CHEM 494 Special Topics in Chemistry University of





































- Slides: 45

CHEM 494 Special Topics in Chemistry University of Illinois at Chicago UIC CHEM 494 - Lecture 7 Prof. Duncan Wardrop October 22, 2012

CHEM 494 Special Topics in Chemistry University of Illinois at Chicago UIC Halogenation of Alkanes Methods and Mechanism Chapter 39

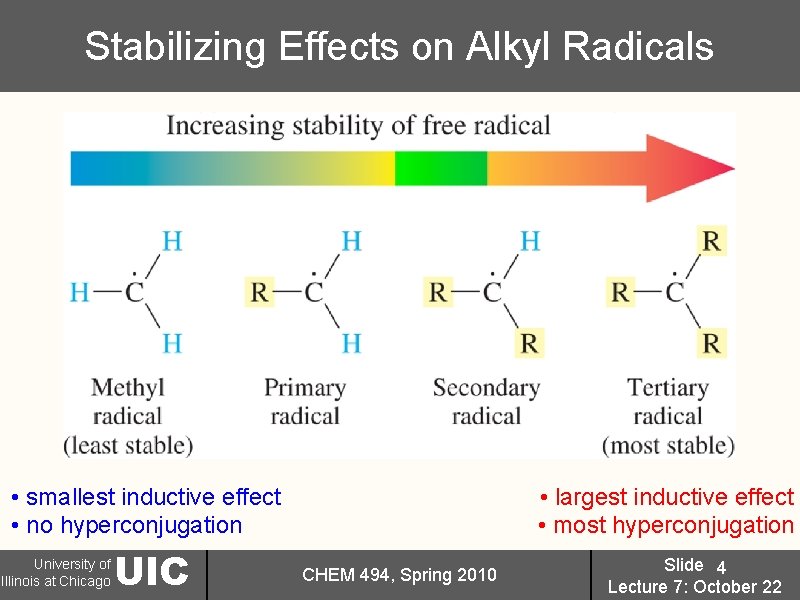
Structure of Alkyl Radical Intermediates • radicals are high energy intermediates; 7 valence electrons; cannot be isolated • sp 2 -hybridized; contain one empty porbital; unpaired electron in the porbital; alkyl radicals can be stabilized by inductive effects and hyperconjugation; similar to carbocations • approximately planar: three bonds to carbon are at ~120º angles from each other and ~90º to half-filled p-orbital • stabilized by inductive effects and hyperconjugation • Stability: 3º > 2º >> 1º > CH 3 University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 3 Lecture 7: October 22

Stabilizing Effects on Alkyl Radicals • smallest inductive effect • no hyperconjugation University of Illinois at Chicago UIC • largest inductive effect • most hyperconjugation CHEM 494, Spring 2010 Slide 4 Lecture 7: October 22

Bromination is More Selective Than Chlorination † † Hammond Postulate • chlorine radicals are higher in energy than bromine radicals = • transition states in chlorination are earlier= • look more like reactants = • less difference in TS energy = • less selective = • greater mixture ‡ ‡ † = early transition state structures ‡ = late transition state structures ∆Ea (bromination) > ∆Ea (chlorination) Bromination is more selective. Relative Rates (krel) of Halogenation chlorination bromination University of Illinois at Chicago R 3 CH (tertiary, 3º) R 2 CH 2 (secondary, 2º) RCH 3 (primary, 1º) 5. 2 1640 3. 9 82 1. 0 UIC • bromine radicals are lower in energy than chlorine radicals = • transition states in bromination are later= • look more like products (radical interm. ) = • greater difference in TS energy = • more selective = • less of a mixture CHEM 494, Spring 2010 Slide 5 Lecture 7: October 22

Quantifying Selectivity Relative Rates (krel) of Halogenation R 3 CH R 2 CH 2 RCH 3 (tertiary, 3º) (secondary, 2º)(primary, 1º) chlorination 5. 2 3. 9 1. 0 bromination 1640 82 1. 0 %= (krel) x (statistical factor) total Product Relative Yield Absolute Yield chlorination A (2 2º H’s) 2 x 3. 9 = 7. 8/13. 8 = 57% B (6 1º H’s) 6 x 1 = 6. 0/13. 8 = 43% Sum 13. 8 100% bromination Predicted Product Ratios A (2 2º H’s) 2 x 82 = 164/170 = 96% B (6 1º H’s) 6 x 1 = 6. 0/170 = 4% Sum 170 100% University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 6 Lecture 7: October 22

