CHEM 3310 Fuel Cell Change in Gibbs Free

CHEM 3310 Fuel Cell

Change in Gibbs Free Energy, G G is the free energy that is available to do useful work such as electrochemical work. G = - n. FE G < 0 Go = - n. FEo An electrochemical cell is able do work if its cell potential, E, is positive. where n is the number of moles of electrons that flow in the cell, F is Faraday’s constant, 96500 coulombs / mole of electrons, Eo is the standard voltage measured of the electrochemical cell. CHEM 3310 2

Change in Gibbs Free Energy, G G is the free energy that is available to do useful work such as electrochemical work. Go < 0 An electrochemical cell is able do work if its cell potential, Eo, is positive. Go = - n. FEo where n is the number of moles of electrons that flow in the cell, F is Faraday’s constant, 96500 coulombs / mole of electrons, Eo is the standard voltage measured of the electrochemical cell. CHEM 3310 3

Example: Hydrogen fuel cell Use solar energy to break apart water Solar panel CHEM 3310 Electrolyzer to generate H 2 and O 2 as fuel for the fuel cell. Hydroge n Fuel cell Load box 4

Example: Hydrogen fuel cell Electrolyzer Solar panel Hydroge n Fuel cell Load box Eo=1. 23 V Anode: 2 H 2 (g) 4 H+ (g) + 4 e- Cathode: O 2 (g) + 4 H+ (g) + 4 e- 2 H 2 O (l) CHEM 3310 2 H 2 (g) + O 2 (g) 2 H 2 O (l) Eo = 1. 23 V 5
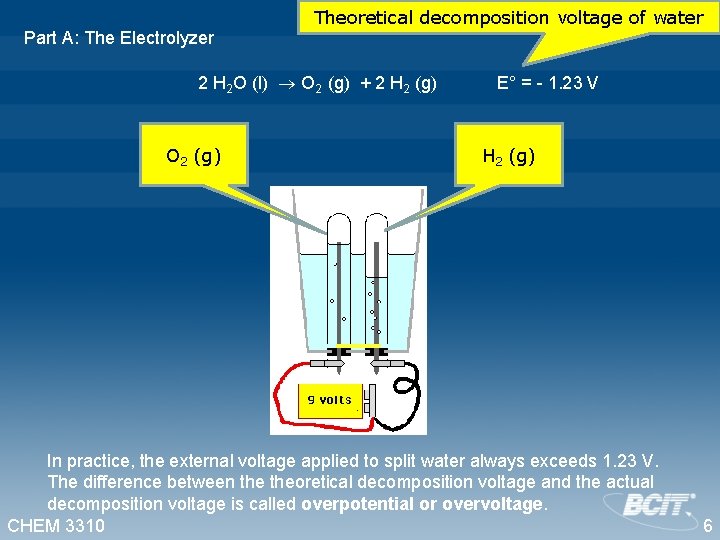
Part A: The Electrolyzer Theoretical decomposition voltage of water 2 H 2 O (l) O 2 (g) + 2 H 2 (g) O 2 (g) E° = - 1. 23 V H 2 (g) In practice, the external voltage applied to split water always exceeds 1. 23 V. The difference between theoretical decomposition voltage and the actual decomposition voltage is called overpotential or overvoltage. CHEM 3310 6

