CHEME 2130 Polymer Properties Steve Spoljaric Contact details












































![Mark-Houwink equation • Mark-Houwink equation is valid for broad molecular weight distribution: [ ] Mark-Houwink equation • Mark-Houwink equation is valid for broad molecular weight distribution: [ ]](https://slidetodoc.com/presentation_image/4a58fc80f2174540b6cf2803db96399c/image-63.jpg)


















- Slides: 91

CHEM-E 2130 Polymer Properties Steve Spoljaric Contact details: steven. spoljaric@aalto. fi Room E 424, Chemical Engineering Building (Kemsitintie 1)

Polymer Properties (5 credits) Learning outcomes: After completing the course, the student understands: 1) The basics of polymer physics and structure hierarchy of polymers 2) The basics of the most important methods of polymer analysis 3) Theories of polymer rheology 4) Stability and degradation of polymers 5) The ability to calculate the above mentioned phenomena

Topics covered: • Structure and average molecular weights • Solid-state properties: – Crystallinity – Thermal transitions • Characterization methods: – Thermoanalytical methods (DSC, DMTA) – Structure analysis (FTIR and Raman, NMR, fractionation, SEC/GPC) • • Rheological properties and their measurement Solubility and gas permeability Microscopy methods, X-ray analysis Stability and degradation

Schedule for the course • 6 Lectures, split into 2 sessions: – Mondays from 10: 15 -12: 00 (Ke 1/A 305) – Tuesdays from 8: 15 -10: 00 (U 4/142) • 5 Problem solving exercises on Thursdays from 8: 00 -10: 00 – Otakaari 1, Room U 4 / U 142 • Textbook for the course: Polymer Science and Technology, Fried JR.

Course assessment • Written examination (70 %) – 12 th December, 9: 00 -13: 00, D-Sali/Y 122 (Otakaari 1) • Problem solving exercises (30 %)

Lecture overview • Active learning / support from teaching staff balance • Broad spectrum of mateiral: – However, key concepts/learning outcomes will be highlighted at the start of each lecture – You will know what the crucial/important/relevant informaiton is • Lecture structure – Going-over and clarifying the course material – Visiting instruments and processing equipment • ’Hands-on’ experience where possible

Problem solving exercises • 5 problem solving exercises, every Thursday morning – All exercises are available on My. Courses • Exercises are submitted by Thursday 17: 00 for correction • You can complete them before hand, or come to the Thursday class and work there • Feel free to ask me and/or work with one another!

Lectures and corresponding book chapters (Fried) • Lecture I (Structure and Molecular Weight) – Chapters 1 and 3. 3 • Lecture II (Crystallinity) – Chapter 4 • Lecture III (Spectroscopic analysis techniques) – Chapters 2. 6 and 4 • Lecture IV (Rheology) – Chapters 5 and 11 • Lecture V (Solution fractionation and permeation) – Chapters 3. 2 and 12. 1 • Lecture VI (Stability, degradation and microscopy) – Chapter 6

Other recommended references • Spectroscopy of Polymers, Jack L. Koenig – Good for further information about spectrsocopy and characterisaton (Lecture III) • Polymer Physics, Ulf Gedde – A good all-round textbook, includes some information about microscopy (Lecture VI)

Questions? 1. Have you completed the course ‘Polymeerien valmistus’ (Polymer Synthesis)? 2. For those who haven’t completed Polymer Synthesis, have you completed any organic chemistry course? 3. What would you like to know more about regarding polymers? What do you think will be the topics in this course? 4. Any comments or wishes at this moment?

Polymer Structure

Key learning outcomes • Tacticity and isomers • Different molecular weights – How they influence polymer properties – How the can be determined – What needs to be known to determine them • Instrumentation used to determine molecular weight – Operating principles – Data obtained – Advantages and limitations

Skeletal (’line’) formula • • Every angle represents a carbon bond Hydrogens are not shown Single line is a single bond Atoms/functional groups added as needed = Solid lines represent bonds in the plane of the paper or screen Solid wedges represent bonds that point out of the plane of the paper or screen, towards the observer Dashed wedges or dashed lines (thick or thin) represent bonds that point into the plane of the paper or screen, away from the observer Wavy lines represent either unknown stereochemistry or a mixture of the two possible stereoisomers

Polymer Stereochemistry • Stereochemistry: Three-dimensional arrangement of atoms and molecules, and their effect on properties This consists of two distinct factors: • Configuration: – Relative position of atoms in a polymer that can only be changed by breaking and reforming primary chemical bonds • Conformation: – Different orientations of atoms and substituents in a molecule – Result from rotations around single bonds

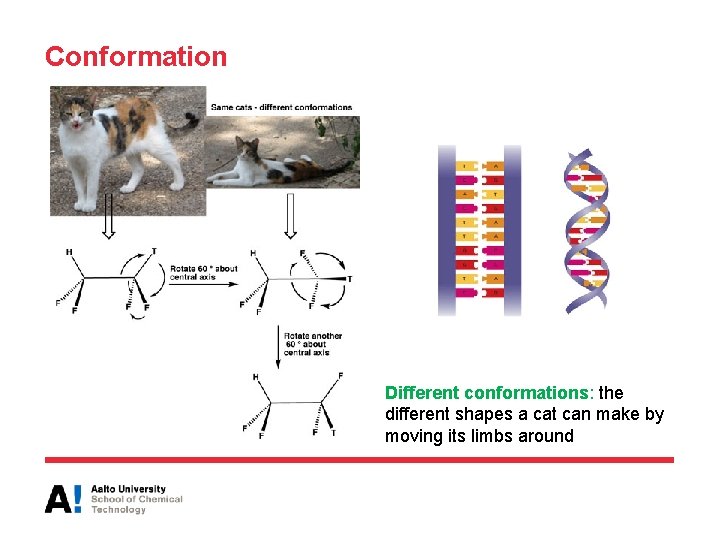
Conformation Different conformations: the different shapes a cat can make by moving its limbs around

Configuration Different configurations: the same number of limbs, but different order and locations where they are connected

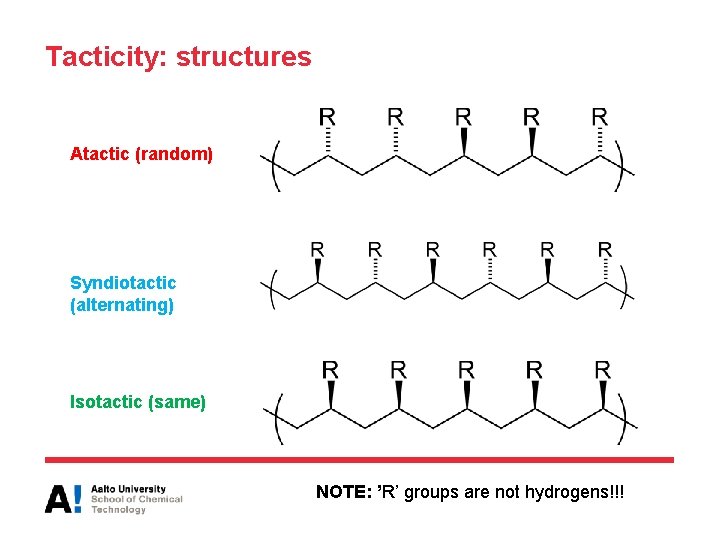
Tacticity • Stereochemical arrangement of units in the polymer main (backbone) chain: not including hydrogens • This determines the overall order of the polymer chain: – If the ‘R’ groups are randomly distributed on the two sides of the planar backbone, the polymer does not have order and is atactic • Ordered structures are isotactic and syndiotactic – When the sterocenter of each repeating unit has the same configuration, the structure is termed isotactic – When the stereocenter alternates from one repeating unit to the next, the structure is syndiotactic

Tacticity: structures Atactic (random) Syndiotactic (alternating) Isotactic (same) NOTE: ’R’ groups are not hydrogens!!!

