Chem Bio Pharm Innovative combination of Quality by

Chem. Bio. Pharm Innovative combination of Quality by Design and green analytical chemistry for analytical methods in pharmaceutical sciences Ludivine Ferey, Karen Gaudin Laboratory of Analytical Chemistry – Faculty of Pharmaceutical Sciences – Bordeaux Chem. Bio. Pharm – ARNA INSERM U 1212 / UMR CNRS 5320 Industrial partner Christine Boussès Unither Développement Bordeaux 1

REGULATORY REQUIREMENTS (1/2) Chem. Bio. Pharm § ICH Q 8(R 2): pharmaceutical development Quality by Design (Qb. D) Q 8(R 2) Guidelines « a systematic approach to development that begins with predefined objectives and emphasizes product and process understanding and process control, based on sound science and quality risk management » Ø « Quality cannot be tested into products, quality should be built in by design » 2

REGULATORY REQUIREMENTS (2/2) Chem. Bio. Pharm § ICH Q 8(R 2): pharmaceutical development Design Space (DS) Q 8(R 2) Guidelines « the multidimensional combination and interaction of input variables and process parameters that have been demonstrated to provide assurance of quality » Ø Use of design of experiments 3

Qb. D & ANALYTICAL CHEMISTRY Chem. Bio. Pharm § Analytical methods Ø used for quality control of drugs => patient safety Ø should provide reliable scientific data for a better knowledge of the product all along its development Ø need a controlled risk-based development to guarantee quality § Design Space Ø sub-region of the experimental domain in which the objectives of the method are reached with a defined probability Ø robustness domain 4