Part A: The Electrolyzer 2 H 2 O (l) O 2 (g) + 2 H 2 (g) E° = - 1. 23 V Adjust the power supply to obtain, roughly, the following current readings: 0. 01, 0. 02, 0. 03, 0. 04, 0. 05, 0. 06, 0. 08, 0. 10, 0. 15, 0. 20, 0. 25, 0. 30, 0. 35, 0. 40, 0. 45 A CHEM 3310 7
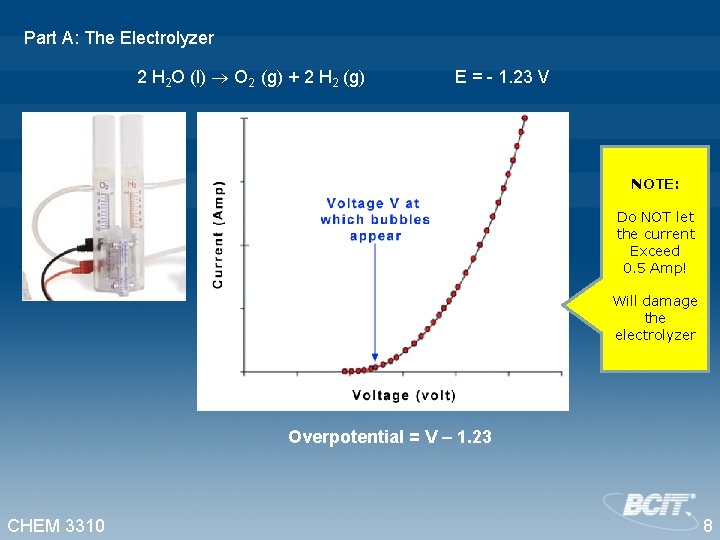
Part A: The Electrolyzer 2 H 2 O (l) O 2 (g) + 2 H 2 (g) E = - 1. 23 V NOTE: Do NOT let the current Exceed 0. 5 Amp! Will damage the electrolyzer Overpotential = V – 1. 23 CHEM 3310 8
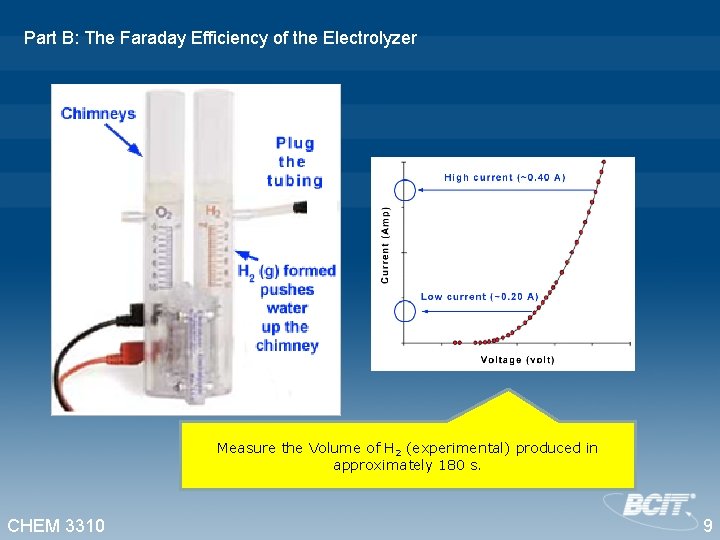
Part B: The Faraday Efficiency of the Electrolyzer Measure the Volume of H 2 (experimental) produced in approximately 180 s. CHEM 3310 9

