CHEM 515 Spectroscopy Vibrational Spectroscopy III Normal Modes

CHEM 515 Spectroscopy Vibrational Spectroscopy III
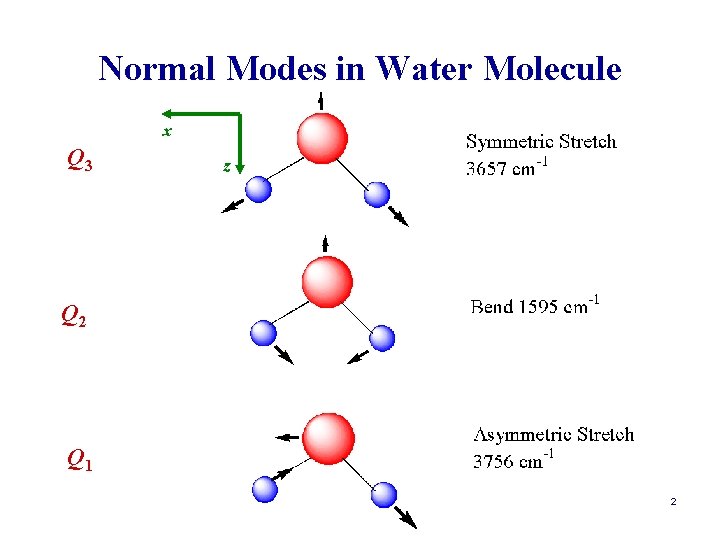
Normal Modes in Water Molecule x Q 3 z Q 2 Q 1 2
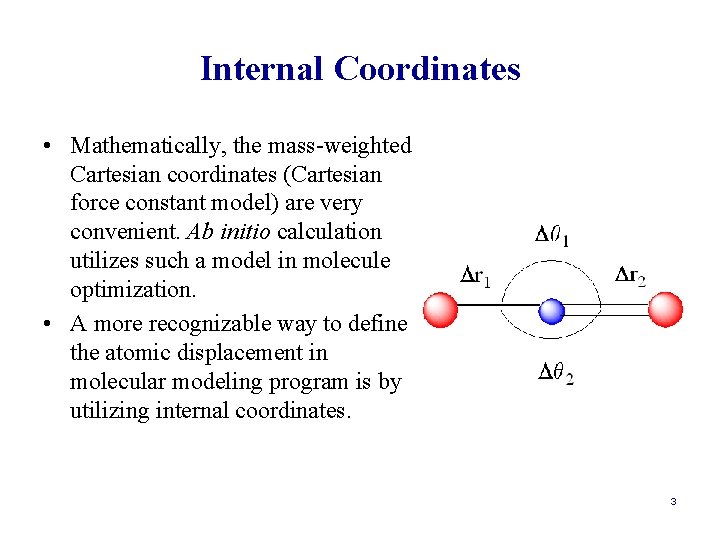
Internal Coordinates • Mathematically, the mass-weighted Cartesian coordinates (Cartesian force constant model) are very convenient. Ab initio calculation utilizes such a model in molecule optimization. • A more recognizable way to define the atomic displacement in molecular modeling program is by utilizing internal coordinates. 3
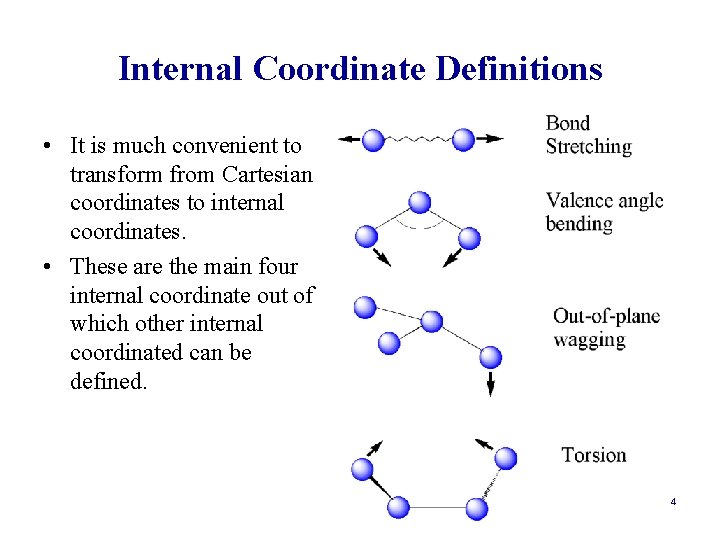
Internal Coordinate Definitions • It is much convenient to transform from Cartesian coordinates to internal coordinates. • These are the main four internal coordinate out of which other internal coordinated can be defined. 4