Self Test Question Determine the predicted product distribution for A in the following chlorination. Relative Rates (krel) of Halogenation R 3 CH R 2 CH 2 RCH 3 (tertiary, 3º) (secondary, 2º)(primary, 1º) chlorination 5. 2 3. 9 1. 0 bromination 1640 82 1. 0 University of Illinois at Chicago UIC CHEM 494, Spring 2010 A. 99% B. 97% C. 95% D. 93% E. 91% Slide 7 Lecture 7: October 22

Defining Regioselectivity (regioselective) A regioselective reaction is one in which one direction of bond making or breaking occurs preferentially over all other possible directions. Reactions are termed completely (100%) regioselective if the discrimination is complete, or partially (<100%), if the product of reaction at one site predominates over the product of reaction at other sites. The discrimination may also semiquantitatively be referred to as high or low regioselectivity. IUPAC Compendium of Chemical Terminology 2 nd Edition (1997) University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 8 Lecture 7: October 22

Mechanism of Alkane Chlorination University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 9 Lecture 7: October 22

Factors Governing Regioselectivity • Strength of C-H Bond Broken • Number & Type of Substrate C-H Bonds • Strength of C-X Bond Formed and X-X Bond Broken University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 10 Lecture 7: October 22

Bromination is More Selective Than Chlorination † † Hammond Postulate • chlorine radicals are higher in energy than ‡ bromine radicals = • transition states in chlorination are earlier = • look more like reactants = • less difference in TS energy = • less selective = • greater mixture ‡ † = early transition state structures ‡ = late transition state structures ∆Ea (bromination) > ∆Ea (chlorination) Bromination is more selective. Relative Rates (krel) of Halogenation chlorination bromination University of Illinois at Chicago R 3 CH (tertiary, 3º) R 2 CH 2 (secondary, 2º) RCH 3 (primary, 1º) 5. 2 1640 3. 9 82 1. 0 UIC • bromine radicals are lower in energy than chlorine radicals = • transition states in bromination are later = • look more like products (radical interm. ) = • greater difference in TS energy = • more selective = • less of a mixture CHEM 494, Spring 2010 Slide 11 Lecture 7: October 22

Quantifying Selectivity of Halogenation Relative Rates (krel) of Halogenation R 3 CH R 2 CH 2 RCH 3 (tertiary, 3º) (secondary, 2º)(primary, 1º) chlorination 5. 2 3. 9 1. 0 bromination 1640 82 1. 0 %= (krel) x (statistical factor) total Product Relative Yield Absolute Yield chlorination A (2 2º H’s) 2 x 3. 9 = 7. 8/13. 8 = 57% B (6 1º H’s) 6 x 1 = 6. 0/13. 8 = 43% Sum 13. 8 100% bromination Predicted Product Ratios A (2 2º H’s) 2 x 82 = 164/170 = 96% B (6 1º H’s) 6 x 1 = 6. 0/170 = 4% Sum 170 100% University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 12 Lecture 7: October 22

Self Test Question Determine the predicted product distribution for A in the following chlorination. Relative Rates (krel) of Halogenation R 3 CH R 2 CH 2 RCH 3 (tertiary, 3º) (secondary, 2º)(primary, 1º) chlorination 5. 2 3. 9 1. 0 bromination 1640 82 1. 0 University of Illinois at Chicago UIC CHEM 494, Spring 2010 A. >99% B. 97% C. 95% D. 93% E. 91% Slide 13 Lecture 7: October 22

CHEM 494 Special Topics in Chemistry University of Illinois at Chicago UIC Nomenclature & Stereoisomerism in Alkenes