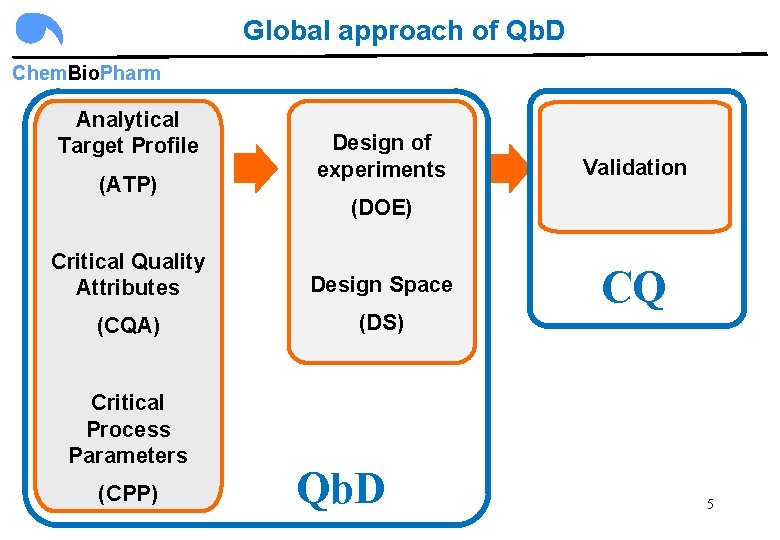
Global approach of Qb. D Chem. Bio. Pharm Analytical Target Profile (ATP) Design of experiments (DOE) Critical Quality Attributes Design Space (CQA) (DS) Critical Process Parameters (CPP) Validation Qb. D CQ 5
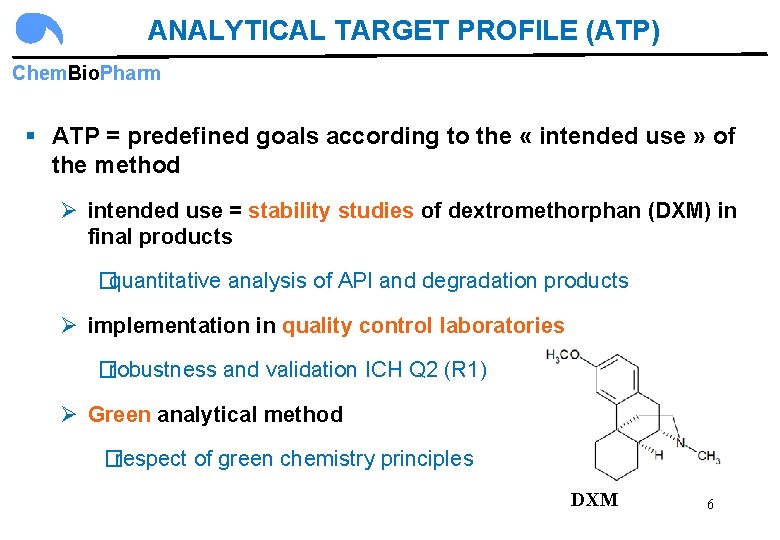
ANALYTICAL TARGET PROFILE (ATP) Chem. Bio. Pharm § ATP = predefined goals according to the « intended use » of the method Ø intended use = stability studies of dextromethorphan (DXM) in final products �quantitative analysis of API and degradation products Ø implementation in quality control laboratories �robustness and validation ICH Q 2 (R 1) Ø Green analytical method �respect of green chemistry principles DXM 6
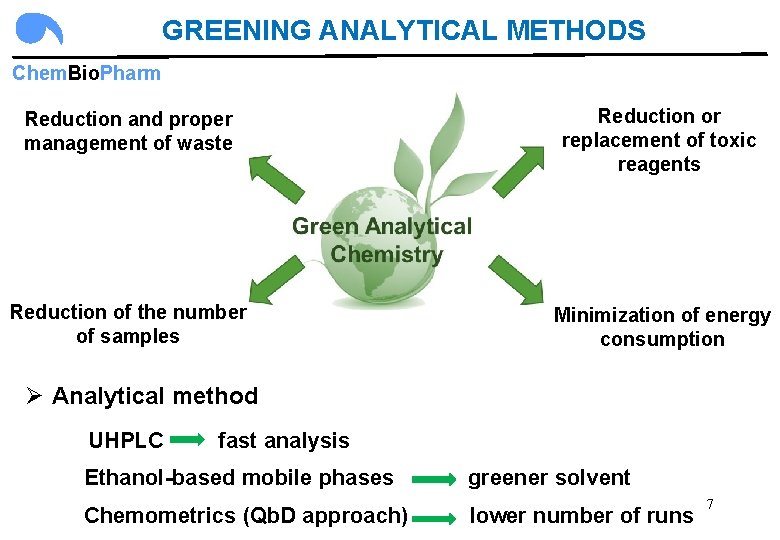
GREENING ANALYTICAL METHODS Chem. Bio. Pharm Reduction and proper management of waste Reduction or replacement of toxic reagents Reduction of the number of samples Minimization of energy consumption Ø Analytical method UHPLC fast analysis Ethanol-based mobile phases greener solvent 7 Chemometrics (Qb. D approach) lower number of runs

CRITICAL QUALITY ATTRIBUTES (CQAs) Chem. Bio. Pharm § CQAs = measurable attributes of the chromatogram that should be within an appropriate limit or range to ensure the desired quality of the method Must have Intend to have Ø Rs between DXM and impurities A, B & C Rs > 2 > 2. 5 Ø Peak efficiency impurity A N > 1500 Ø Peak efficiency impurity C N > 20000 > 3000 Ø Minimization of solvent consumption § Et. OH volume < 0. 4 m. L < 0. 35 m. L § Mobile phase volume < 2 m. L < 1. 2 m. L Assassi et al, Green analytical method development for statin analysis, J. Chromatogr. A, 37 (2015) 61 -72. 8