Internal Coordinate for Water Molecule • The internal displacement of atoms can be defined as change in the three coordinates. • Δr 1 • Δr 2 • Δθ r 1 r 2 θ 5

Symmetry Coordinates • More preferably than internal coordinates is the use of symmetry coordinates. • The concept of molecule symmetry “group theory” is applied with help of projection operators to generate the required 3 N – 6 symmetry coordinates. Symmetry specie Projection operator Normalization factor Operation Character for symmetry specie γ New coordinate obtained from Inti upon operation R 6

Symmetry Coordinates • This totally symmetric projection operator is used to get a set of symmetric coordinates by linear combinations of internal coordinates. • This method is also known as “Symmetry-Adapted Linear Combinations; SALCs)” as proposed by A. F. Cotton. Symmetry specie Normalization factor Operation Character for symmetry specie γ New coordinate obtained from Inti upon operation R 7
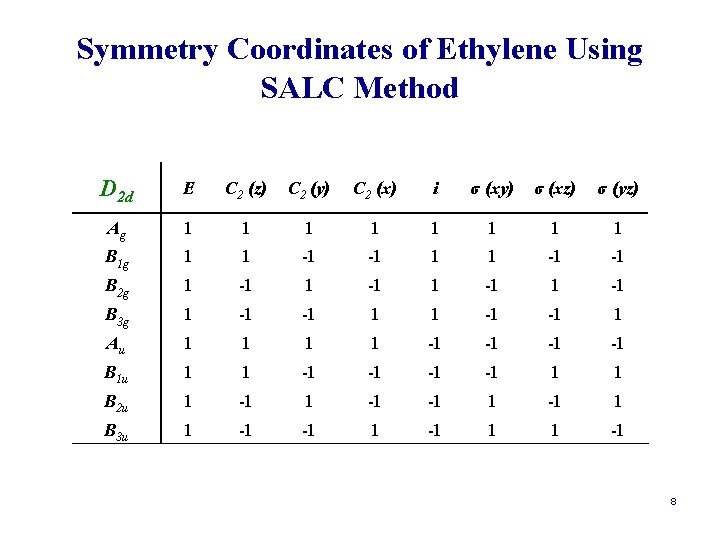
Symmetry Coordinates of Ethylene Using SALC Method D 2 d E C 2 (z) C 2 (y) C 2 (x) i σ (xy) σ (xz) σ (yz) Ag 1 1 1 1 B 1 g 1 1 -1 -1 B 2 g 1 -1 B 3 g 1 -1 -1 1 Au 1 1 -1 -1 B 1 u 1 1 -1 -1 1 1 B 2 u 1 -1 -1 1 B 3 u 1 -1 -1 1 1 -1 8
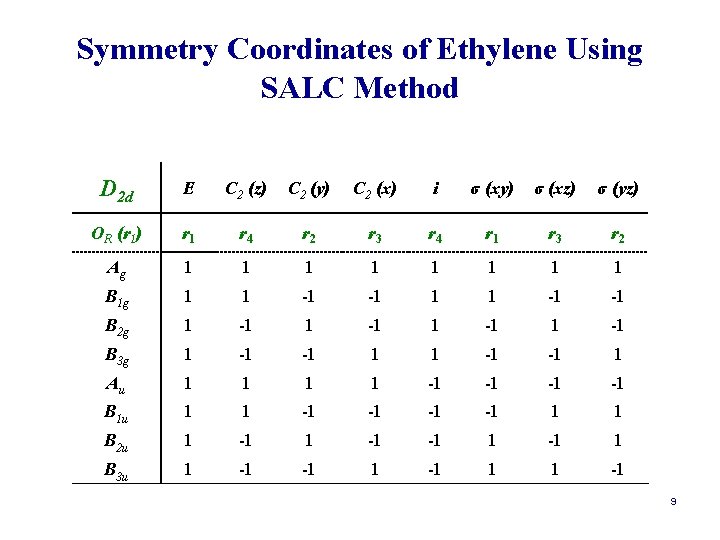
Symmetry Coordinates of Ethylene Using SALC Method D 2 d E C 2 (z) C 2 (y) C 2 (x) i σ (xy) σ (xz) σ (yz) OR (r 1) r 1 r 4 r 2 r 3 r 4 r 1 r 3 r 2 Ag 1 1 1 1 B 1 g 1 1 -1 -1 B 2 g 1 -1 B 3 g 1 -1 -1 1 Au 1 1 -1 -1 B 1 u 1 1 -1 -1 1 1 B 2 u 1 -1 -1 1 B 3 u 1 -1 -1 1 1 -1 9