Tacticity: influences • In general: – isotactic and syndiotactic polymers are partly crystalline – atactic are amorphous • Tacticity affects also other properties such as thermal and mechanical properties • The tacticity of a polymer can be controlled by several factors: – Polymerization temperature – Pressure – Catalyst – Solvent

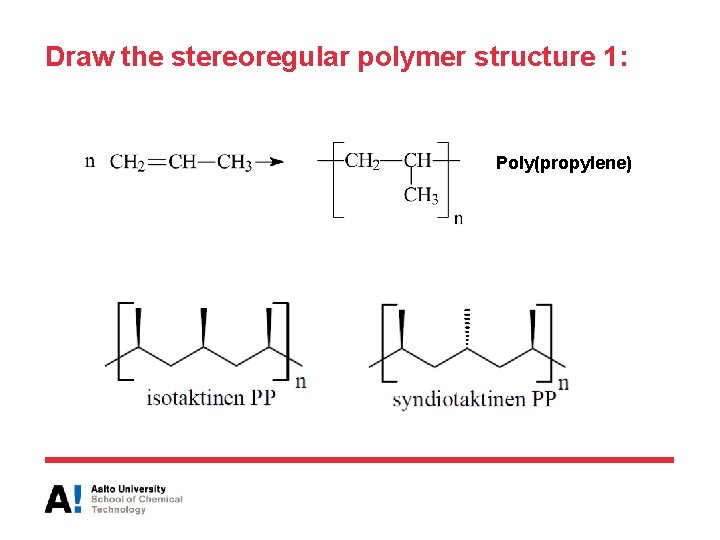
Draw the stereoregular polymer structure 1: Poly(propylene)

Tacticity in commercial polymers • Polypropylene (PP) used and sold is mostly isotactic but it is also possible to produce syndiotactic or atactic polymer Isotactic Atactic Syndiotactic

Draw the stereoregular polymer structure 2: Poly(isobutylene) No stereoregularity differences: • When there are two identical substituents on the same carbon • Polyethylene is the other exception

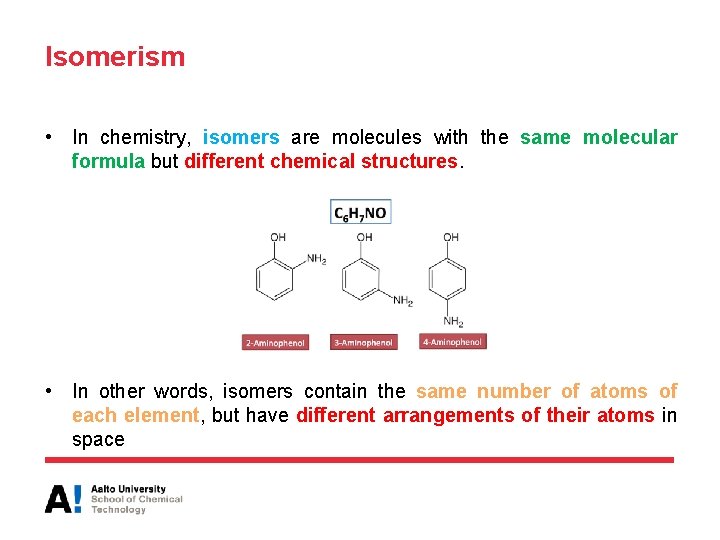
Isomerism • In chemistry, isomers are molecules with the same molecular formula but different chemical structures. • In other words, isomers contain the same number of atoms of each element, but have different arrangements of their atoms in space

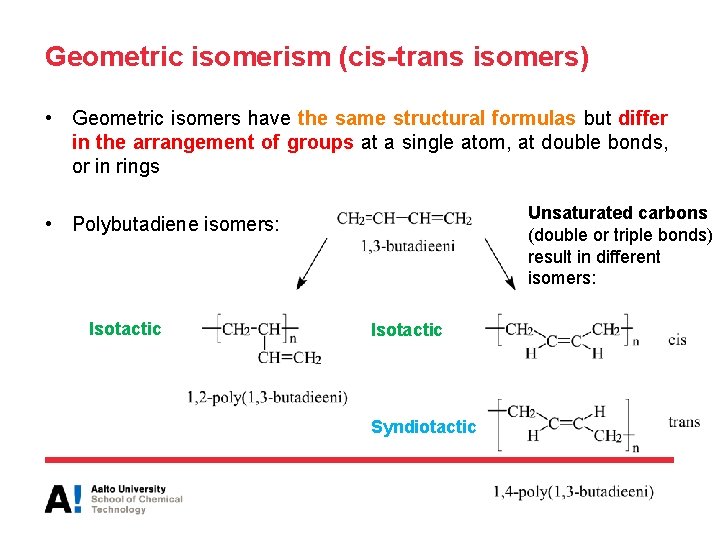
Geometric isomerism (cis-trans isomers) • Geometric isomers have the same structural formulas but differ in the arrangement of groups at a single atom, at double bonds, or in rings Unsaturated carbons (double or triple bonds) result in different isomers: • Polybutadiene isomers: Isotactic Syndiotactic

Optical isomerism (enantiomerism or chirality) • Optical isomers (also called enantiomers) are mirror images that are not superimposable (that is, they can’t be put on top of one another – the phenomenon is called chirality) • They differ in only one characteristic; their interaction with plane polarized light (specifically, the direction in which they rotate it) https: //www. youtube. com/watch? v=6 vd 7 S 3 T 7 R 8 k

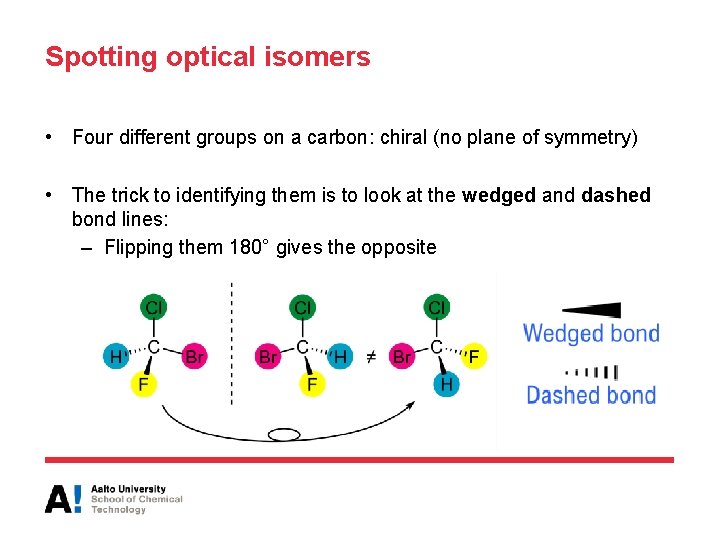
Spotting optical isomers • Four different groups on a carbon: chiral (no plane of symmetry) • The trick to identifying them is to look at the wedged and dashed bond lines: – Flipping them 180° gives the opposite