Part B: The Faraday Efficiency, ŋ, of the Electrolyzer Cathode: 4 H+ (g) + 4 e- 2 H 2 (g) Anode: 2 H 2 O (l) O 2 (g) + 4 H+ (g) + 4 e 2 H 2 O (l) O 2 (g) + 2 H 2 (g) 2 moles of electrons per mole of H 2 O E = - 1. 23 V . • One mole of electrons has a charge equal to 96500 coulombs. • At 20 o. C, the molar volume of H 2 (g) is 24000 m. L. Determine ŋ for the each of the two currents. CHEM 3310 In commercial electrolyzers, the Faraday efficiency must be close to 1 (i. e. 100%). 10
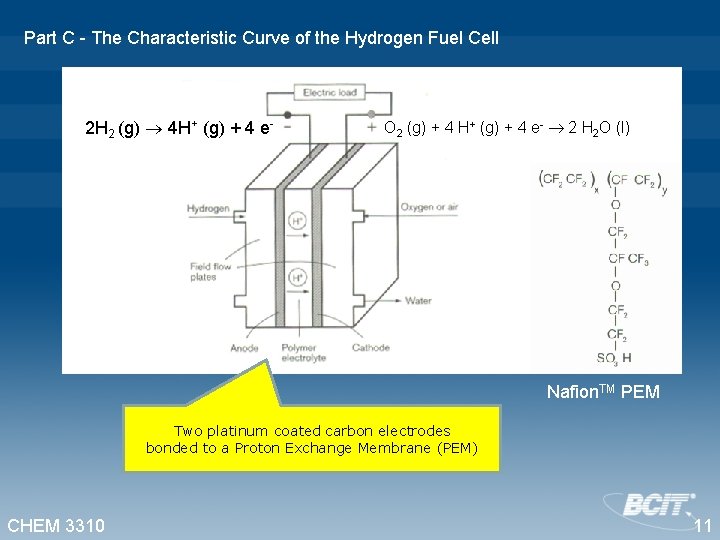
Part C - The Characteristic Curve of the Hydrogen Fuel Cell 2 H 2 (g) 4 H+ (g) + 4 e- O 2 (g) + 4 H+ (g) + 4 e- 2 H 2 O (l) Nafion. TM PEM Two platinum coated carbon electrodes bonded to a Proton Exchange Membrane (PEM) CHEM 3310 11
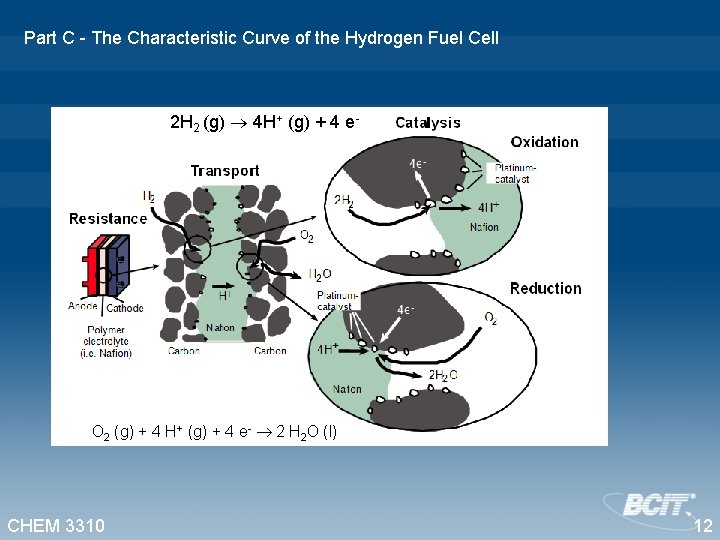
Part C - The Characteristic Curve of the Hydrogen Fuel Cell 2 H 2 (g) 4 H+ (g) + 4 e- O 2 (g) + 4 H+ (g) + 4 e- 2 H 2 O (l) CHEM 3310 12
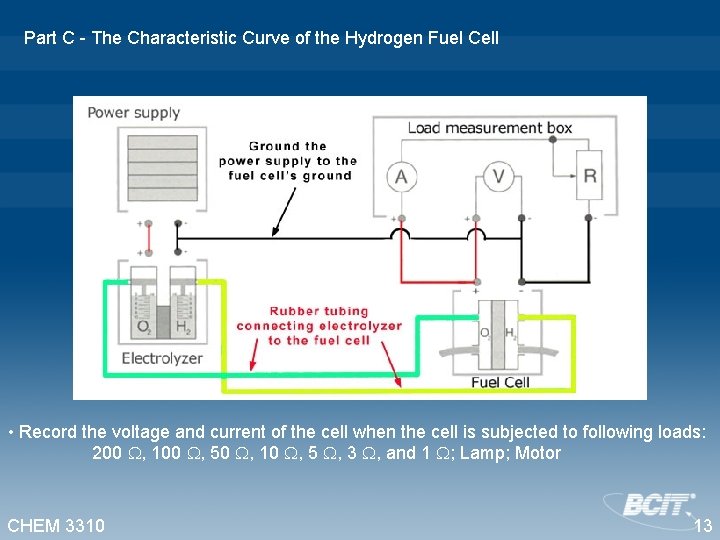