Determining the Symmetry Species for the Vibrations in a Molecule • We are very concerned with the symmetry of each normal mode of vibration in a molecule. • Each normal mode of vibration will form a basis for an irreducible representation (Γ) of the point group of the molecule. • The objective is to determine what the character (trace) is for the transformation matrix corresponding to a particular operation in a specific molecule. 10

Symmetry of Normal Modes of Vibrations in H 2 O • H 2 O has C 2 v symmetry. • Operation E results in the following transformations: 11

Symmetry of Normal Modes of Vibrations in H 2 O • The transformations in the x, y and z modes can be represented with the following matrix transformation: • Trace of E matrix is equal to 9. 12

Symmetry of Normal Modes of Vibrations in H 2 O • The operation C 2 is more interesting! • Operation C 2 results in the following transformations: 13

Symmetry of Normal Modes of Vibrations in H 2 O • The transformations in the x, y and z modes can be represented with the following matrix transformation: • Trace of C 2 matrix is equal to – 1. 14

Determining the Symmetry Species for the Vibrations in a Molecule: a Shorter Way • The matrix transformation method is very cumbersome. However, it can be streamlined tremendously another procedure. • Alternative Method: 1. Count unshifted atoms per each operation. 2. Multiply by contribution per unshifted atom to get the reducible representation (Γ). 3. Determine (Γ) for each symmetry operation. 4. Subtract Γtrans and Γrot from Γtot. Γvib = Γtot – Γtrans – Γrot. 15