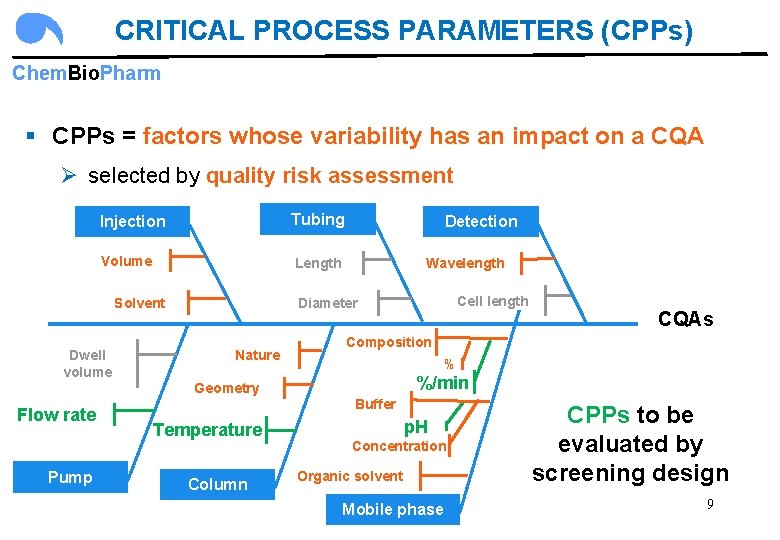
CRITICAL PROCESS PARAMETERS (CPPs) Chem. Bio. Pharm § CPPs = factors whose variability has an impact on a CQA Ø selected by quality risk assessment Injection Tubing Volume Length Solvent Dwell volume Detection Wavelength Nature % %/min Pump Buffer Temperature Column CQAs Composition Geometry Flow rate Cell length Diameter p. H Concentration Organic solvent Mobile phase CPPs to be evaluated by screening design 9
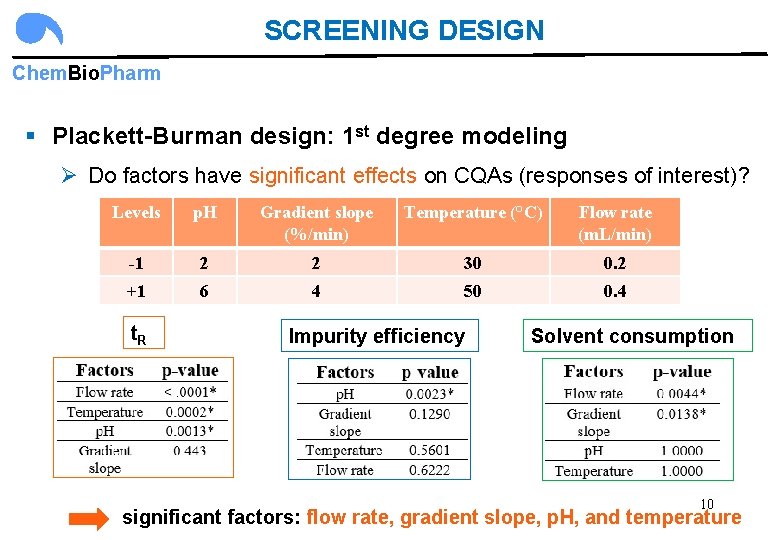
SCREENING DESIGN Chem. Bio. Pharm § Plackett-Burman design: 1 st degree modeling Ø Do factors have significant effects on CQAs (responses of interest)? Levels p. H Gradient slope (%/min) Temperature (°C) Flow rate (m. L/min) -1 2 2 30 0. 2 +1 6 4 50 0. 4 t. R Impurity efficiency Solvent consumption 10 significant factors: flow rate, gradient slope, p. H, and temperature
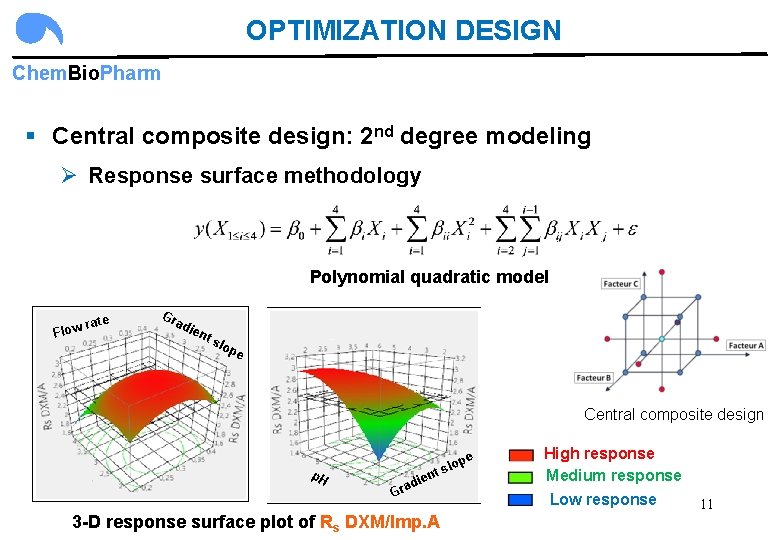
OPTIMIZATION DESIGN Chem. Bio. Pharm § Central composite design: 2 nd degree modeling Ø Response surface methodology Polynomial quadratic model rate Flow Gr ad ien t s lop e Central composite design e p. H die a r G p slo t n 3 -D response surface plot of Rs DXM/Imp. A High response Medium response Low response 11
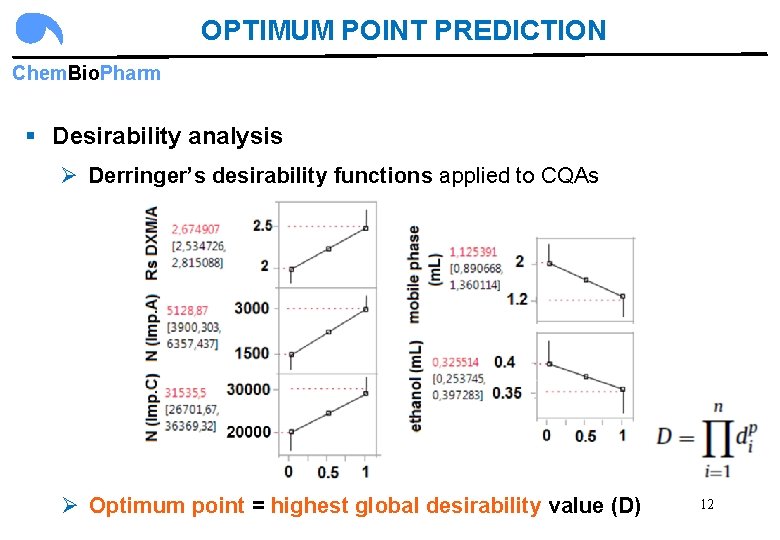
OPTIMUM POINT PREDICTION Chem. Bio. Pharm § Desirability analysis Ø Derringer’s desirability functions applied to CQAs Ø Optimum point = highest global desirability value (D) 12
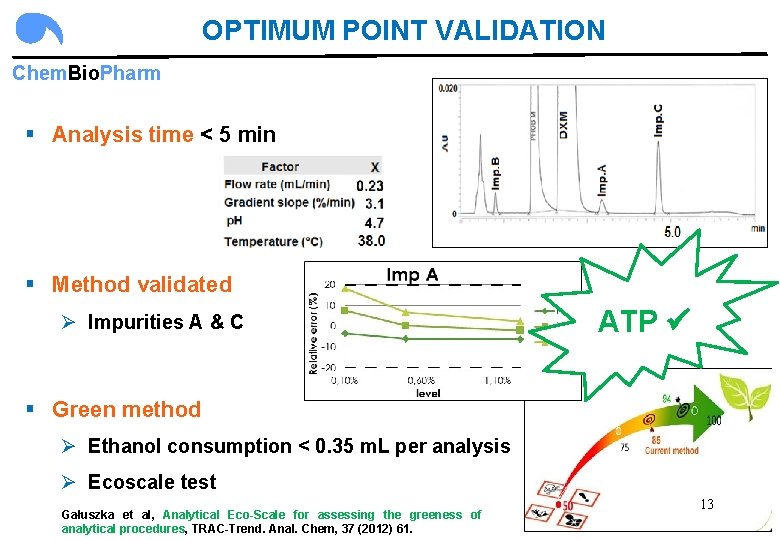
