Chem E 260 Entropy Balances On Open and

Chem. E 260 Entropy Balances On Open and Closed Systems Dr. William Baratuci Senior Lecturer Chemical Engineering Department University of Washington TCD 8: A & B CB 6: 10 + Supplement May 10, 2005

Entropy Balance: Closed System • 1 st Law: • 2 nd Law, Internally Reversible Processes: • Boundary Work, Internally Reversible Processes: • Gibbs 1 st Equation: • Entropy Balance Equation – Integral Form: – Differential Form: – Rate Form: Baratuci Chem. E 260 May 10, 2005 (Usually assume Wtot = Wb. )

Entropy Balance: Open System • General: • Steady-state, SISO: • Entropy generation within the system: Baratuci Chem. E 260 May 10, 2005

The • st 1 Law and Entropy 1 st Law, Steady-state, Internally Reversible, SISO: • From the definition of entropy: • Gibbs 2 nd Equation: • Integrating for an open system: Baratuci Chem. E 260 May 10, 2005

Mechanical Energy Balance Equation • Combine all the equations from the previous slide: • The MEBE: • If Wnot b = 0 : Bernoulli Equation (incompressible fluid) • If Ekin = Epot = 0 : Baratuci Chem. E 260 May 10, 2005 Usually, Wnot b = Wsh
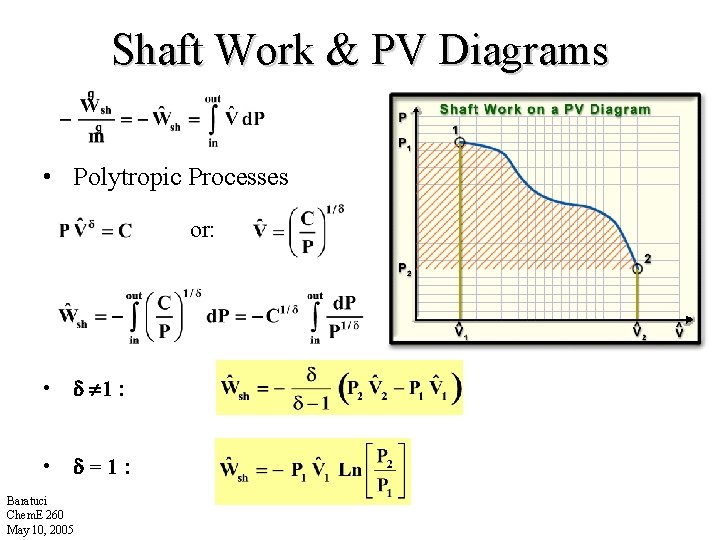
Shaft Work & PV Diagrams • Polytropic Processes or: • 1 : • =1: Baratuci Chem. E 260 May 10, 2005
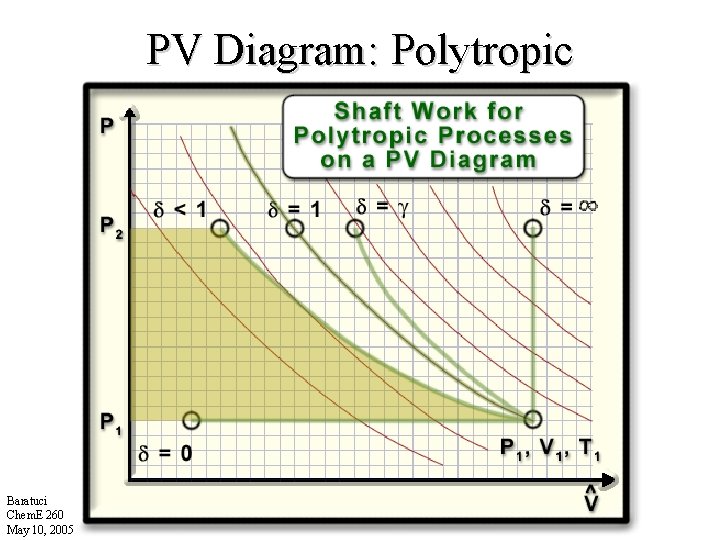
PV Diagram: Polytropic Baratuci Chem. E 260 May 10, 2005

Summary: Wsh Polytropic Processes Process Type Real Fluids Ideal Gases =0 =1 Isothermal 1: Polytropic = = constant = : Isochoric Baratuci Chem. E 260 May 10, 2005 Isentropic • Wsh is actially all work other than flow work.

Next Class … • Problem Session • After that… – Isentropic Efficiency • Define an efficiency for a process by comparing actual performance to the performance of an isentropic process • Nozzles, compressors and turbines • New diagram: HS Diagram – Multi-Stage Compressors • Intercooler HEX’s reduce work input requirement Baratuci Chem. E 260 May 10, 2005

Example #1 • Air is compressed from 1 bar and 310 K to 8 bar. Calculate the specific work and heat transfer if the air follows a polytropic process path with = 1. 32. Assume air is an ideal gas in this process. Baratuci Chem. E 260 May 10, 2005

Example #2 • A turbine lets down steam from 5 MPa and 500 o. C to saturated vapor at 100 k. Pa while producing 720 k. J/kg of shaft work. The outer surface of the turbine is at an average temperature of 200 o. C. Determine the heat losses from the turbine and the entropy generation in the turbine in k. J/kg-K. Baratuci Chem. E 260 May 10, 2005
- Slides: 11