Part C - The Characteristic Curve of the Hydrogen Fuel Cell • Record the voltage and current of the cell when the cell is subjected to following loads: 200 , 100 , 50 , 10 , 5 , 3 , and 1 ; Lamp; Motor CHEM 3310 13
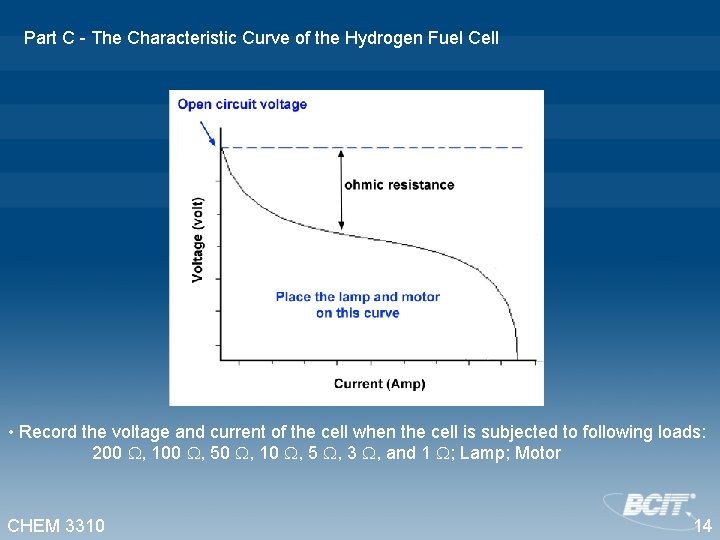
Part C - The Characteristic Curve of the Hydrogen Fuel Cell • Record the voltage and current of the cell when the cell is subjected to following loads: 200 , 100 , 50 , 10 , 5 , 3 , and 1 ; Lamp; Motor CHEM 3310 14
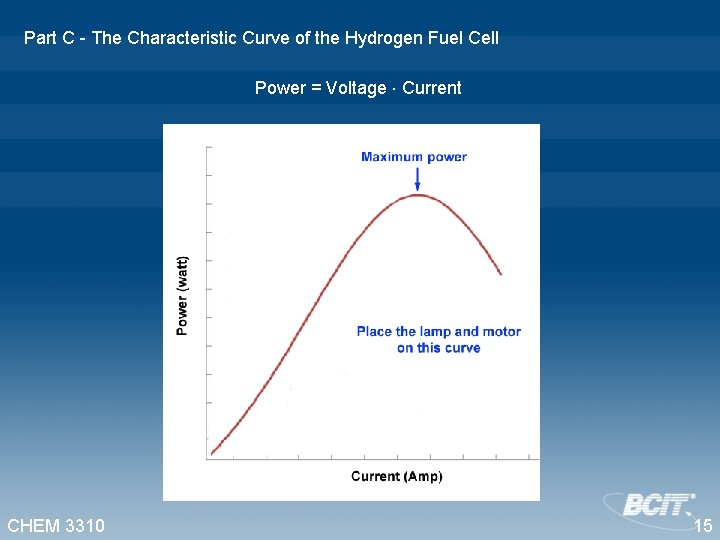
Part C - The Characteristic Curve of the Hydrogen Fuel Cell Power = Voltage Current CHEM 3310 15
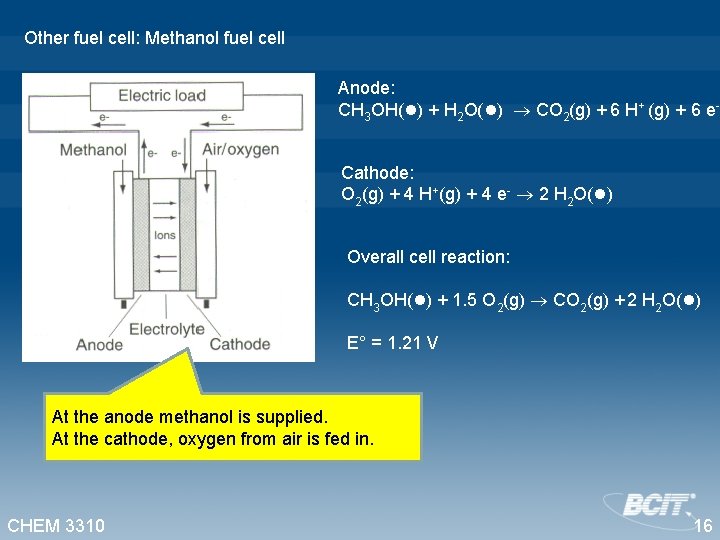
Other fuel cell: Methanol fuel cell Anode: CH 3 OH(l) + H 2 O(l) CO 2(g) + 6 H+ (g) + 6 e. Cathode: O 2(g) + 4 H+(g) + 4 e- 2 H 2 O(l) Overall cell reaction: CH 3 OH(l) + 1. 5 O 2(g) CO 2(g) + 2 H 2 O(l) E° = 1. 21 V At the anode methanol is supplied. At the cathode, oxygen from air is fed in. CHEM 3310 16
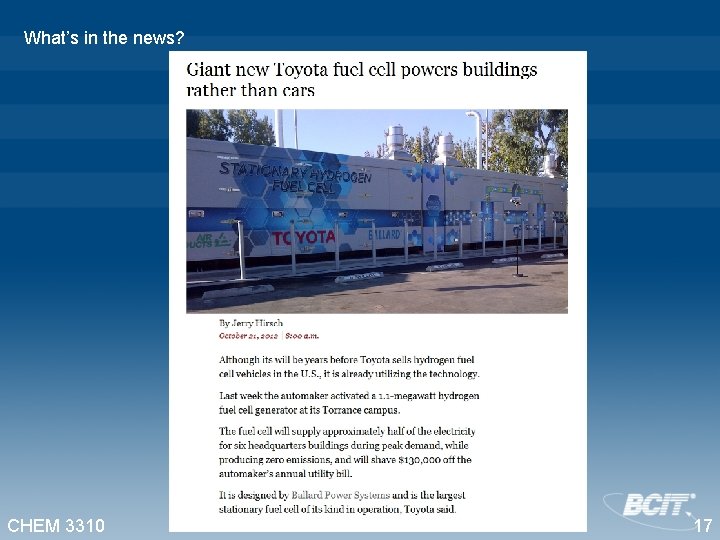
What’s in the news? CHEM 3310 17

