Chem 31 1011 Lecture Announcements Statistical Calculations Lab

Chem. 31 – 10/11 Lecture

Announcements • Statistical Calculations Lab – Due today • AA Lab – Tap water data posted online • Today’s Lecture – Chapter 6 • Parts missed last time • Sparingly Soluble Salts – Solubility in water – Solubility in common ion – Precipitation + Selective Precipitation • Complex Ions

Example question The reaction N 2(g) + O 2 (g) ↔ 2 NO(g) has a positive DH. Under what conditions is this process spontaneous? - all temperatures - low temperatures - high temperatures - never

Thermodynamics ΔG and Equilibrium ΔG = ΔG° + RTln. Q Q = Reaction Quotient (for A ↔ B, Q = [B]/[A]) At equilibrium, ΔG = 0 and Q = K ΔG° = -RTln. K

Solubility Product Problems - Solubility in Water Example: solubility of Mg(OH)2 in water Solubility defined as mol Mg(OH)2 dissolved/L sol’n or g Mg(OH)2 dissolved/L sol’n or other units Use ICE approach: Mg(OH)2(s) ↔ Mg 2+ + 2 OHInitial 0 0 Change +x +2 x Equilibrium x 2 x Note: x = [Mg 2+] = solubility

Solubility Product Problems - Solubility of Mg(OH)2 in water Equilibrium Equation: Ksp = [Mg 2+][OH-]2 Ksp = 7. 1 x 10 -12 = x(2 x)2 = 4 x 3 (see Appendix F for Ksp) x = (7. 1 x 10 -12/4)1/3 = 1. 2 x 10 -4 M Solubility = 1. 2 x 10 -4 M = [Mg 2+] Conc. [OH-] = 2 x = 2. 4 x 10 -4 M

Solubility Product Problems - Solubility of Mg(OH)2 in Common If we dissolve Mg(OH)2 in a common ion (OH- or Mg 2+), from Le Châtelier’s principle, we know the solubility will be reduced Example 1) What is the solubility of Mg(OH)2 in a p. H = 11. 0 buffer? No ICE table needed because, from p. H, we know [OH-]eq and buffer means dissolution of Mg(OH)2 doesn’t affect p. H.
![Solubility Product Problems - Solubility of Mg(OH)2 at p. H 11 – cont. [H+] Solubility Product Problems - Solubility of Mg(OH)2 at p. H 11 – cont. [H+]](http://slidetodoc.com/presentation_image_h2/1487cb6ed9441b5cc6c0b907c303d12a/image-8.jpg)
Solubility Product Problems - Solubility of Mg(OH)2 at p. H 11 – cont. [H+] = 10 -p. H = 10 -11 M and [OH-] = Kw/[H+] = 10 -3 M Ksp = [Mg 2+][OH-]2 Moles Mg(OH)2 dissolved = moles Mg 2+ [Mg 2+] = Ksp/[OH-]2 = 7. 1 x 10 -12/(10 -3)2 [Mg 2+] = 7 x 10 -6 M

Solubility Product Problems - Solubility of Mg(OH)2 in Common Ion Example 2) Solubility of Mg(OH)2 in 5. 0 x 103 M Mg. Cl. 2

Precipitations Used for Separations Example: If we wanted to know the concentrations of Ca 2+ and Mg 2+ in a water sample. EDTA titration gives [Ca 2+] + [Mg 2+]. However, if we could selectively remove Ca 2+ or Mg 2+ (e. g. through titration) and re-titrate, we could determine the concentrations of each ion. Determine if it is possible to remove 99% of Mg 2+ through precipitation as Mg(OH)2 without precipitating out any Ca(OH)2 if a tap water solution initially has 1. 0 x 10 -3 M Mg 2+ and 1. 0 x 10 -3 M Ca 2+.

Solubility Product Problems Precipitation Problems What occurs if we mix 50 m. L of 0. 020 M Ba. Cl 2 with 50 m. L of 3. 0 x 10 -4 M (NH 4)2 SO 4? Does any solid form from the mixing of ions? What are the concentrations of ions remaining?