The Terms Alkene and Olefin are Synonymous • Alkenes are hydrocarbons that are characterized by a C-C double bond • Also called “olefins” • General molecular formula = Cn. H 2 n ethylene C 2 H 4 University of Illinois at Chicago UIC • Described as unsaturated since they have two fewer H atoms than equivalent alkanes CHEM 494, Spring 2010 Slide 15 Lecture 7: October 22

Index of Hydrogen Deficiency (IHD) IHD is synonymous with degrees of unsaturation and indicates the number of double/triple bonds and/or rings in a molecule IHD = 1 (1 double bond) IHD = 4 (3 double bonds & 1 ring) IHD = 2 (2 rings) IHD = 1 (one ring) University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 16 Lecture 7: October 22

Calculating IHD . Hydrocarbons (Cn. Hx) and Oxygenates (Cn. Hx. Oy): Ignore (2 n+2) - X IHD = 2 . Compounds with N (Cn. Hx. Ny): Subtract # N from # H (2 n+2) - (X-Y) IHD = 2 3. Halogens (Cn. Hx. Xy): Add # Halogens to # H (2 n+2) - (X+Y) IHD = 2 University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 17 Lecture 7: October 22

IHD & The Chemistry of Margarine Employing equation 1 (slide 12). . . . University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 18 Lecture 7: October 22

IUPAC Naming of Alkenes 4 -chloro-1 -pentene or 4 -chloropent-1 -ene Steps: 1. Number longest chain that includes both alkene carbons so that they have the lowest locants. 2. Replace -ane ending of parent alkane with -ene (alkene). 3. In the name, list the locant of only the first alkene carbon. Conventions/Rules: • • • alkenes have higher priority over alkane and halide substituents when numbering the longest chain alcohols have higher priority over alkenes when numbering the longest chain locant may be placed in front of parent name (e. g. 2 -pentene) or in front of suffix (e. g. pent-2 -ene). locant University of Illinois at Chicago substituent 1 st alphabetically UIC parent base CHEM 494, Fall 2012 alkene locant “ene” Slide 19 Lecture 7: October 22

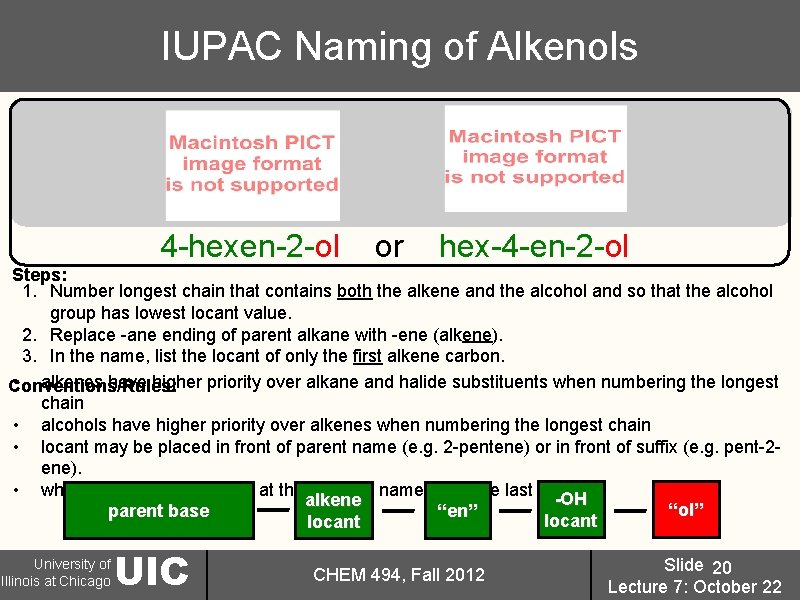
IUPAC Naming of Alkenols 4 -hexen-2 -ol or hex-4 -en-2 -ol Steps: 1. Number longest chain that contains both the alkene and the alcohol and so that the alcohol group has lowest locant value. 2. Replace -ane ending of parent alkane with -ene (alkene). 3. In the name, list the locant of only the first alkene carbon. • alkenes have higher priority over alkane and halide substituents when numbering the longest Conventions/Rules: chain • alcohols have higher priority over alkenes when numbering the longest chain • locant may be placed in front of parent name (e. g. 2 -pentene) or in front of suffix (e. g. pent-2 ene). • when “ene” does not occur at the end of a name, drop the last “e”-OH alkene “ol” parent base “en” locant University of Illinois at Chicago UIC CHEM 494, Fall 2012 Slide 20 Lecture 7: October 22

