Chem 1140 Catalysis General Principles ZieglerNatta Olefin Polymerization


















- Slides: 29

Chem 1140; Catalysis • General Principles • Ziegler-Natta Olefin Polymerization • Mechanism of Hydrogenation with Wilkinson’s Catalyst • Asymmetric Hydrogenation

Catalysis • Catalysts increase reaction rate without themselves being changed • Can accelerate a reaction in both directions • Do not affect the state of equilibrium of reaction – simply allow equilibrium to be reached faster

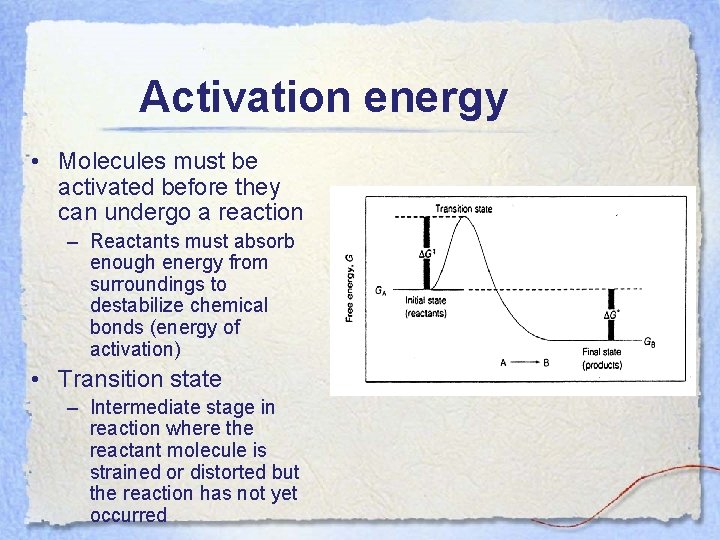
Activation energy • Molecules must be activated before they can undergo a reaction – Reactants must absorb enough energy from surroundings to destabilize chemical bonds (energy of activation) • Transition state – Intermediate stage in reaction where the reactant molecule is strained or distorted but the reaction has not yet occurred

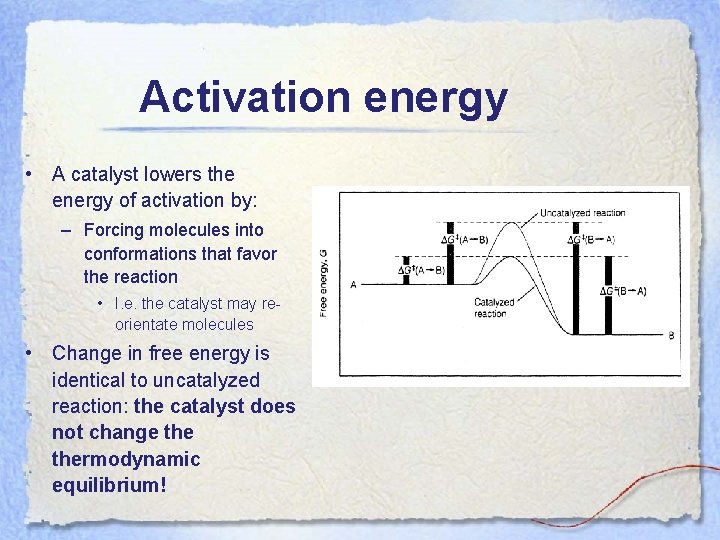
Activation energy • A catalyst lowers the energy of activation by: – Forcing molecules into conformations that favor the reaction • I. e. the catalyst may reorientate molecules • Change in free energy is identical to uncatalyzed reaction: the catalyst does not change thermodynamic equilibrium!