Complex Ions Example Reaction: Ag+ + 2 NH 3(aq) ↔ Ag(NH 3)2+ Metal Ligand Complex Ion Why does reaction occur? Metal is a Lewis acid (electron pair acceptor) NH 3 is a Lewis base (electron pair donator) Metal-ligand bonds are intermediate strength
Complex Ions – Why Study? Crown ether (12 -crown-4) • Useful in separations – Complexed metals become more organic soluble • Effects on metal solubility Na+ (e. g. addition of NH 3 on Ag. Cl solubility) Crown ether added • Complexometric titrations (e. g. water hardness titration) • Some Complexes are Colored Diethyl ether (use as indicators or for spectroscopic measurements) Sodium conc. given by gray shading water

Complex Ions Step-wise vs. full reactions: Example: addition of NH 3 to Ag+ Reaction occurs in steps: 1) Ag+ + NH 3(aq) ↔ Ag. NH 3+ K 1 (= β 1) 2) Ag. NH 3+ + NH 3(aq) ↔ Ag(NH 3)2+ K 2 Net) Ag+ + 2 NH 3(aq) ↔ Ag(NH 3)2+ β 2 = K 1·K 2

Complex Ions Due to large exponents on ligand concentration, a small change in ligand concentration has a big effect on how metal exists Example: Al 3+ + 3 C 2 O 42 - ↔ Al(C 2 O 4)33 - β 3 = 4. 0 x 1015 [C 2 O 42 -] [Al(C 2 O 4)33 -]/[Al 3+] 10 -4 M 4000 10 -5 M 4 10 -6 M 0. 004

Complex Ions – “U” Shaped Solubility Curves Many sparingly soluble salts release cations and anions that form complexes with each other Example: calcium oxalate (Ca. C 2 O 4) Ca. C 2 O 4(s) ↔ Ca 2+ + C 2 O 42 - (Ksp = 1. 3 x 10 -8 M) increased [C 2 O 42 -] decreases Ca 2+ solubility for above reaction only, but. . . Ca 2+ + C 2 O 42 - ↔ Ca. C 2 O 4(aq) K 1 = 46 Ca. C 2 O 4(aq) + C 2 O 42 - ↔ Ca(C 2 O 4)22 - K 2 = 490 β 2 = K 1·K 2 = 2. 3 x 104 = [Ca(C 2 O 4)22 -]/([Ca 2+][C 2 O 42 -]2)
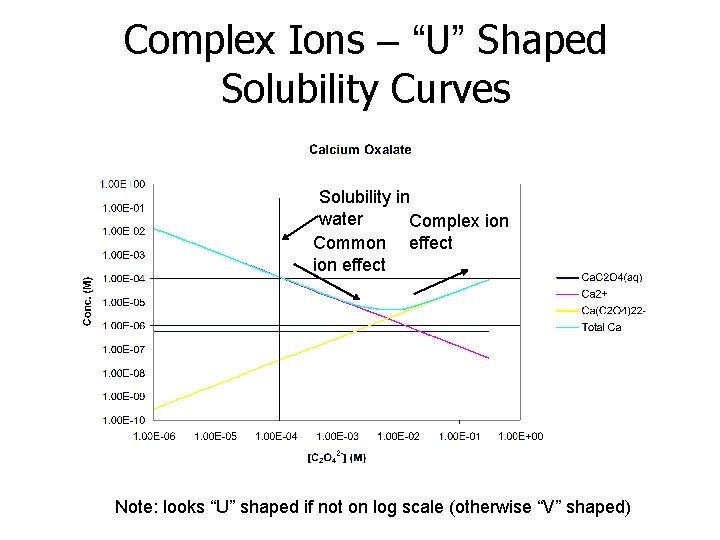
Complex Ions – “U” Shaped Solubility Curves Solubility in water Complex ion Common effect ion effect Note: looks “U” shaped if not on log scale (otherwise “V” shaped)

Some Questions 1. 2. 3. 4. In the reaction: Ca 2+ + Y 4 - ↔ Ca. Y 2 - (where Y 4 - = EDTA), which species is the Lewis acid? List two applications in which the formation of a complex ion would be useful for analytical chemists. List two applications in the lab in which you used or are using complex ions. Ag. CN is a sparingly soluble salt. However, a student observed that adding a little of a Na. CN solution to a saturated solution of Ag. CN did not result in more precipitation of solid. Addition of more Na. CN solution resulted in total dissolution of the Ag. CN. Explain what is happening.
- Slides: 18