What’s in the news? CHEM 3310 18
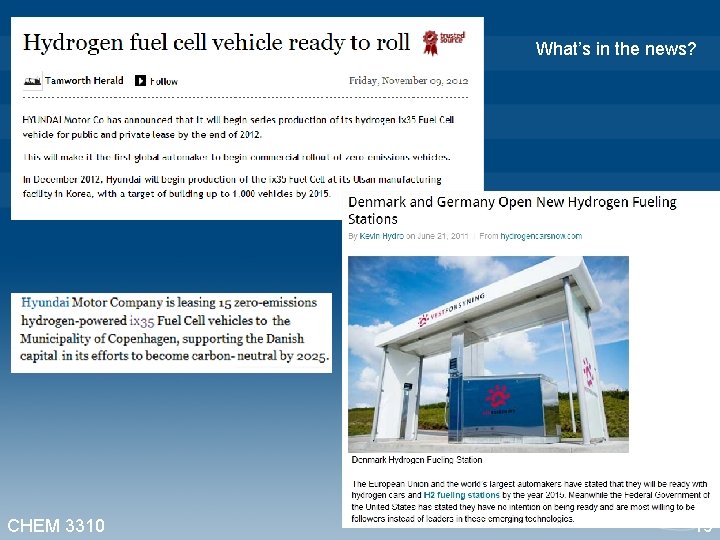
What’s in the news? CHEM 3310 19

- Slides: 21