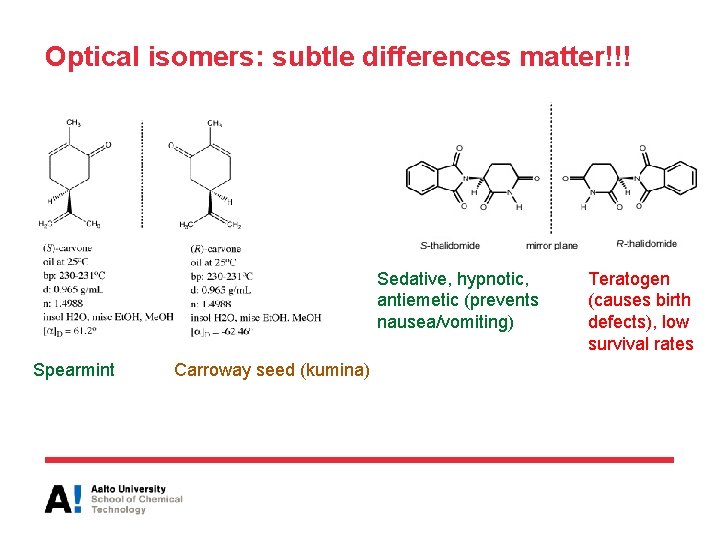
Optical isomers: subtle differences matter!!! Sedative, hypnotic, antiemetic (prevents nausea/vomiting) Spearmint Carroway seed (kumina) Teratogen (causes birth defects), low survival rates

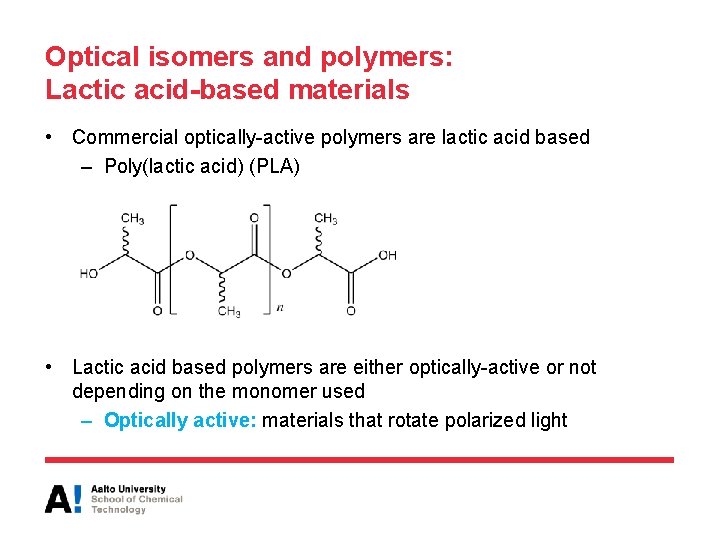
Optical isomers and polymers: Lactic acid-based materials • Commercial optically-active polymers are lactic acid based – Poly(lactic acid) (PLA) • Lactic acid based polymers are either optically-active or not depending on the monomer used – Optically active: materials that rotate polarized light

Optical isomers in polymers: applications • • Chromatographic/separation applications Catalysts Liquid crystals Microwave absorbents Membrane separation technology Optical switches Biomedical equipment Optoelectronics

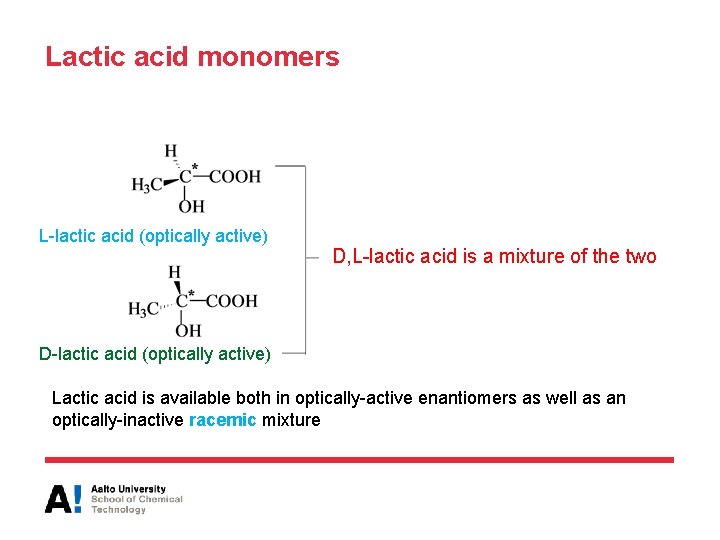
Lactic acid monomers L-lactic acid (optically active) D, L-lactic acid is a mixture of the two D-lactic acid (optically active) Lactic acid is available both in optically-active enantiomers as well as an optically-inactive racemic mixture

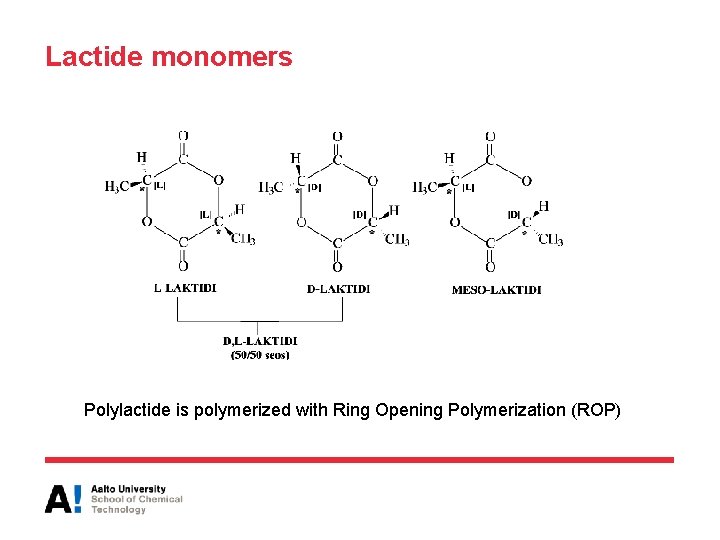
Lactide monomers Polylactide is polymerized with Ring Opening Polymerization (ROP)

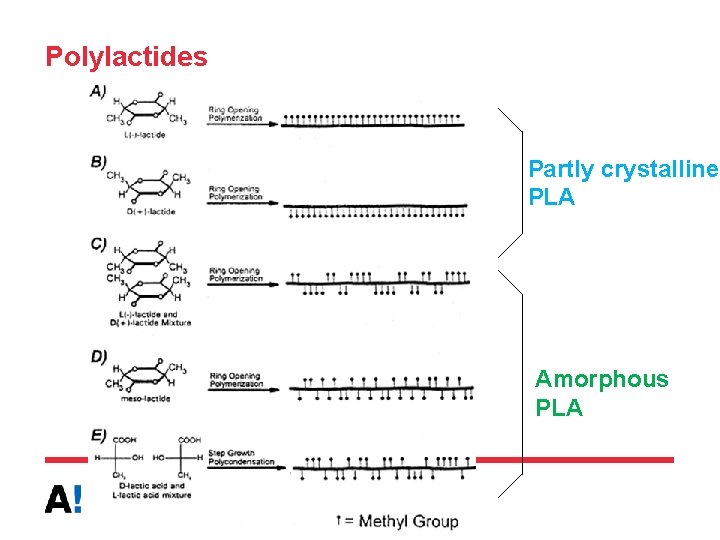
Polylactides Partly crystalline PLA Amorphous PLA