OPTIMUM POINT VALIDATION Chem. Bio. Pharm § Analysis time < 5 min § Method validated Ø Impurities A & C ATP § Green method Ø Ethanol consumption < 0. 35 m. L per analysis Ø Ecoscale test Gałuszka et al, Analytical Eco-Scale for assessing the greeness of analytical procedures, TRAC-Trend. Anal. Chem, 37 (2012) 61. 13
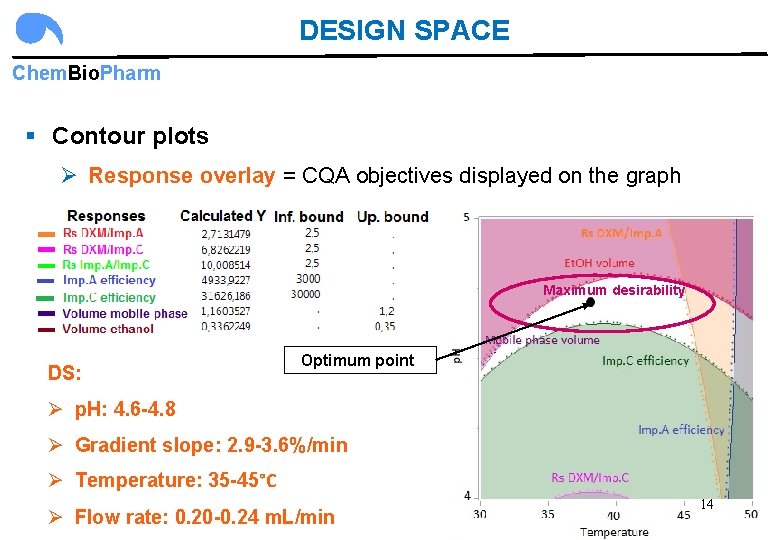
DESIGN SPACE Chem. Bio. Pharm § Contour plots Ø Response overlay = CQA objectives displayed on the graph Maximum desirability DS: Optimum point Ø p. H: 4. 6 -4. 8 Ø Gradient slope: 2. 9 -3. 6%/min Ø Temperature: 35 -45°C Ø Flow rate: 0. 20 -0. 24 m. L/min 14

ROBUSTNESS Chem. Bio. Pharm § New Plackett-Burman design performed around the optimum point (24 -1) § 8 validation points experimentally tested by varying CPPs from extreme limits of the DS range § CQAs met the specifications for all 8 points (D = 1) Ø DS = robustness domain 15

RISK-BASED DESIGN SPACE Mean based Chem. Bio. Pharm S > 1 min § Mean responses used for optimization Ø Do not provide any clue about method reliability Ø How well and how often the method can meet the specifications? What guarantee? § Optimized robust assay S = t. R, begin – t. R, end Ø Take into account model uncertainty Risk based P(S > 1 min) Ø Monte-Carlo study of the propagation of the model’s predictive errors Ø Quality is ensured by assessing the risk of not being within the acceptance limits E. Rozet et al, Design Spaces for Analytical Methods: what, why, how? , Tr. ACTrend. Anal. Chem, 42 (2013) 157 16