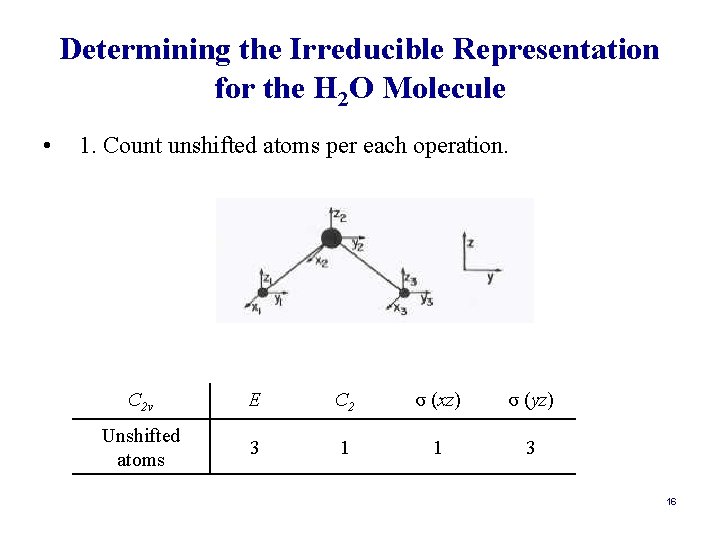
Determining the Irreducible Representation for the H 2 O Molecule • 1. Count unshifted atoms per each operation. C 2 v E C 2 σ (xz) σ (yz) Unshifted atoms 3 1 1 3 16
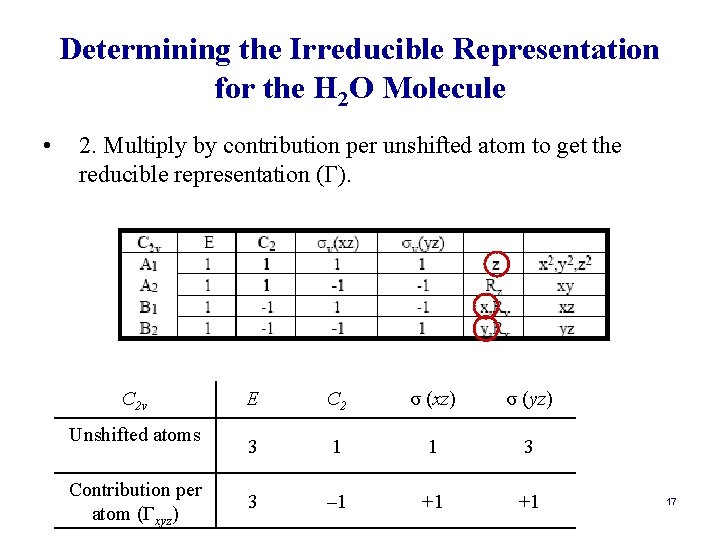
Determining the Irreducible Representation for the H 2 O Molecule • 2. Multiply by contribution per unshifted atom to get the reducible representation (Γ). C 2 v Unshifted atoms Contribution per atom (Γxyz) E C 2 σ (xz) σ (yz) 3 1 1 3 3 – 1 +1 +1 17
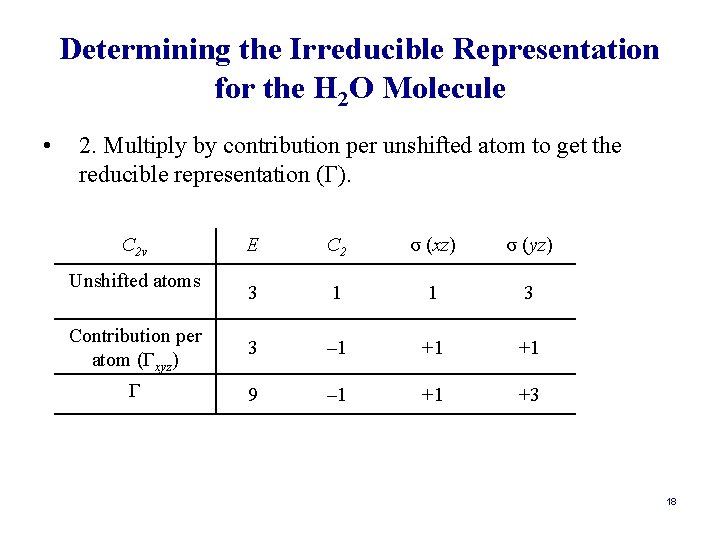
Determining the Irreducible Representation for the H 2 O Molecule • 2. Multiply by contribution per unshifted atom to get the reducible representation (Γ). C 2 v E C 2 σ (xz) σ (yz) 3 1 1 3 Contribution per atom (Γxyz) 3 – 1 +1 +1 Γ 9 – 1 +1 +3 Unshifted atoms 18

Determining the Irreducible Representation for the H 2 O Molecule • 3. Determine (Γ) for each symmetry operation. ηi : number of times the irreducible representation (Γ) appears for the symmetry operation i. h : order of the point group. R : an operation of the group. χR : character of the operation R in the reducible represent. χi. R : character of the operation R in the irreducible represent. CR : number of members of class to which R belongs. 19