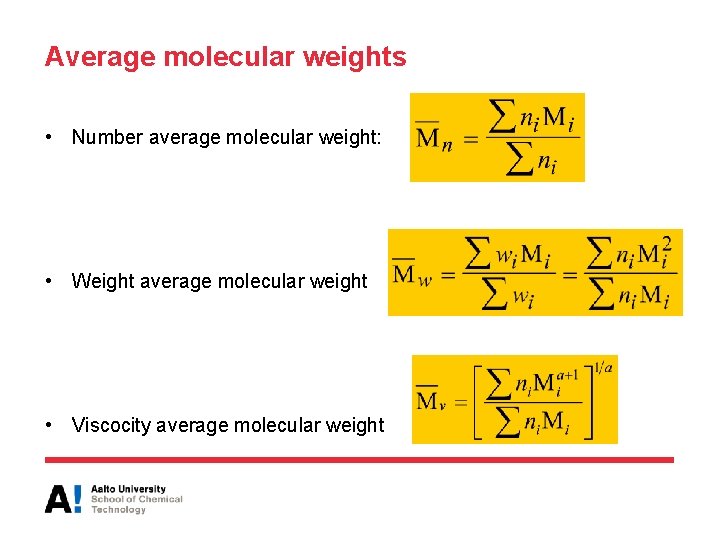
Average molecular weights

Molecular weight vs. molar mass • Molecular weight is the mass of one molecule of a substance • Usually provided as an average distribution: – Following polymerisation, polymer chains rarely have the same degree of polymerisation (number of repeating monomer units) and molar mass • Molar mass is the mass of one mole of a substance

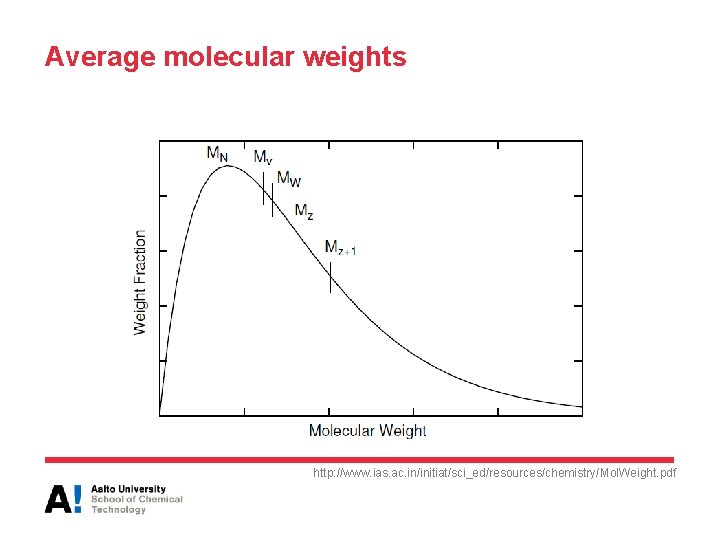
Average molecular weights http: //www. ias. ac. in/initiat/sci_ed/resources/chemistry/Mol. Weight. pdf

Average molecular weights • Number average molecular weight: • Weight average molecular weight • Viscocity average molecular weight

Average molecular weights • Z-average molecular weight (Z = zentrifuge/centrifuge) • Polydispersity – If PDI = 1, the polymer is monodisperse ni = Number of molecules with molecular weight Mi wi = weight fraction with molecular weight Mi a = constant, depends on polymer/solvent combination

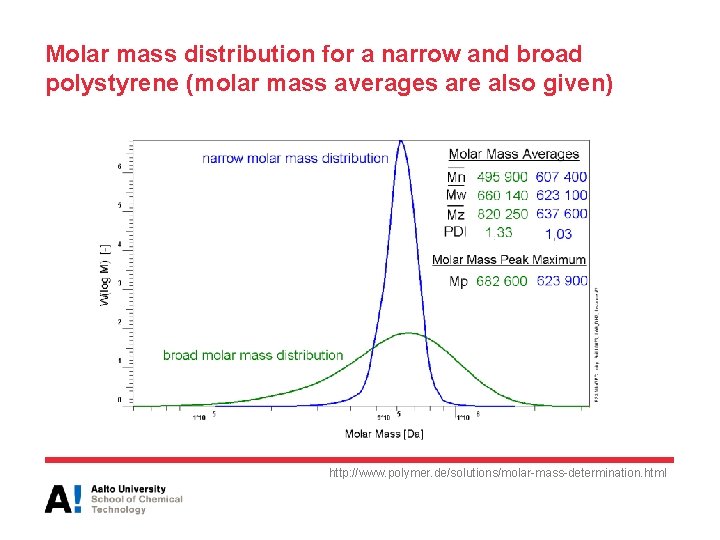
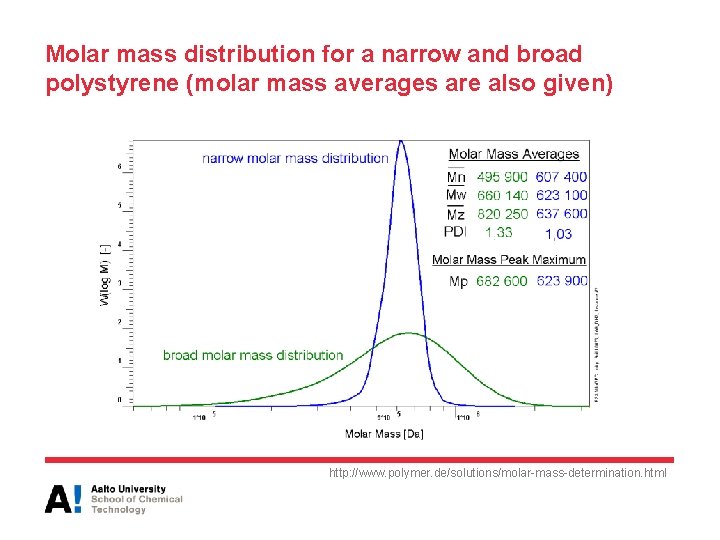
Molar mass distribution for a narrow and broad polystyrene (molar mass averages are also given) http: //www. polymer. de/solutions/molar-mass-determination. html

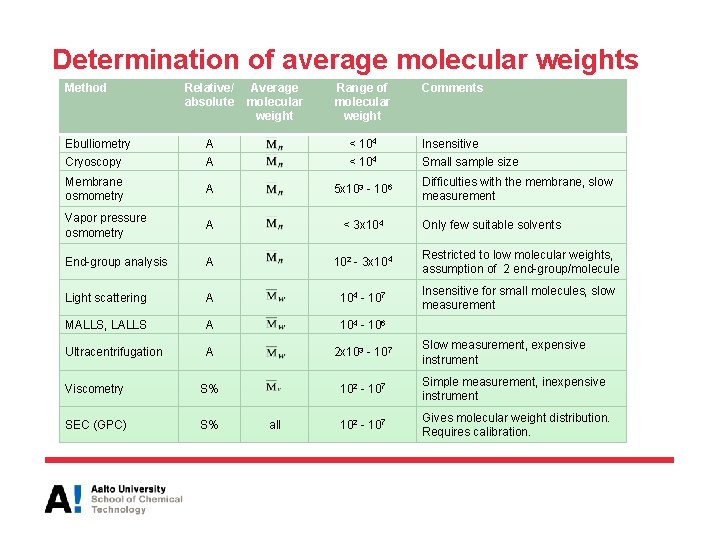
Determination of average molecular weights • Primary (absolute values) methods: – Osmometry (Mn) – Scattering (Mw) – Sedimentation (Mz) Z-average molecular weight is obtained from centrifugation data • Secondary (relevant to reference or calibration) methods: – Gel permeation chromatography (GPC), also called size exclusion chromatography (SEC) to obtain molecular weight distribution – Intrinsic viscosity for determining viscosity average molecular weight