CONCLUSION Chem. Bio. Pharm § Rapid development of optimal and robust chromatographic methods § In line with ICH Q 8 § Better knowledge and easier introduction of new concepts (green chemistry) § Design space = robustness but method validation still necessary C. Boussès, L. Ferey, E. Vedrines, K. Gaudin, Using an innovative of Qb. D and Green analytical chemistry approaches for the developpement of a stability indicating UHPLC 17 mehod in pharmaceutical products, J. Pharm. Biomed. Anal. 115 (2015) 114 -122

THANK YOU FOR YOUR ATTENTION 18
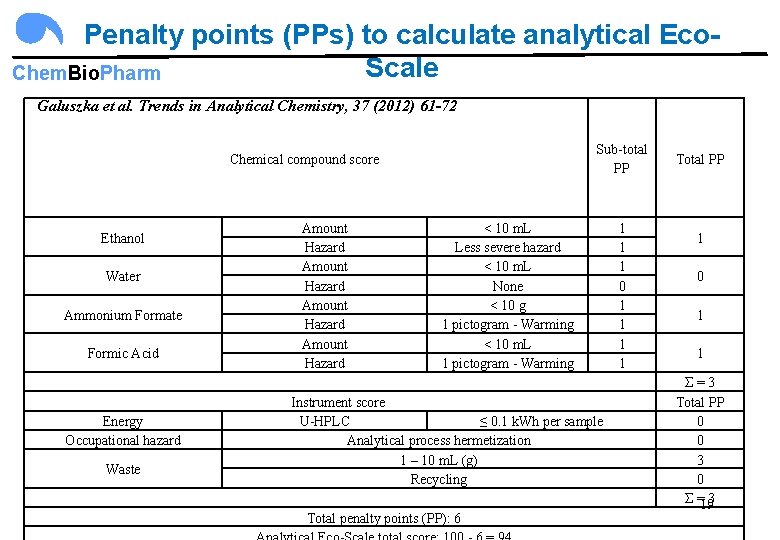
Penalty points (PPs) to calculate analytical Eco. Scale Chem. Bio. Pharm Galuszka et al. Trends in Analytical Chemistry, 37 (2012) 61 -72 Chemical compound score Ethanol Water Ammonium Formate Formic Acid Energy Occupational hazard Waste Sub-total PP Amount < 10 m. L Hazard Less severe hazard Amount < 10 m. L Hazard None Amount < 10 g Hazard 1 pictogram - Warming Amount < 10 m. L Hazard 1 pictogram - Warming Instrument score U-HPLC ≤ 0. 1 k. Wh per sample Analytical process hermetization 1 – 10 m. L (g) Recycling Total penalty points (PP): 6 1 1 1 0 1 1 Total PP 1 0 1 1 Σ = 3 Total PP 0 0 3 0 Σ = 3 19

METHOD VALIDATION Chem. Bio. Pharm § Protocol Calibration set: for each impurity, the stock solutions of each impurity (0. 1 mg/m. L in ethanol/water mixtures, (25/75, v/v) was diluted 20 fold to obtain a solution at 0. 5% of impurity. This dilution was done induplicate for each impurity which constituted the calibration set. Validation set: dilutions of the stock solutions by 100, 20, and 8 fold were prepared in triplicate in the presence of excipients and dextromethorphan to obtain limit of quantitation (LOQ), 0. 5 and 1. 25% concentration levels, respectively. All these solutions were 10 fold diluted while adding 6. 7 m. L of a DXM syrup at 3 mg m. L− 1. This procedure was repeated at 3 different days and each solution was injected in the optimal chromatographic conditions. 20