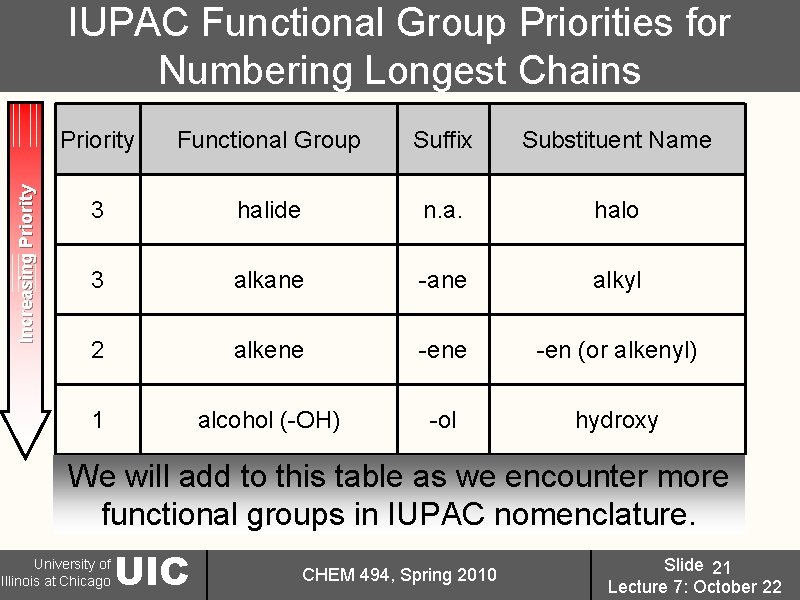
Increasing Priority IUPAC Functional Group Priorities for Numbering Longest Chains Priority Functional Group Suffix Substituent Name 3 halide n. a. halo 3 alkane -ane alkyl 2 alkene -en (or alkenyl) 1 alcohol (-OH) -ol hydroxy We will add to this table as we encounter more functional groups in IUPAC nomenclature. University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 21 Lecture 7: October 22

Common Alkenyl Group Names University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 22 Lecture 7: October 22

Alkene Structure & Bonding • carbons in an alkenes are sp 2 hybridized • bond angles are ~120º around sp 2 hybridized carbons • geometry around sp 2 -carbons is planar (flat) University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 23 Lecture 7: October 22

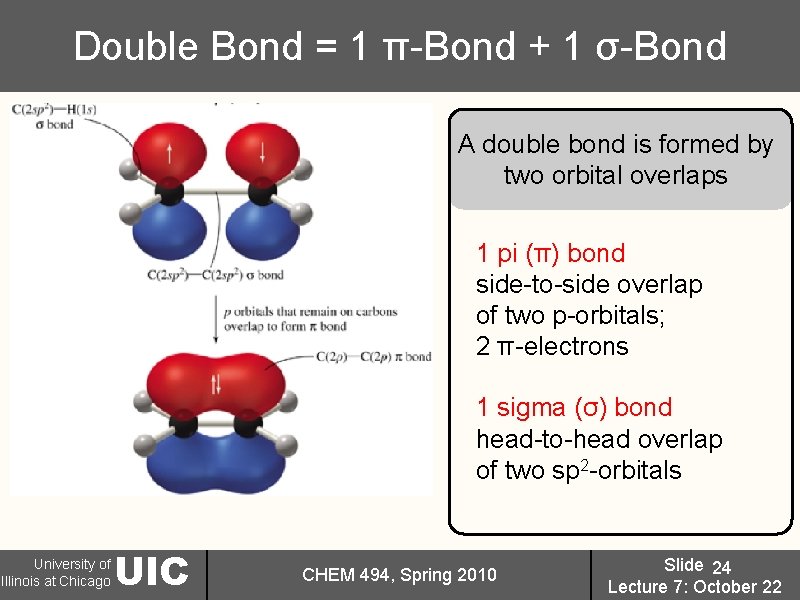
Double Bond = 1 π-Bond + 1 σ-Bond A double bond is formed by two orbital overlaps 1 pi (π) bond side-to-side overlap of two p-orbitals; 2 π-electrons 1 sigma (σ) bond head-to-head overlap of two sp 2 -orbitals University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 24 Lecture 7: October 22

