lilac Cream No precipitate reaction C Yellow. Orange White No precipitate reaction D Blue-green White No precipitate reaction Insoluble No reaction Identity of substance
- Slides: 8