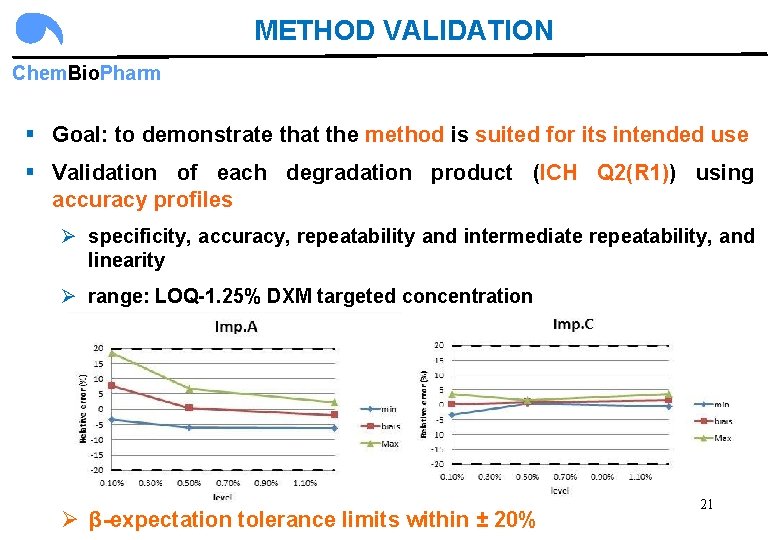
METHOD VALIDATION Chem. Bio. Pharm § Goal: to demonstrate that the method is suited for its intended use § Validation of each degradation product (ICH Q 2(R 1)) using accuracy profiles Ø specificity, accuracy, repeatability and intermediate repeatability, and linearity Ø range: LOQ-1. 25% DXM targeted concentration Ø β-expectation tolerance limits within ± 20% 21

DEXTROMETHORPHAN Chem. Bio. Pharm v Cyclic tertiary amine: model compound for the study of basic substances v p. Ka = 8. 2, a component of a cough medicine, has been analyzed by reversed-phase HPLC with some difficulties, and actually included as one of the test solutes aiming at the effect of silanols on elution of basic compounds. It has been eluted at acidic p. H or in the presence of an ion-pair reagent in most reported determinations to avoid severe tailing at neutral p. H in the absence of another amine. v Sensitive probe for silanol effect detection v Typical pharmaceutical case study relevant for the evaluation of robust conditions in green analytical chemistry 22
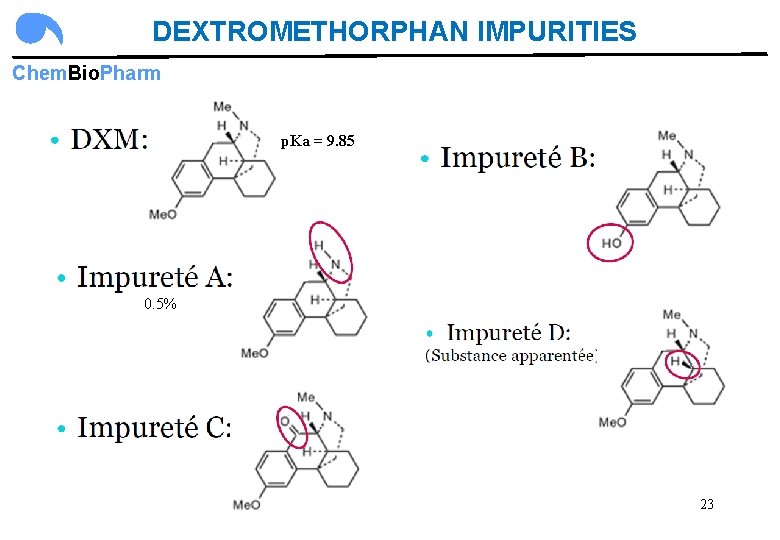
DEXTROMETHORPHAN IMPURITIES Chem. Bio. Pharm p. Ka = 9. 85 0. 5% 23

CHROMATOGRAPHIC CONDITIONS Chem. Bio. Pharm v Screening of 3 columns: Acquity BEH C 18 (50 x 2. 1 mm, 1. 7 µm) = hybrid silica, Acquity BEH Phenyl (50 x 2. 1 mm, 1. 7 µm) = covalently modified by phenyl grafting, Hypersil gold AQ (50 x 2. 1 mm, 1. 