Bond Rotation of Alkanes vs. Alkenes University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 25 Lecture 7: October 22

Alkenes Resist Rotation Round C=C Bond • π-bond prevents full rotation around double bond • 90º rotation would break π-bond to generate diradical • substituents on alkene “locked” in spacial relationships • some rotation is possible through pyramidalization (more on this topic later) University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 26 Lecture 7: October 22

Photolysis Mediates C=C Bond Rotation University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 27 Lecture 7: October 22

Geometrical Stereoisomers of Alkenes • restricted rotation around double bond gives cis and trans geometrical isomers (stereoisomers) • cis and trans: relationship between two vicinal groups on alkene • cis: same side; trans: opposite sides University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 28 Lecture 7: October 22

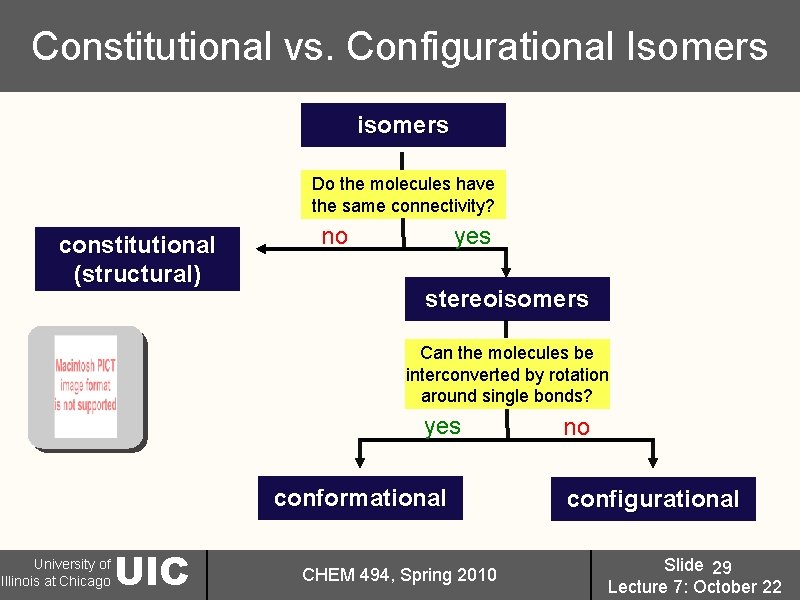
Constitutional vs. Configurational Isomers isomers Do the molecules have the same connectivity? constitutional (structural) no yes stereoisomers Can the molecules be interconverted by rotation around single bonds? yes conformational University of Illinois at Chicago UIC CHEM 494, Spring 2010 no configurational Slide 29 Lecture 7: October 22

Stereoisomers stereoisomers Can the molecules be interconverted by rotation around single bonds? yes conformational no configurational Is the isomerism at a doulbe bond? yes geometrical University of Illinois at Chicago UIC CHEM 494, Spring 2010 no optical Slide 30 Lecture 7: October 22

E-Z Nomenclature for Alkenes University of Illinois at Chicago • • • Groups are ranked according to atomic number • E (entgegen) = opposite; highest priority group on each alkene carbon on opposite sides Higher atomic number = higher rank Z (zusammen) = together; highest priority group on each alkene carbon on same side UIC CHEM 494, Spring 2010 Slide 31 Lecture 7: October 22