Determination of average molecular weights Method Relative/ absolute Average molecular weight Range of molecular weight Comments Ebulliometry A < 104 Insensitive Cryoscopy A < 104 Small sample size Membrane osmometry A 5 x 103 - 106 Vapor pressure osmometry A < 3 x 104 End-group analysis A 102 - 3 x 104 Restricted to low molecular weights, assumption of 2 end-group/molecule Light scattering A 104 - 107 Insensitive for small molecules, slow measurement MALLS, LALLS A 104 - 106 Ultracentrifugation A 2 x 103 - 107 Viscometry S% 102 - 107 Simple measurement, inexpensive instrument SEC (GPC) S% 102 - 107 Gives molecular weight distribution. Requires calibration. all Difficulties with the membrane, slow measurement Only few suitable solvents Slow measurement, expensive instrument

Effect of molecular weight on polymer properties

Properties dependent on Molecular Weight and Molecular Weight Distribution • • • Melt Viscosity Tensile Strength Toughness or Impact Strength Resistance to heat Corrosive properties Polymer Monodisperse (uniform) Polydisperse (non -uniform) Proteins, nucleic acids All synthetic polymers

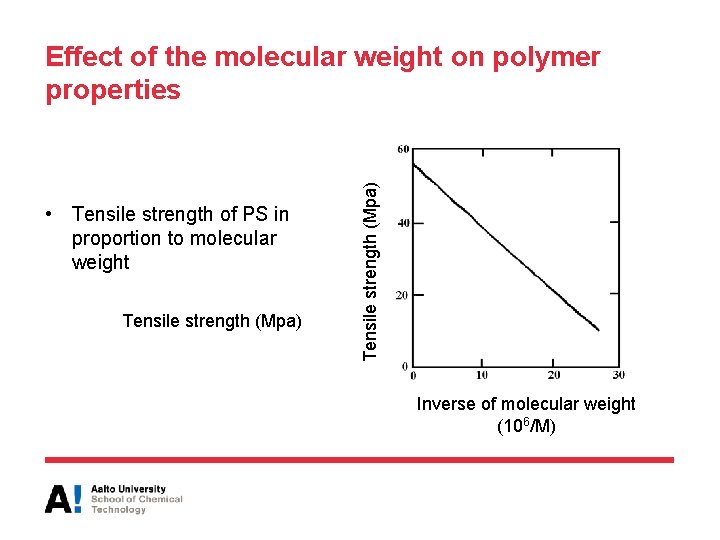
Effect of the molecular weight on polymer properties • Huge effect on properties!!! – Some properties have a limit (eg. Tg and strength at break) – Some can change without any limit • Dependence of glass transition temperature (Tg) on number average molecular weight (Mn): - A and B are experimental constants - Tg, is glass transition temperature of a extremely high molecular weight polymer

Effect of the molecular weight on polymer properties • Many mechanical properties exhibit a similar dependence on molecular weight: P = property value (strength, toughness, stiffness …) K’ = empirical constant Mn = number average molecular weight

• Tensile strength of PS in proportion to molecular weight Tensile strength (Mpa) Effect of the molecular weight on polymer properties Inverse of molecular weight (106/M)

Effect of molecular weight and PDI on properties • Examples of how average molecular weight and molecular weight distribution affect different properties: Property Molecular weight increases Molecular weight distribution narrows Tensile strength + + Tensile strain + - Strength at yield + - Ductility + + Brittleness + - Hardness + - Softening point + + Melt viscosity + + Adhesion - - Chemical endurance + + Solubility - o Property: + = improves - = worse o = little effect

Number average molecular weight

Number average molecular weight • Determination methods based on various phenomena or factors: – End groups on polymer backbone – Property changes • Osmotic pressure • Increase in boiling point • Decrease in freezing point or vapour pressure

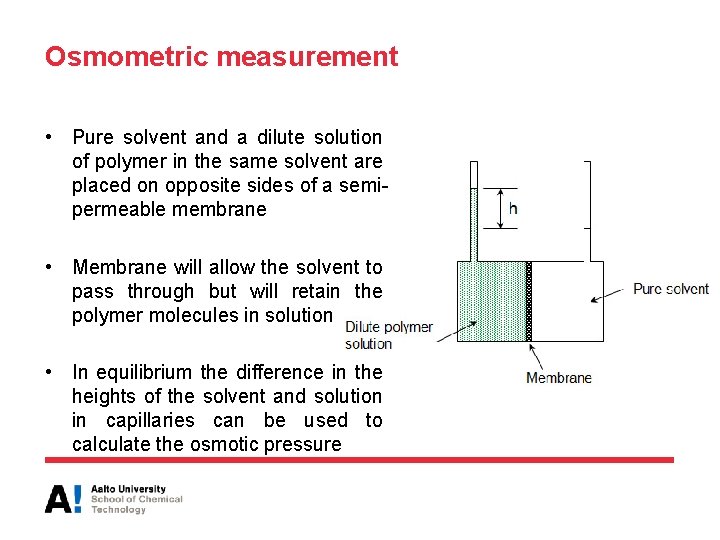
Osmometric measurement • Pure solvent and a dilute solution of polymer in the same solvent are placed on opposite sides of a semipermeable membrane • Membrane will allow the solvent to pass through but will retain the polymer molecules in solution • In equilibrium the difference in the heights of the solvent and solution in capillaries can be used to calculate the osmotic pressure

Osmometric measurement • Van’t Hoff equation for the osmotic pressure of an ideal, dilute solution: p = osmotic pressure c = concentration R = gas constant 8. 314 J/mol/K T = temperature (K) = number average molecular weight (g/mol) g = gravitational constant 9. 80665 m/s 2 ρ = solvent density

Osmometric measurement • Van’t Hoff equation is for ideal, dilute solutions. In real solutions the equation will be following: B, C are virial coefficients • For the determination of molecular weight, 4 -6 pressure measurements with different concentrations are required. When solutions are dilute enough, p/c can be obtained by extrapolation of c to 0. Average molecular weight can be calculated from: • Polymer concentration is g/dm 3 and p/c in J/kg

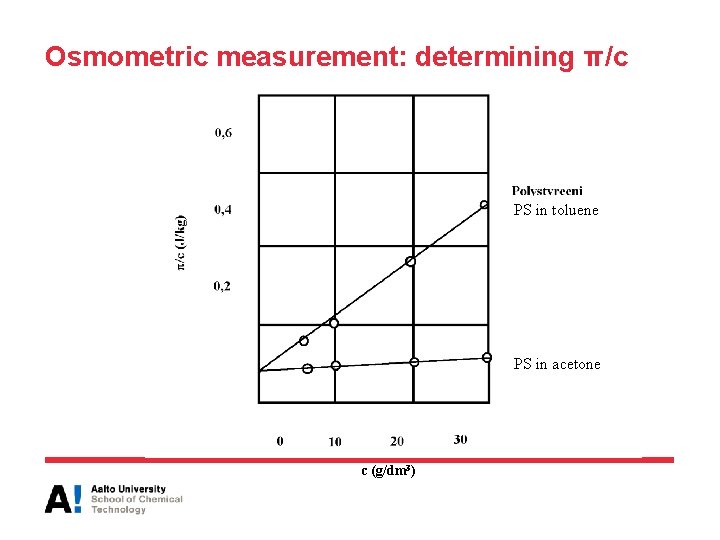
Osmometric measurement: determining π/c PS in toluene PS in acetone c (g/dm 3)