Determining the Irreducible Representation for the H 2 O Molecule C 2 v E C 2 σ (xz) σ (yz) Γ 9 – 1 +1 +3 20

Determining the Irreducible Representation for the H 2 O Molecule C 2 v E C 2 σ (xz) σ (yz) Γ 9 – 1 +1 +3 21

Determining the Irreducible Representation for the H 2 O Molecule • 3. Determine (Γ) for each symmetry operation. Γtot = 3 A 1 + A 2 + 2 B 1 + 3 B 2 • Number of irreducible representations Γtot must equal to 3 N for the molecule. 22

Determining the Irreducible Representation for the H 2 O Molecule • Subtract Γtrans and Γrot from Γtot = 3 A 1 + A 2 + 2 B 1 + 3 B 2 23
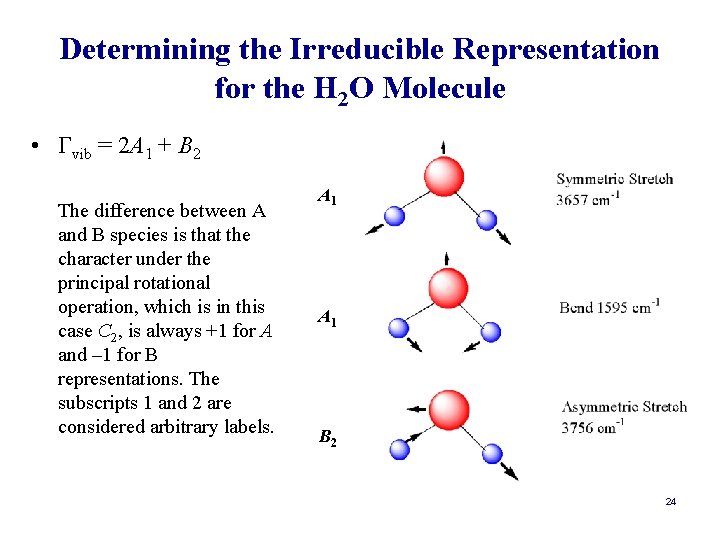
Determining the Irreducible Representation for the H 2 O Molecule • Γvib = 2 A 1 + B 2 The difference between A and B species is that the character under the principal rotational operation, which is in this case C 2, is always +1 for A and – 1 for B representations. The subscripts 1 and 2 are considered arbitrary labels. A 1 B 2 24
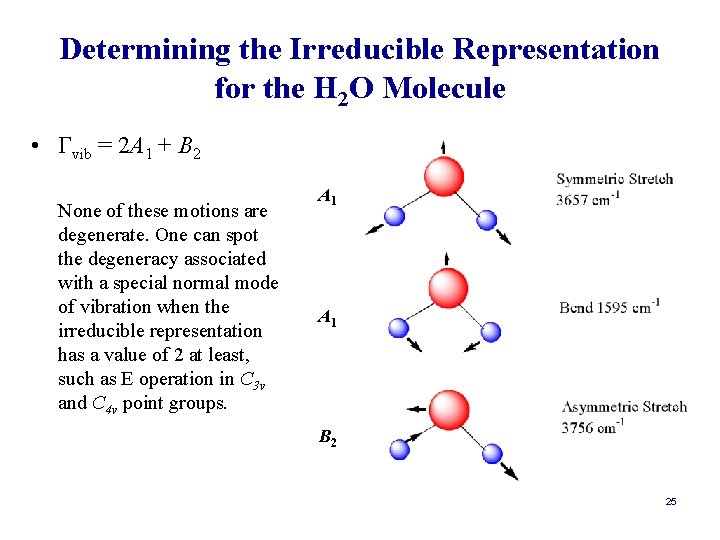
Determining the Irreducible Representation for the H 2 O Molecule • Γvib = 2 A 1 + B 2 None of these motions are degenerate. One can spot the degeneracy associated with a special normal mode of vibration when the irreducible representation has a value of 2 at least, such as E operation in C 3 v and C 4 v point groups. A 1 B 2 25
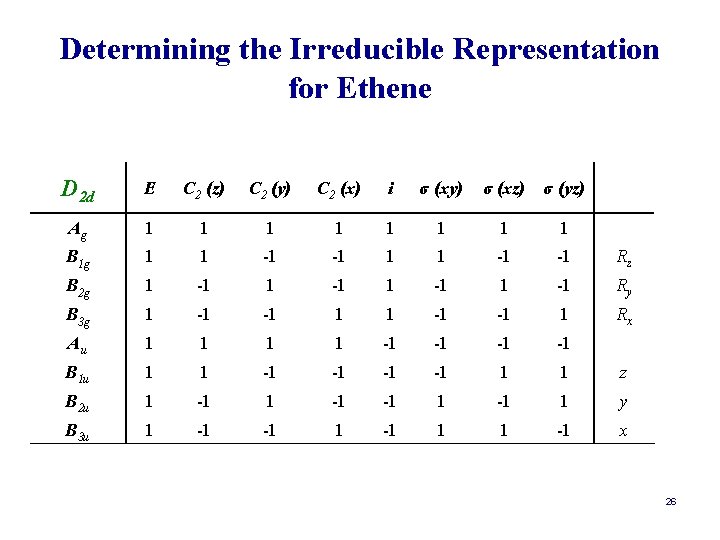
Determining the Irreducible Representation for Ethene D 2 d E C 2 (z) C 2 (y) C 2 (x) i σ (xy) σ (xz) σ (yz) Ag 1 1 1 1 B 1 g 1 1 -1 -1 Rz B 2 g 1 -1 Ry B 3 g 1 -1 -1 1 Rx Au 1 1 -1 -1 B 1 u 1 1 -1 -1 1 1 z B 2 u 1 -1 -1 1 y B 3 u 1 -1 -1 1 1 -1 x 26