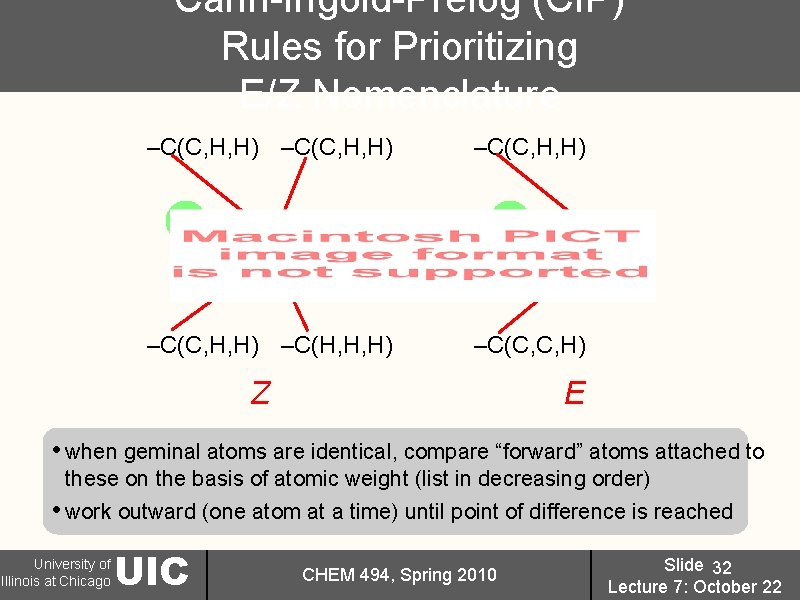
Cahn-Ingold-Prelog (CIP) Rules for Prioritizing E/Z Nomenclature –C(C, H, H) –C(H, H, H) –C(C, C, H) Z E • when geminal atoms are identical, compare “forward” atoms attached to these on the basis of atomic weight (list in decreasing order) • work outward (one atom at a time) until point of difference is reached University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 32 Lecture 7: October 22

Cahn-Ingold-Prelog (CIP) Rules for Prioritizing E/Z Nomenclature –C(O, O, C) –C(O, C, H) E • an atom that is multiply bonded to another atom is listed as two separate atoms for nomenclature purposesremember to list attached atoms in decreasing order of atomic weighthighest priority = highest atomic weight at first point of difference University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 33 Lecture 7: October 22

Why Alkene Geometry Matters Geometry influences molecular shape and, in turn, physical, chemical & biological properties. . Z Configuration Vascular targeting agent Combretastatin A 4 (CA-4) E Configuration Biologically Inactive! S. A. bush willow tree epi-Combretastatin A 4 (CA-4) University of Illinois at Chicago UIC Combretum caffrum CHEM 494, Spring 2010 Slide 34 Lecture 7: October 22

IUPAC: Stereoisomeric Alkenes (E)-3 -methyl-2 -pentene or (E)-3 -methylpent-2 -ene Steps: 1. Number the longest chain. 2. Replace -ane ending of parent alkane with -ene or -en, depending on priority. 3. In the name, list the locant of only the first alkene carbon. 4. Indicate geometrical isomers with (Z) or (E) at the beginning of the name. Conventions/Rules: • Z and E are italicized • Z and E are placed within parentheses at beginning of name: (Z) & (E). • cis and trans may be substituted for E/Z when alkene is 1, 2 -disubstituted “(E)” “(Z)” University of Illinois at Chicago UIC parent base alkene locant CHEM 494, Fall 2012 “ene” Slide 35 Lecture 7: October 22

CHEM 494 Special Topics in Chemistry University of Illinois at Chicago Relative Stabilities of Alkenes UIC