9 µm) grafted C 18 silica possible to use 100% aqueuous phase v Better selectivity of Acquity BEH C 18 = baseline separation of all peaks whereas on others columns coelutions of DXM and Imp. A were observed. v Mobile phase A: 10 m. M ammonium formate adjusted to different p. H with formic acid, and 96% ethanol (95/5, v/v) v Mobile phase B: 10 m. M ammonium formate in ethanol 96% with the same proportion of formic acid as mobile phase A v Gradient: initial hold of 1 min at 21% of mobile phase B followed by a linear ramp from 21% to 36% of mobile phase B, and 6 min of equilibration v Injection volume = 2 µL and detection wavelength = 280 nm. 24
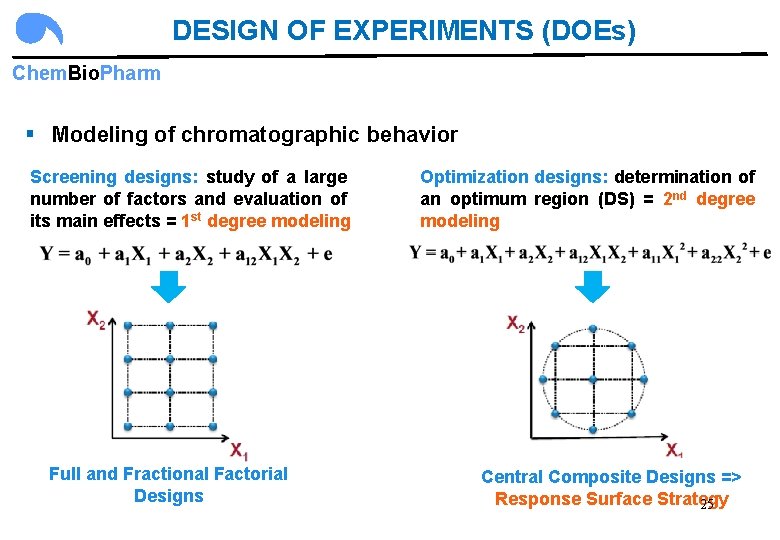
DESIGN OF EXPERIMENTS (DOEs) Chem. Bio. Pharm § Modeling of chromatographic behavior Screening designs: study of a large number of factors and evaluation of its main effects = 1 st degree modeling Full and Fractional Factorial Designs Optimization designs: determination of an optimum region (DS) = 2 nd degree modeling Central Composite Designs => Response Surface Strategy 25

RISK-BASED DESIGN SPACE Mean based Chem. Bio. Pharm S > 1 min § Mean responses used for optimization Ø Do not provide any clue about method reliability Ø How well and how often the method can meet the specifications? What guarantee? § Optimized robust assay S = t. R, begin – t. R, end Ø Take into account model uncertainty Risk based P(S > 1 min) Ø Monte-Carlo study of the propagation of the model’s predictive errors Ø Quality is ensured by assessing the risk of not being within the acceptance limits E. Rozet et al, Design Spaces for Analytical Methods: what, why, how? , Tr. ACTrend. Anal. Chem, 42 (2013) 157 26
- Slides: 26