Determining the Irreducible Representation for Ethene 27
Determining the Irreducible Representation for Ethene 28

Normal Modes in Ethene Physical Chemistry By Robert G. Mortimer 29

Mutual Exclusion Principle • For molecules having a center of symmetry (i), the vibration that is symmetric w. r. t the center of symmetry is Raman active but not IR active, whereas those that are antisymmetric w. r. t the center of symmetry are IR active but not Raman active. 30
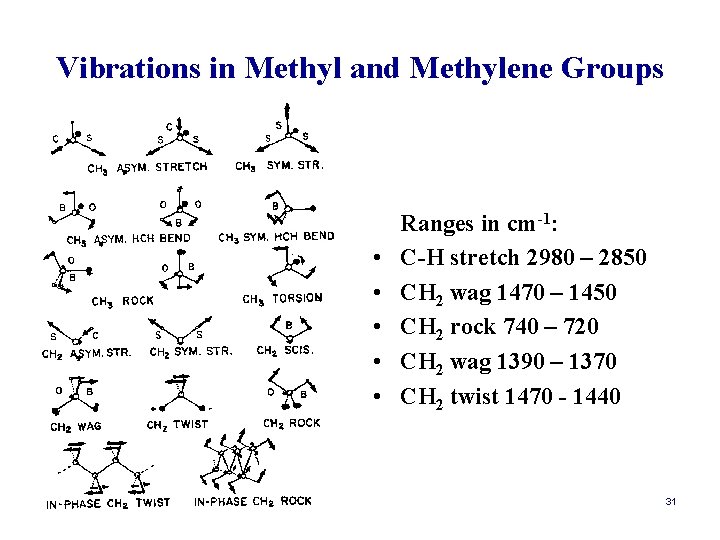
Vibrations in Methyl and Methylene Groups • • • Ranges in cm-1: C-H stretch 2980 – 2850 CH 2 wag 1470 – 1450 CH 2 rock 740 – 720 CH 2 wag 1390 – 1370 CH 2 twist 1470 - 1440 31
- Slides: 31