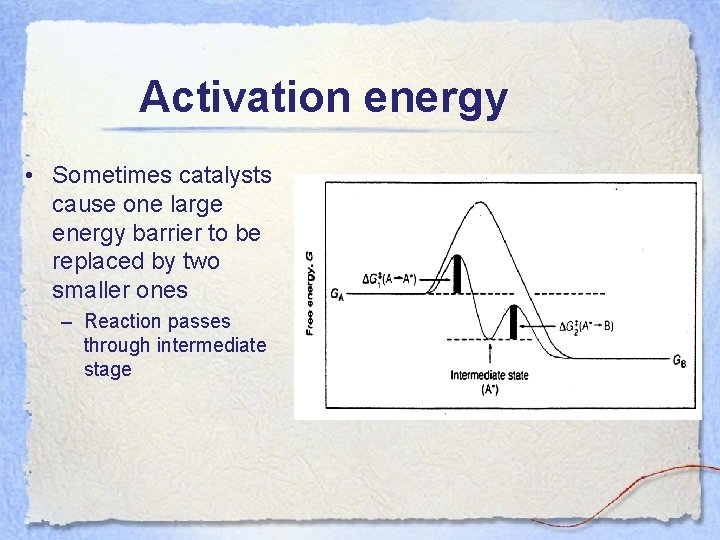
Activation energy • Sometimes catalysts cause one large energy barrier to be replaced by two smaller ones – Reaction passes through intermediate stage

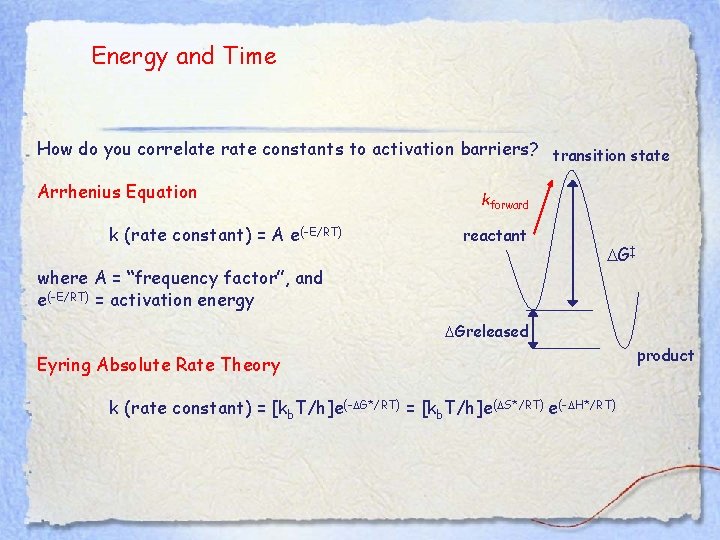
Energy and Time How do you correlate rate constants to activation barriers? transition state Arrhenius Equation k (rate constant) = A e(-E/RT) kforward reactant where A = “frequency factor”, and e(-E/RT) = activation energy DG‡ DGreleased Eyring Absolute Rate Theory k (rate constant) = [kb. T/h]e(-DG*/RT) = [kb. T/h]e(DS*/RT) e(-DH*/RT) product

Ziegler-Natta Catalysis of Alkene Polymerization A typical Ziegler-Natta catalyst is a combination of Ti. Cl 4 and (CH 3 CH 2)2 Al. Cl, or Ti. Cl 3 and (CH 3 CH 2)3 Al. Many Ziegler-Natta catalyst combinations include a metallocene.

Ziegler’s Discovery • 1953 K. Ziegler, E. Holzkamp, H. Breil & H. Martin • Angew. Chem. 67, 426, 541 (1955); 76, 545 (1964). + Ni(Ac. Ac) + Cr(acac) + Zr(acac) Same result White Ppt. (Not reported by Holzkamp) White Ppt. (Eureka! reported by Breil)

Natta’s Discovery • • • 1954 Giulio Natta, P. Pino, P. Corradini, and F. Danusso J. Am. Chem. Soc. 77, 1708 (1955) Crystallographic Data on PP J. Polym. Sci. 16, 143 (1955) Polymerization described in French Isotactic Syndiotactic Ziegler and Natta won Nobel Prize in 1963

