Chem 336 338 Lab Lecture C 336 01

Chem 336 - 338 Lab Lecture C 336 -01 Organic Chemistry Laboratory II and C 338 -01 Organic Chemistry Laboratory II Honors

Dr. Margareta Séquin Office: Science 106 C; Office Hours: Tu 11 -12 pm, Th 1 -2 pm and by appt. e-mail: msequin @ sfsu. edu Phone: 415 -405 -0315

http: //userwww. sfsu. edu/~msequin

First half of semester: Syntheses and methods of evaluation Second half: C 336 “Unknowns”. C 338 Final Project
http: //userwww. sfsu. edu/~msequin


Prerequisites: C 336: passing grade (C or better) in Chem 333, 334; Chem 335 (can be taken concurrently). C 338: B or better. Prerequisites will be strictly applied. A student who withdraws from Chem 335 must also withdraw from Chem 336 or Chem 338. Text: “Chem 334, 336, and 338 Organic Chemistry Laboratory”, SFSU 2005, with Mayo, Pike, Trumper, "Microscale Organic Laboratory", 4 th Ed. ; Wiley Custom Ed. Use your lecture text for reference!

Grading C 336 and C 338: Mini. Quizzes at the end of lab lectures, 2 pts. each C 336 and C 338: One Midterm Exam (100 points) C 336: Final Exam (150 pts. ) C 338: Special Project Final Report, worth 150 pts. Lab reports: 20 points/ lab report 1 - 4. Lab reports V and VI will be combined and will be 30 pts. Unknown reports (C 336) will be 30 pts. each. Lab Notebooks will be checked regularly. Your total class grade will be calculated as follows: 40% Exams % (Midterm, plus Mini Quizzes, plus Final Exam or Project, %) 30% Lab reports % 30% Lab preparation, notebook, effort, attendance, participation. Total class grade: >/=90% A; 85 -<90% A-; 80 -<85% B+; 75 -<80% B; 70 -<75% B-; 65 <70% C+; 60 -<65% C; 55 -<60% C-; 50 -< 55% D+; 45 -<50% D; 40 <45 D-; <40% F

Exp. 1: Solvolysis of tert Butyl Chloride 1. IUPAC name for tert butyl chloride, for the alcohol product? 2. Why does changing the solvent medium affect the rate? 60%water: 40%acetone; 70%water: 30%acetone; 80%water: 20%acetone
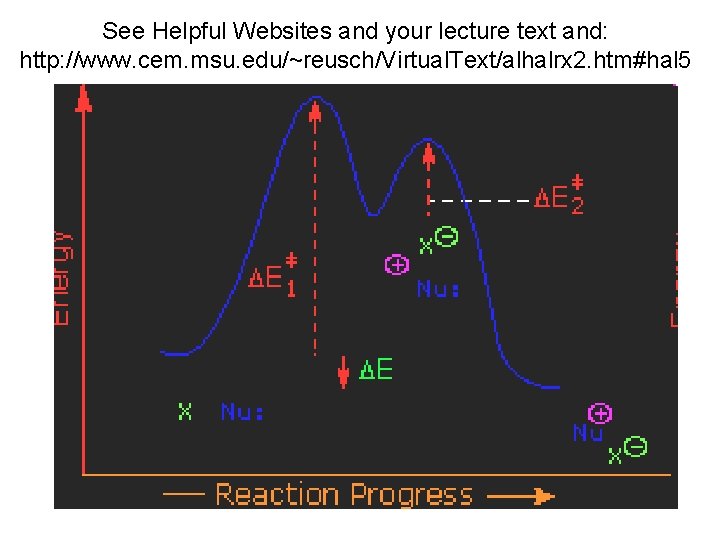
See Helpful Websites and your lecture text and: http: //www. cem. msu. edu/~reusch/Virtual. Text/alhalrx 2. htm#hal 5

Measuring the tenth–life in our reaction What is a possible by-product of our reaction? Does this affect our data? How would a one-third decrease of concentration of RX affect the reaction rate?

Calculations for Exp. 1: Solvolysis of tert Butyl Chloride p. 354 -355

Calculations for Exp. 1: Solvolysis 1. 2. 3. 4.
![Calculations for Exp. 1: Solvolysis at t 10%, [RCl] = 0. 90 [RCl]o 0. Calculations for Exp. 1: Solvolysis at t 10%, [RCl] = 0. 90 [RCl]o 0.](http://slidetodoc.com/presentation_image_h2/80fbef956904480dc2a0dfd23a1dac63/image-14.jpg)
Calculations for Exp. 1: Solvolysis at t 10%, [RCl] = 0. 90 [RCl]o 0. 90[RCl] o -ln = kt 10% [RCl]o -ln 0. 90 = 0. 1053 = kt 10% 0. 1053 t 10% = k Units of k? How could you calculate k by using the half life?

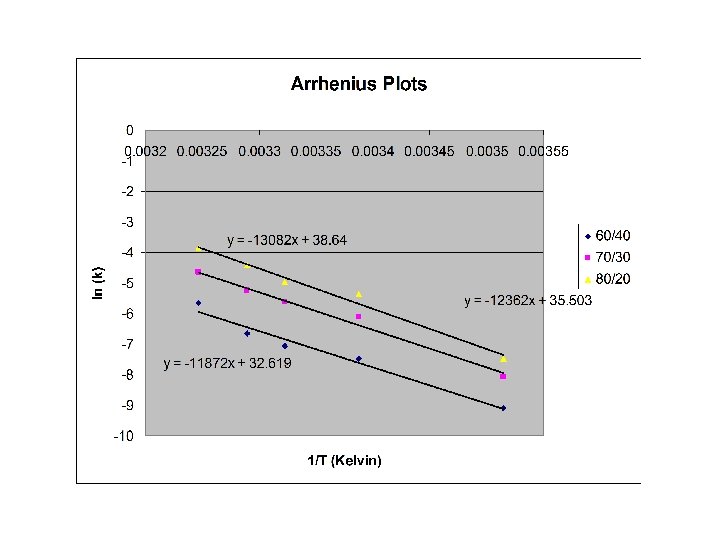
Arrhenius Equation and Calculation of Ea and A R=1. 987216 cal/K mol What does Ea tell us about the reaction? ln k = ln A -Ea/RT y= b + mx ln k Plot ln k vs. 1/T for each solvent mixture 1/T

Next time: • Kinetics (conclusion) • Friedel-Crafts (Exp. 2) • Mini. Quiz on today’s class (Exp. 1) Please fill out roll form and hand it in. Thank you.
- Slides: 17