CHEM 433 11111 V Phase Equilibria Pure Substances

CHEM 433 – 11/1/11 V. Phase Equilibria (Pure Substances) • Intro & definitions. • Phase diagrams - CO 2 , H 2 O, C • Phase Stability vs. T READ: CHAPTER 4 HW #6: via e-mail – DUE Th. EXAM #3 on Tuesday – review(s) Monday Web up to date with review sheet today.

Phase: A form of mater, uniform in chemical composition and physical state. Phase Transition: The spontaneous conversion of one phase to another (e. g. g —> l) Transition Temperature (Ttrs): Chemical potentials of 2 phases are equal and the 2 phases are in equilibrium Phase Diagram: Shows regions of P and T in which phases are stable Phase boundaries: p and T where 2 phases co-exist (in eq. , ’s are =). Vapor pressure: pressure of vapor in eq. with liquid (the l-v boundary). Sublimation vapor pressure: pressure of vapor in eq. with solid (s-v phase boundary)

Boiling: A condition of free vaporization throughout the liquid. Boiling temperature(Tb): T at which vapor pressure equals external pressure Normal boiling: boiling T at 1 atm external pressure Standard boiling point: boiling T at 1 bar external pressure. Freezing/Melting Temperature (Tf): T at which liquid and solid are in EQ at a given p. Normal/Standard Tf: as above, for 1 atm or 1 bar, respectively.