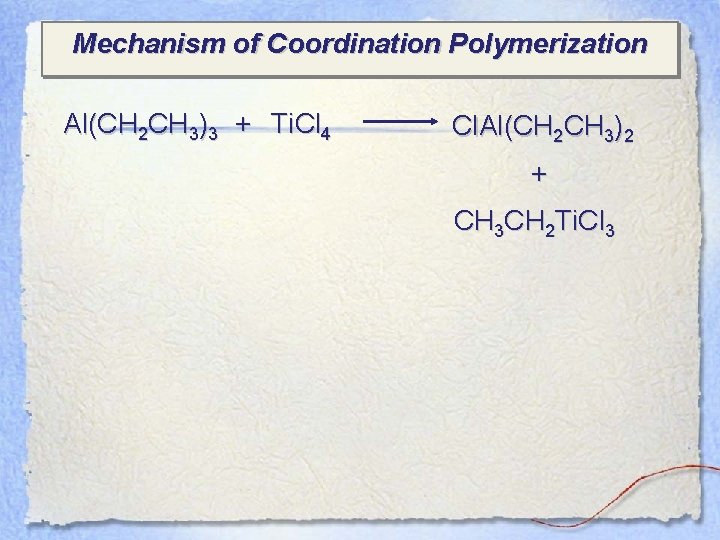
Mechanism of Coordination Polymerization Al(CH 2 CH 3)3 + Ti. Cl 4 Cl. Al(CH 2 CH 3)2 + CH 3 CH 2 Ti. Cl 3

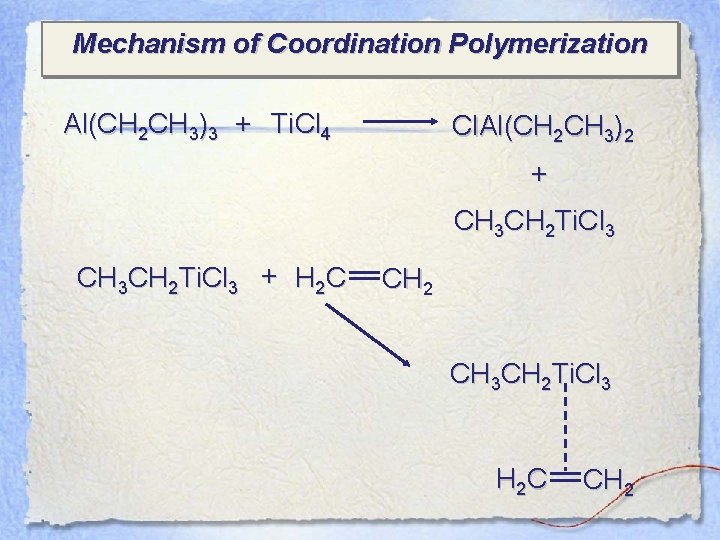
Mechanism of Coordination Polymerization Al(CH 2 CH 3)3 + Ti. Cl 4 Cl. Al(CH 2 CH 3)2 + CH 3 CH 2 Ti. Cl 3 + H 2 C CH 2 CH 3 CH 2 Ti. Cl 3 H 2 C CH 2

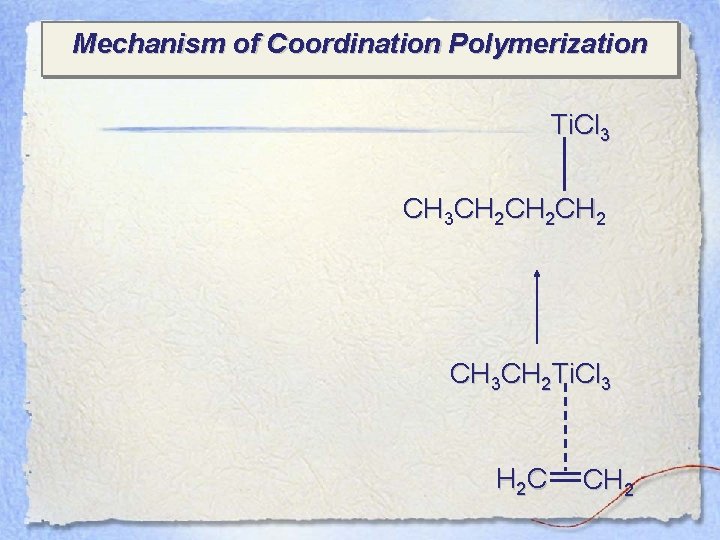
Mechanism of Coordination Polymerization Ti. Cl 3 CH 3 CH 2 CH 2 CH 3 CH 2 Ti. Cl 3 H 2 C CH 2