Osmometric measurement: challenges • Simple experimental procedure, but can be very time consuming • Performance of the membrane can be a problem • Membrane can let some smaller polymer molecules through and this will result in an artificially-higher Mn value • Thus, the method is considered accurate for molecular weights above 20, 000 g/mol • The upper limit for molecular weight is 500, 000 g/mol due to inaccuracy in measuring small osmotic pressures

Vapor-pressure osmometry • When a polymer is added to a solvent, the vapor pressure of the solvent is lowered due to the decrease in solvent activity • Osmometry uses two matched thermistors that are in a closed chamber containing saturated solvent vapor • A drop of solvent and a drop of dilute polymer solution is placed on thermistors • Condensation of solvent vapor onto the solution causes the temperature of the solution thermistor increase until vapor pressure of solution equals to that of a solvent

Vapor-pressure osmometry: measurement principles Closed chamber Pure solvent • For the characterization of low molecular weight oligomers and polymers, vapor-pressure osmometry is recommended

Vapor-pressure osmometry cont. • The difference in temperatures is recorded in terms of difference in resistance DR, which is calibrated by use of a standard low molecular weight sample • Several polymer concentrations are measured and extrapolation of DR/c to zero concentration yield Mn (requires calibration constant obtained with the standard sample hence VPO is secondary method)

Ebullioscopic method • The determination of molecular weight, in which the differential thermometer is used to measure the rise in boiling point • Boiling point of the polymer solution is compared to the boiling point of the solvent. An increase in boiling point is comparable to polymer properties: Tb = increase in Tb Kb = ebullioscopy constant WA = mass of the polymer WS = mass of the solvent • Top limit of the measurement is 50 000 g/mol

Cryoscopic method • Resembles ebullioscopy method • Compares the freezing point of a polymer solution and pure solvent • For dilute solutions: Kf = cryoscopic constant • Top limit of the measurement is 50 000 g/mol

End group method • The structure of the polymer chain must be known!!! • End groups to be either quantitatively determined with appropriate reagents (titration) or determined by IR spectroscopy or NMR • IR spectroscopy: Number average molecular weight can be calculated assuming the chain is linear with no branch points – Number of end groups of the polymer is thus one or two per molecule – End groups are not spectroscopically coupled to the remainder of the chain, low molecular weight analogs can be used for calibration – Particularly useful for insoluble systems (eg. fluorocarbons)

Viscosity average molecular weight

Viscosity average molecular weight • Widely used for routine molecular weight determination – Cheap equipment and simple measurement procedure • In this method, the viscosities of pure solvent and dilute polymer solutions are compared • To determine the molecular weight, the empirical constants K and a, that are specific for a given polymer, solvent and temperature, need to be known – Extensive libraries of the values for constants exist for different polymer solvent combinations

Staudinger equation • Staudinger equation is valid only for a relatively narrow molecular weight distribution sp = specific viscosity c = solution concentration Km = constant for specific polymer-solvent combination
![MarkHouwink equation MarkHouwink equation is valid for broad molecular weight distribution Mark-Houwink equation • Mark-Houwink equation is valid for broad molecular weight distribution: [ ]](https://slidetodoc.com/presentation_image/4a58fc80f2174540b6cf2803db96399c/image-63.jpg)
Mark-Houwink equation • Mark-Houwink equation is valid for broad molecular weight distribution: [ ] KM, a = intrinsic viscosity = viscosity average molecular weight = constants for particular polymer-solvent combination For flexible polymer chains a is 0. 5 - 0. 8, for stiff and rod like chains a is 2. 0.

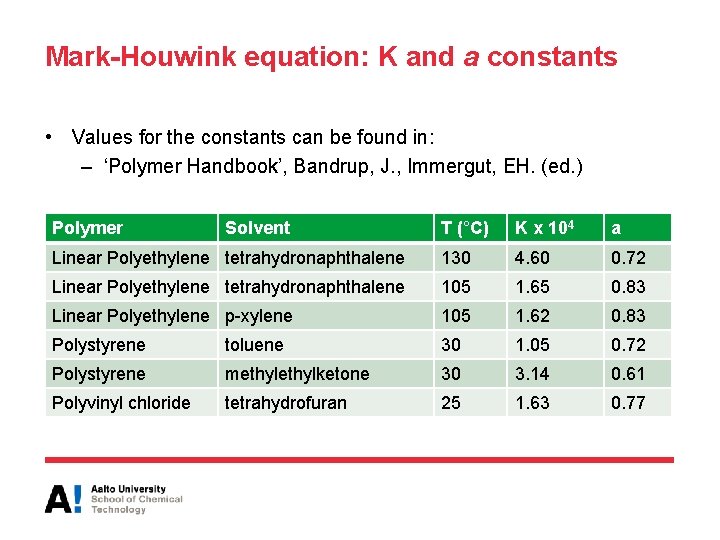
Mark-Houwink equation: K and a constants • Values for the constants can be found in: – ‘Polymer Handbook’, Bandrup, J. , Immergut, EH. (ed. ) Polymer Solvent T (°C) K x 104 a Linear Polyethylene tetrahydronaphthalene 130 4. 60 0. 72 Linear Polyethylene tetrahydronaphthalene 105 1. 65 0. 83 Linear Polyethylene p-xylene 105 1. 62 0. 83 Polystyrene toluene 30 1. 05 0. 72 Polystyrene methylketone 30 3. 14 0. 61 Polyvinyl chloride tetrahydrofuran 25 1. 63 0. 77

Viscosity parameters Capillary viscometers are used to carry out the measurement: – Ostwald-Fenske or Ubbelodde type capillary – Constant temperature, typically 25 or 30°C – The time required for a dilute solution and pure solvent to pass through the capillary is measured – 4 -6 polymer solutions are measured – Efflux times need to be more than 100 s, which can then be used to calculate the relative viscosity: t = efflux time for dilute polymer solution T 0 = efflux time for pure solvent = dynamic viscosity of the solution 0 = dynamic viscosity of the solvent

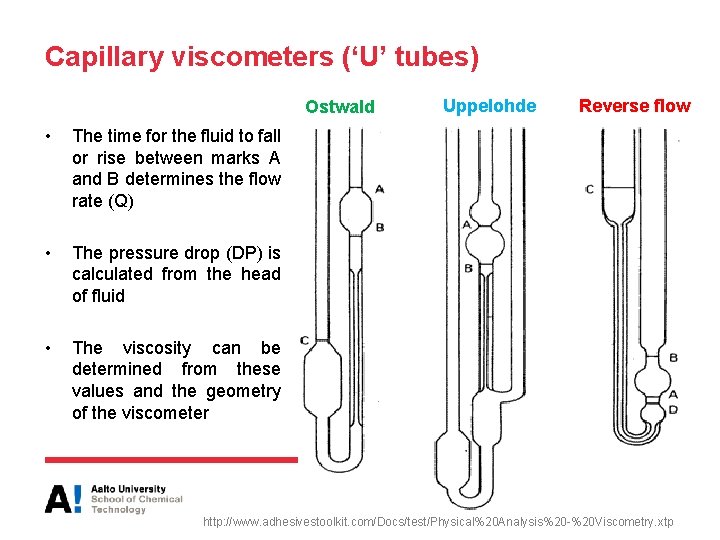
Capillary viscometers (‘U’ tubes) Ostwald • The time for the fluid to fall or rise between marks A and B determines the flow rate (Q) • The pressure drop (DP) is calculated from the head of fluid • The viscosity can be determined from these values and the geometry of the viscometer Uppelohde Reverse flow http: //www. adhesivestoolkit. com/Docs/test/Physical%20 Analysis%20 -%20 Viscometry. xtp