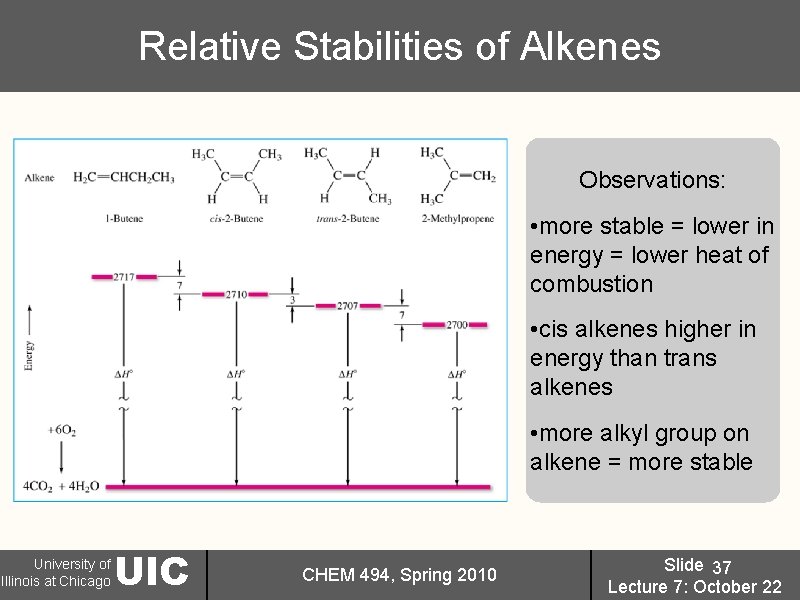
Relative Stabilities of Alkenes Observations: • more stable = lower in energy = lower heat of combustion • cis alkenes higher in energy than trans alkenes • more alkyl group on alkene = more stable University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 37 Lecture 7: October 22

Relative Stabilities of Alkenes 1. Steric Effects van der Waals strain between cis groups University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 38 Lecture 7: October 22

Relative Stabilities of Alkenes 2. Substituent Effects alkyl groups stabilize alkenes through donation (via induction and hyperconjugation) to the more electronegative sp 2 -hybridized carbon atoms Stability University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 39 Lecture 7: October 22

CHEM 494 Special Topics in Chemistry University of Illinois at Chicago UIC Preparation of Alkenes Elimination Chapter 19

β-Elimination Reactions Summary of β-Elimination (1, 2 -elimination) Reactions University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 41 Lecture 7: October 22

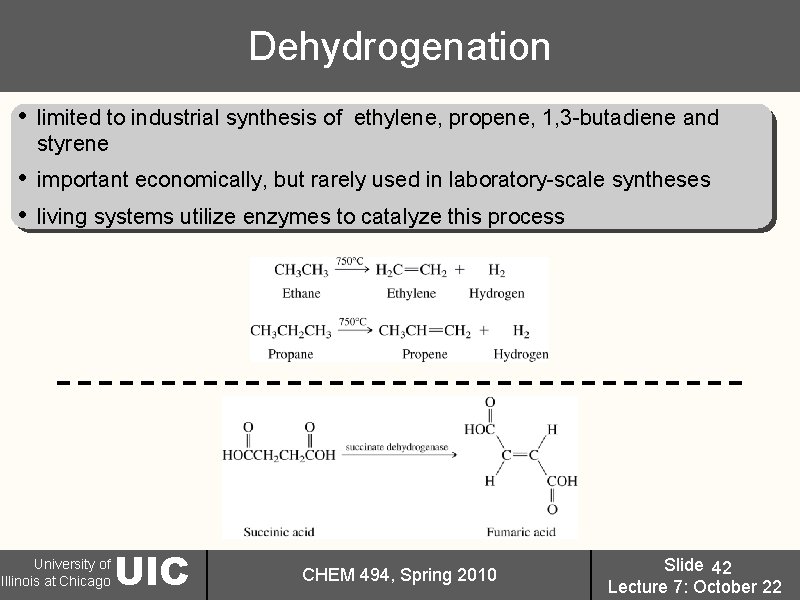
Dehydrogenation • limited to industrial synthesis of ethylene, propene, 1, 3 -butadiene and styrene • • important economically, but rarely used in laboratory-scale syntheses living systems utilize enzymes to catalyze this process University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 42 Lecture 7: October 22

Strong Acids Catalyze the Dehydration of 2° & 3 ° Alcohols • H 2 SO 4 and H 3 PO 4 are most common acids used for dehydration: protonation of OH group is first step • dehydration is reversible (hydration) in aqueous acid (Chapter 6) University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 43 Lecture 7: October 22

Industrial Example of Dehydration University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 44 Lecture 7: October 22

Dehydration can be “Coupled” with Other Chemical Transformation Two-step, one-pot transformation involves a Friedel-Crafts reaction (see, Chapter 12) and dehydration of the resulting 3° alcohol University of Illinois at Chicago UIC CHEM 494, Spring 2010 Slide 45 Lecture 7: October 22