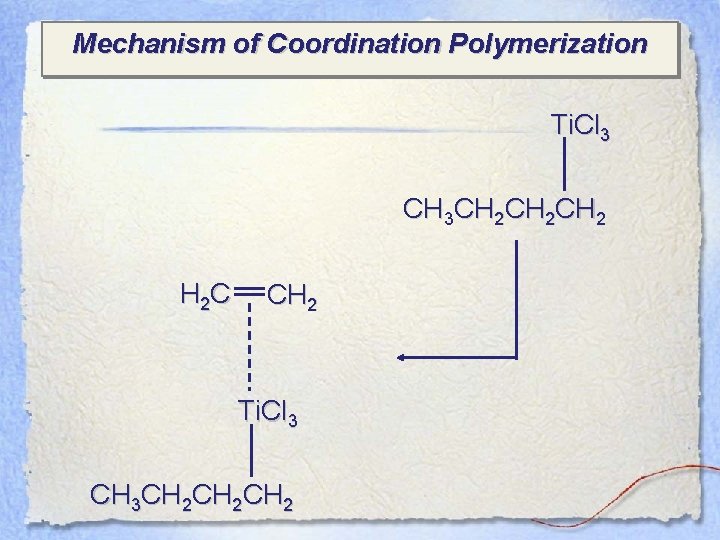
Mechanism of Coordination Polymerization Ti. Cl 3 CH 3 CH 2 CH 2 H 2 C CH 2 Ti. Cl 3 CH 3 CH 2 CH 2

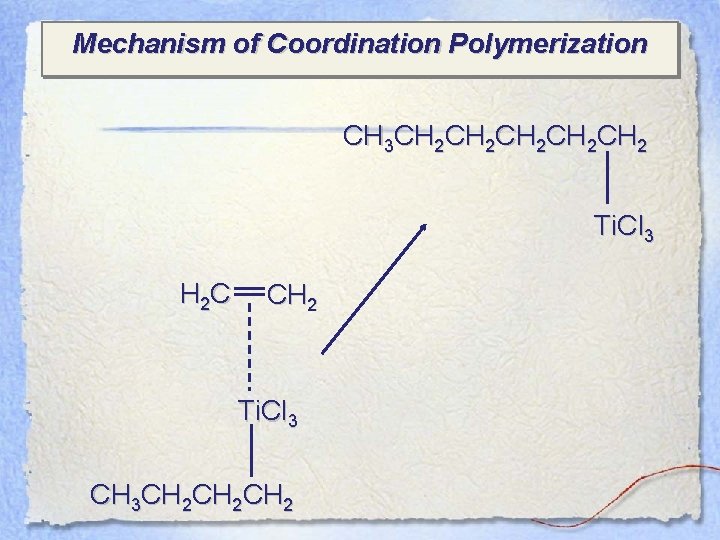
Mechanism of Coordination Polymerization CH 3 CH 2 CH 2 CH 2 Ti. Cl 3 H 2 C CH 2 Ti. Cl 3 CH 3 CH 2 CH 2