Viscosity parameters • Specific viscosity can be calculated from viscosities or the efflux times: • The viscosity number or reduced viscosity is obtained by dividing specific viscosity by the solution concentration: Molecular weight determination, Rheological measurements in Polymers: Polymer Characterization and Analysis, Ed. J. I. Krowchwitz, John Wiley & Sons Inc, 1990

Viscosity parameters • Logarithmic viscosity number, inherent viscosity: • Limiting viscosity number, intrinsic viscosity is obtained from the parameters of logarithmic viscosities

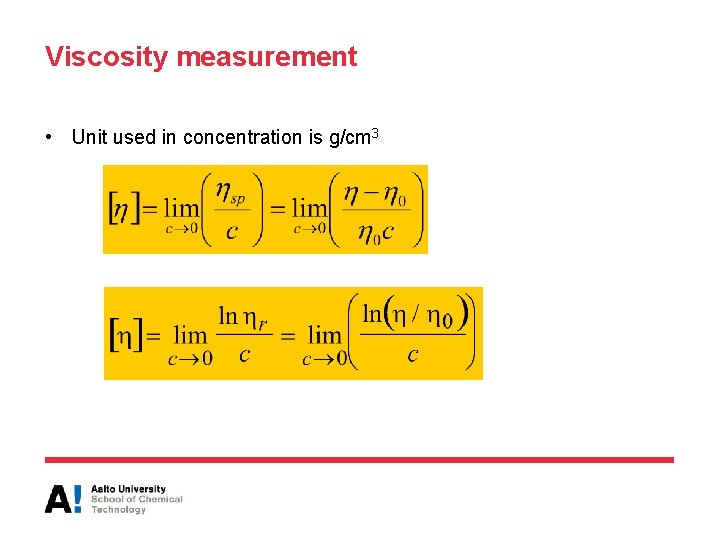
Viscosity measurement • Unit used in concentration is g/cm 3

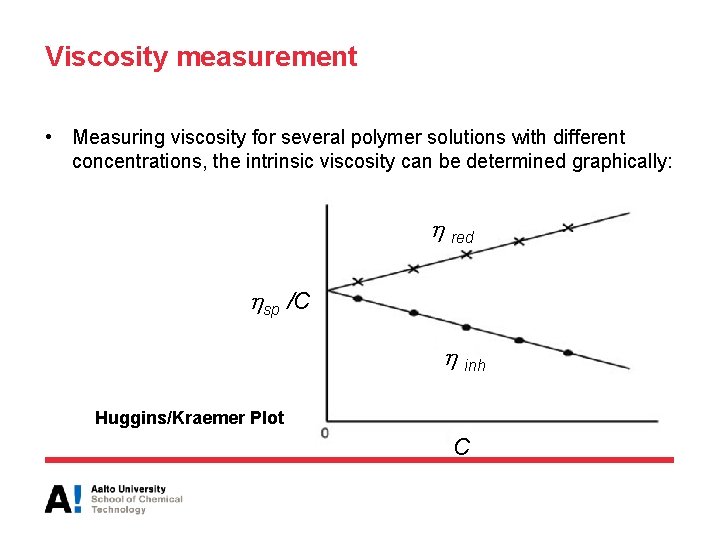
Viscosity measurement • Measuring viscosity for several polymer solutions with different concentrations, the intrinsic viscosity can be determined graphically: red sp /C inh Huggins/Kraemer Plot C

Weight average molecular weight

Light scattering in nature • Why is the sky blue? • How can we see clouds? incident beam scattered light • When light passes through matter, most of the light continues in its original direction but a small fraction is scattered in other directions

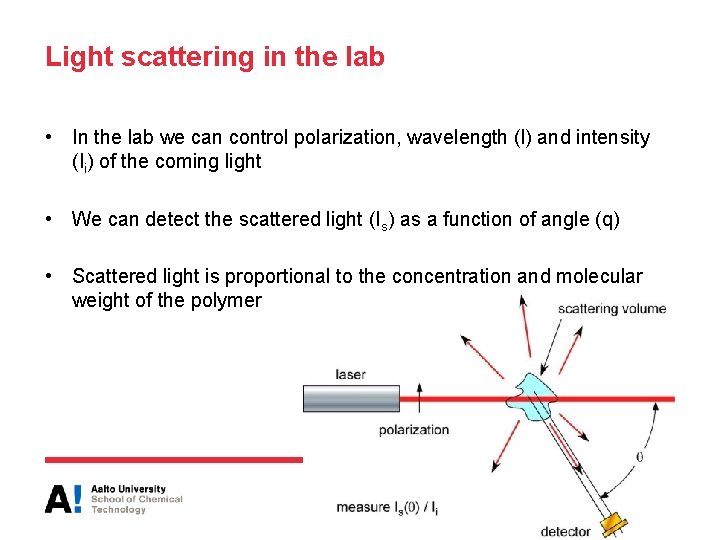
Light scattering in the lab • In the lab we can control polarization, wavelength (l) and intensity (Ii) of the coming light • We can detect the scattered light (Is) as a function of angle (q) • Scattered light is proportional to the concentration and molecular weight of the polymer

Light scattering • The weight average molecular weight can be obtained directly only by scattering experiments • Most commonly used is light scattering from dilute polymer solution • Polymer molecules in solution cause stronger scattering than solvent molecules – The intensity of the light is proportionate to the absolute molecular weight of the particle causing the scattering

Light scattering procedure • Polymer is dissolved in appropriate solvent • The particles have interactions in solution and self organize – Local changes in concentration and density in solution • Light scattering is observed in different angles • Measurements with several concentrations and angles

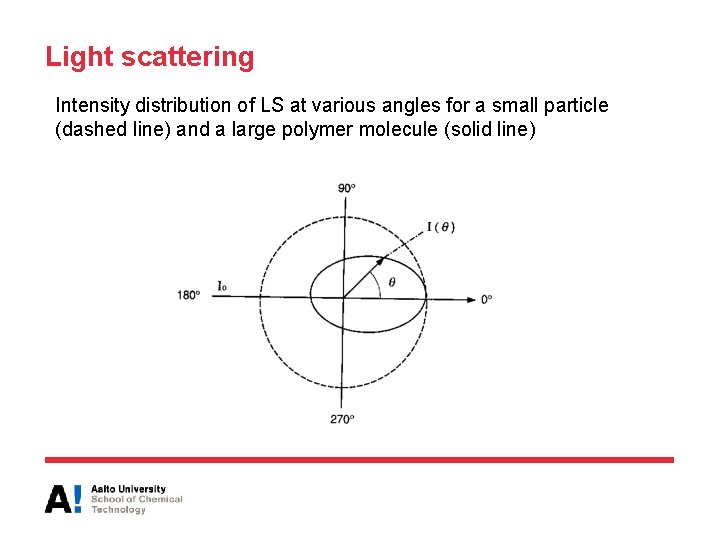
Light scattering Intensity distribution of LS at various angles for a small particle (dashed line) and a large polymer molecule (solid line)