Triple point: Temperature and pressure at which 3 phases coexist (usually s, l, g). Critical temperature: T when surface between phases disappears (closed system). Density of (l) and (g) become equal! Critical pressure: vapor pressure at critical temperature. Supercritical Fluid: The name of the phase above the critical point.
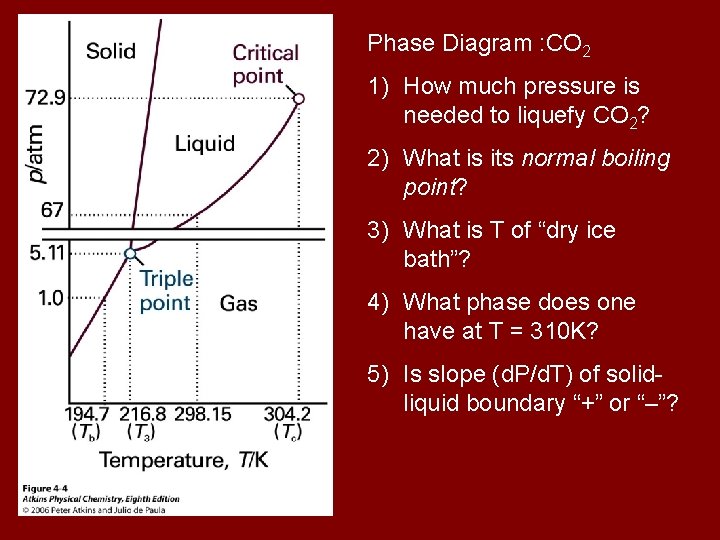
Phase Diagram : CO 2 1) How much pressure is needed to liquefy CO 2? 2) What is its normal boiling point? 3) What is T of “dry ice bath”? 4) What phase does one have at T = 310 K? 5) Is slope (d. P/d. T) of solidliquid boundary “+” or “–”?
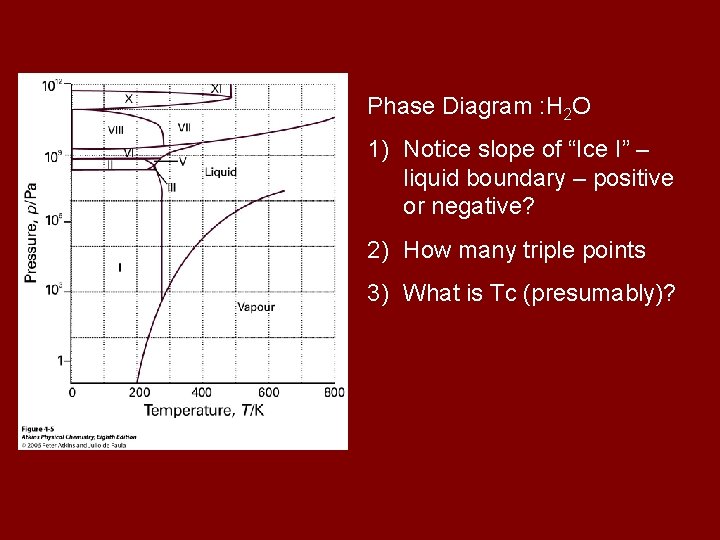
Phase Diagram : H 2 O 1) Notice slope of “Ice I” – liquid boundary – positive or negative? 2) How many triple points 3) What is Tc (presumably)?
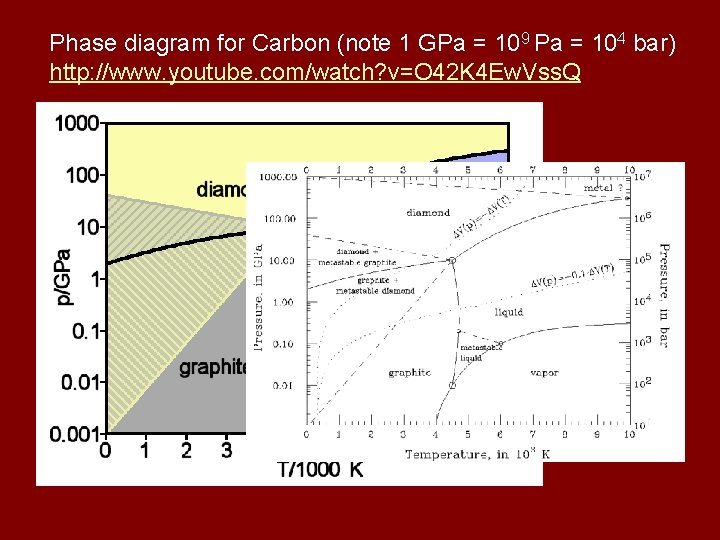
Phase diagram for Carbon (note 1 GPa = 109 Pa = 104 bar) http: //www. youtube. com/watch? v=O 42 K 4 Ew. Vss. Q
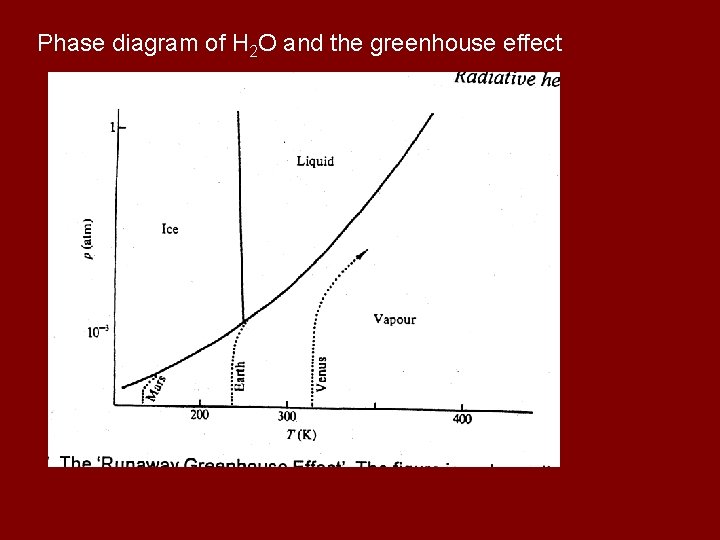
Phase diagram of H 2 O and the greenhouse effect
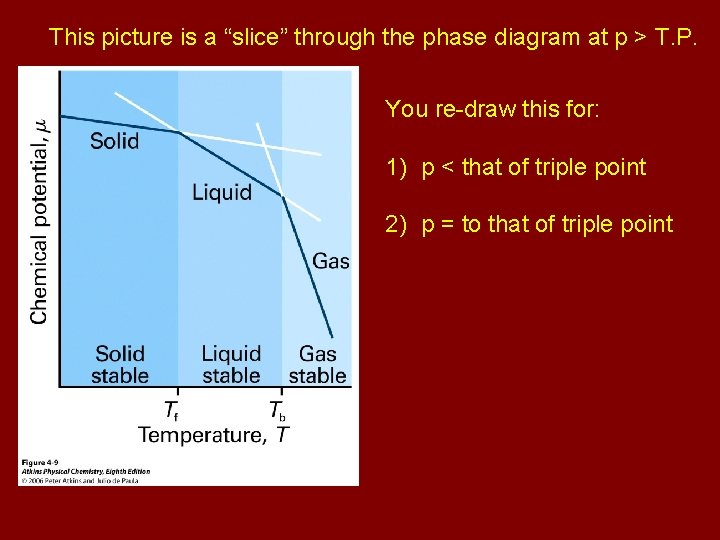
This picture is a “slice” through the phase diagram at p > T. P. You re-draw this for: 1) p < that of triple point 2) p = to that of triple point
- Slides: 9