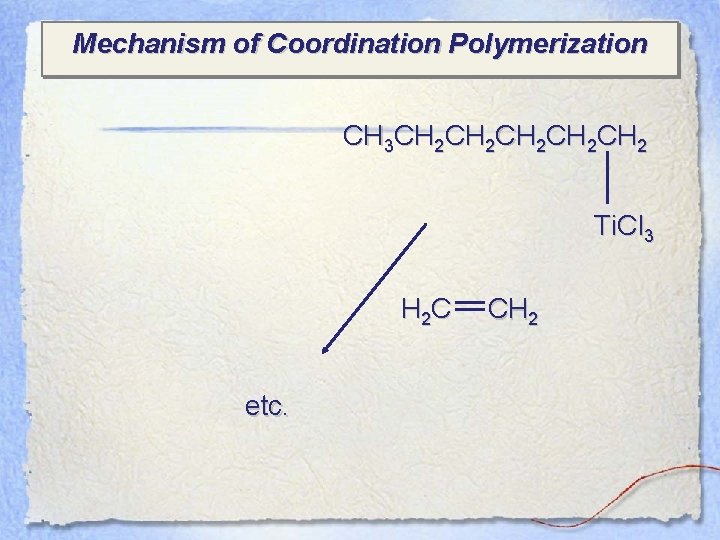
Mechanism of Coordination Polymerization CH 3 CH 2 CH 2 CH 2 Ti. Cl 3 H 2 C etc. CH 2

General Composition of Catalyst System Group I – III Metals Al. Et 3 Et 2 Al. Cl Et. Al. Cl 2 i-Bu 3 Al Et 2 Mg Et 2 Zn Et 4 Pb Transition Metals Additives Ti. Cl 4 a, g, d Ti. Cl 3 Mg. Cl 2 Support VCl 3, Vo. CL 3, V(Ac. Ac)3 Titanocene dichloride Ti(Oi. Bu)4 H 2 O 2, H 2 O (Mo, Cr, Zr, W, Mn, Ni) HMPA, DMF R-OH Phenols R 3 N, R 2 O, R 3 P Aryl esters

Kaminsky Catalyst System W. Kaminsky et. al. Angew. Chem. Eng. Ed. 19, 390, (1980); Angew. Chem. 97, 507 (1985) Al: Zr = 1000 Me = Ti, Zr, Hf Atactic polypropylene Activity = 106 g/mol Zr Linear HD PE Activity = 107 g/mol Zr

Methylaluminoxane: the Key Cocatalyst n = 10 -20 MAO Proposed structure

Nature of active catalyst MAO Transition metal alkylation Ionization to form active sites Noncoordinating Anion, NCA

Alkene Hydrogenation with Wilkinson’s Catalyst

Mechanism

Enantiomerically Enriched Phosphines

Asymmetric Hydrogenation

Asymmetric Hydrogenation

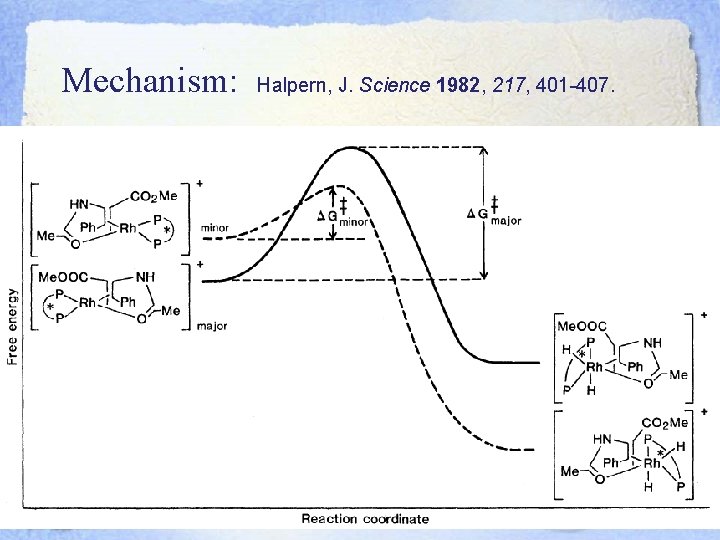
Mechanism: Halpern, J. Science 1982, 217, 401 -407.

Mechanism: Halpern, J. Science 1982, 217, 401 -407.

Mechanism: Halpern, J. Science 1982, 217, 401 -407.

Mechanism: Halpern, J. Science 1982, 217, 401 -407.

Mechanism: Halpern, J. Science 1982, 217, 401 -407.