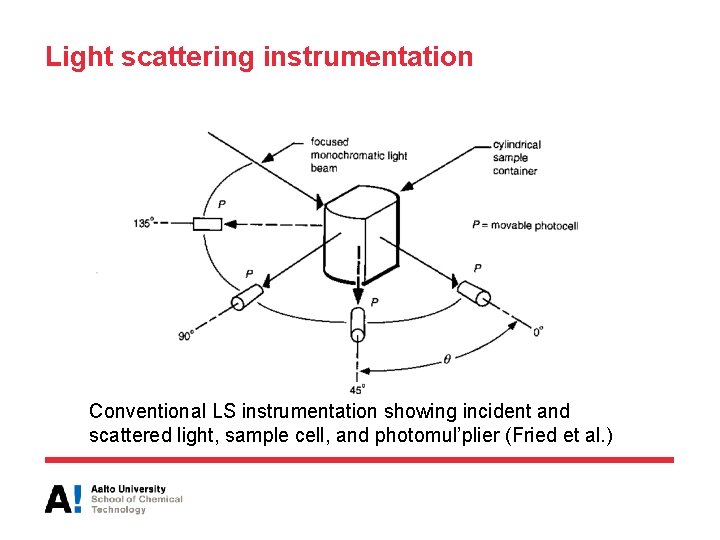
Light scattering instrumentation Conventional LS instrumentation showing incident and scattered light, sample cell, and photomul’plier (Fried et al. )

Light scattering In order to determine molecular weight, the following information must be known/determined: • • Refractive index for the solvent Difference between refractive indices for the solution and solvent Polymer concentration Absolute light intensity for certain scattering angles

Light scattering • Rayleigh formula: n 0 = refractive index for solvent l = wavelength of the light N 0= Avogadro number Rb= Rayleigh constant I 0= light intensity at 90 o angle that scattered from solvent c = polymer concentration in solution = measuring angle R = measured intensity, contains the angle and correction for concentration • Problem is determining R( ) value, which should be known for all values

Low-Angle Laser Light scattering (LALLS) • Helium-Neon lasers have replaced conventional light sources in scattering instruments • The high intensity of the light sources permits measurements at much smaller angles (2– 10 o). Then, the scattering by small spherical particles can be expressed: • Only at one single angle chain dimensions cannot be obtained

Multi-Angle Laser Light-Scattering (MALLS) • Several detectors (18 angles, 20– 150 o ) • Absolute molar mass (Mn, Mw, - Mz) and size (Rz , rh) • No standard references, more reliable than calibration techniques – Information about molecules and particles (conformation, branching, partly cross-linked fractions) – Co-polymer, protein, aggregate detection – Easy and fast to use

DAWN 8+ • 8 angles (23 -155 °) • Molar mass range <10³-107 g/mol • Molecular size range 10 -500 nm • Laser wavelength 658 nm – (50 m. W Ga. As linear polarized laser) • All solvents compatible (aqueous and organic) incident beam photodiode detectors

Gel permeation chromatography (GPC ) aka: Size-exclusion chromatography (SEC)

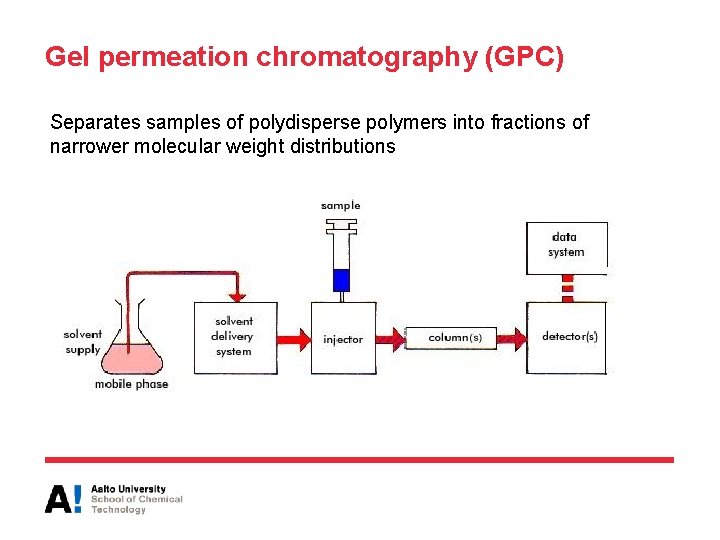
Gel permeation chromatography (GPC) • GPC is the most widely used technique for the analysis of polymers • Can be used for samples soluble in organic and aqueous eluents and molecular weights from approximately 100 to several million • Yields all molecular weight averages and the molecular weight distribution • Room temperature GPC or operating at higher temperature (for polyolefins 120 ᵒC)

Gel permeation chromatography (GPC) Separates samples of polydisperse polymers into fractions of narrower molecular weight distributions

Principle of GPC: The largest fractions of polymer come through the column first Polystyrene gel Polymer molecules

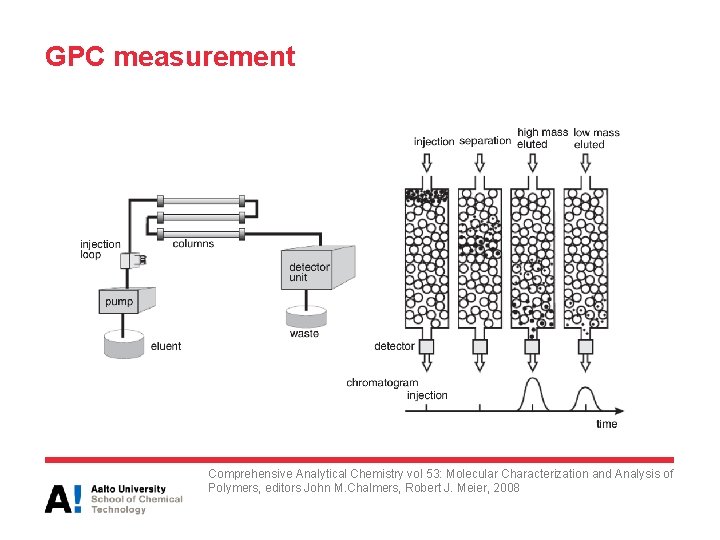
GPC measurement • Columns are packed with small, highly porous beads or gel – Pore diameters of the beads range from 10 to 107 Å – Approximately the dimensions of polymer molecules in solution • During GPC operation, pure pre-filtered solvent is continuously pumped through the columns at a constant flow rate. Then a small amount of dilute polymer solution is injected into the solvent stream and carried to through the columns • Polymer molecules diffuse from this mobile phase into the stationary phase in the pores. The smallest polymer molecules penetrate deeply into the pores whereas the largest molecules pass through the columns first

GPC measurement Comprehensive Analytical Chemistry vol 53: Molecular Characterization and Analysis of Polymers, editors John M. Chalmers, Robert J. Meier, 2008

GPC measurement • The concentration of polymer molecules in each eluting fraction is monitored by a detector, such as IR • The elution volumes are calibrated with know Mn standards (PS and PMMA standards are most common) • Only apparent molecular weights are determined unless: – Molecular weight standards exist of the same composition and topology as the samples – LALLS or viscometry is used – Mark–Houwink parameters of the standards and sample are known

Molar mass distribution for a narrow and broad polystyrene (molar mass averages are also given) http: //www. polymer. de/solutions/molar-mass-determination. html

Next week: Crystallinity Thermal transitions