Chem 108 Saturated Hydrocarbons Alkanes Chapter 2 General

Chem. 108 Saturated Hydrocarbons: Alkanes Chapter 2
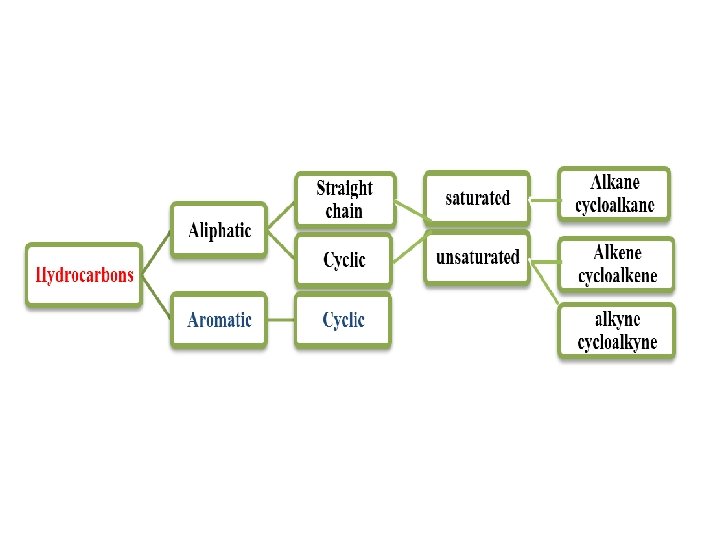

General Molecular Formula of Hydrocarbons (Homologous Series) Alkanes Cn. H 2 n+2 Saturated Cycloalkanes Cn. H 2 n (containing a single ring) Alkenes Cn. H 2 n (containing one double bond) Alkynes Cn. H 2 n-2 (containing one triple bond)
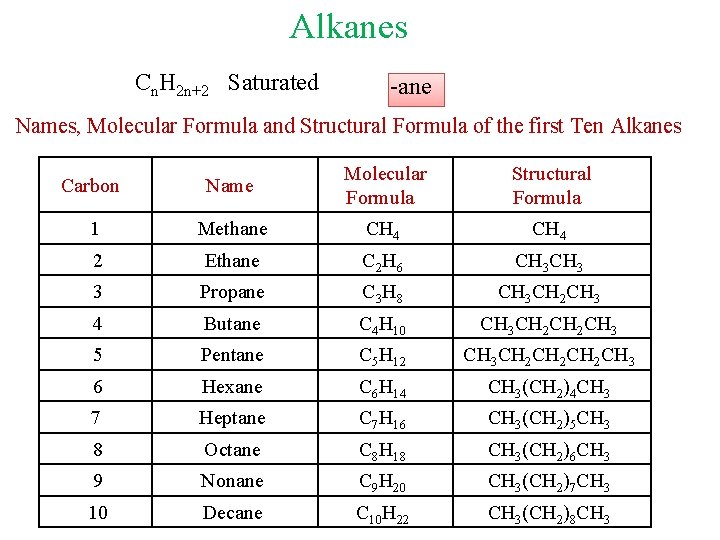
Alkanes Cn. H 2 n+2 Saturated -ane Names, Molecular Formula and Structural Formula of the first Ten Alkanes Carbon Name Molecular Formula Structural Formula 1 Methane CH 4 2 Ethane C 2 H 6 CH 3 3 Propane C 3 H 8 CH 3 CH 2 CH 3 4 Butane C 4 H 10 CH 3 CH 2 CH 3 5 Pentane C 5 H 12 CH 3 CH 2 CH 2 CH 3 6 Hexane C 6 H 14 CH 3(CH 2)4 CH 3 7 Heptane C 7 H 16 CH 3(CH 2)5 CH 3 8 Octane C 8 H 18 CH 3(CH 2)6 CH 3 9 Nonane C 9 H 20 CH 3(CH 2)7 CH 3 10 Decane C 10 H 22 CH 3(CH 2)8 CH 3
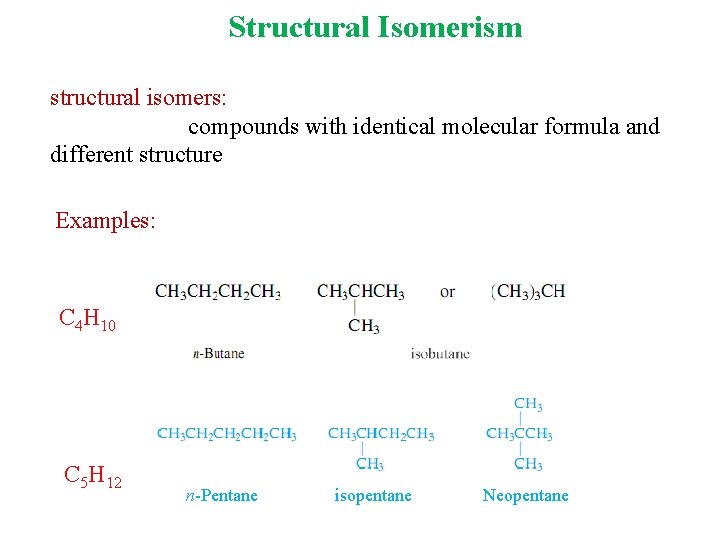
Structural Isomerism structural isomers: compounds with identical molecular formula and different structure Examples: C 4 H 10 C 5 H 12 n-Pentane isopentane Neopentane
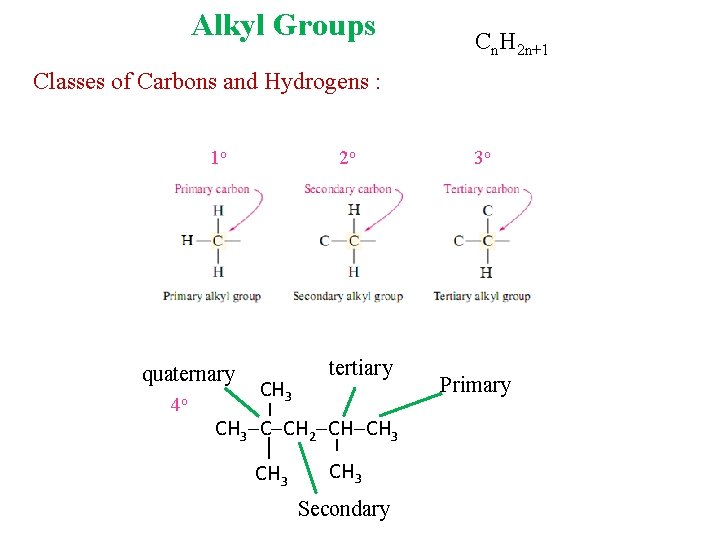
Alkyl Groups Cn. H 2 n+1 Classes of Carbons and Hydrogens : 1 o quaternary 4 o 2 o CH 3 tertiary CH 3 C CH 2 CH CH 3 Secondary 3 o Primary
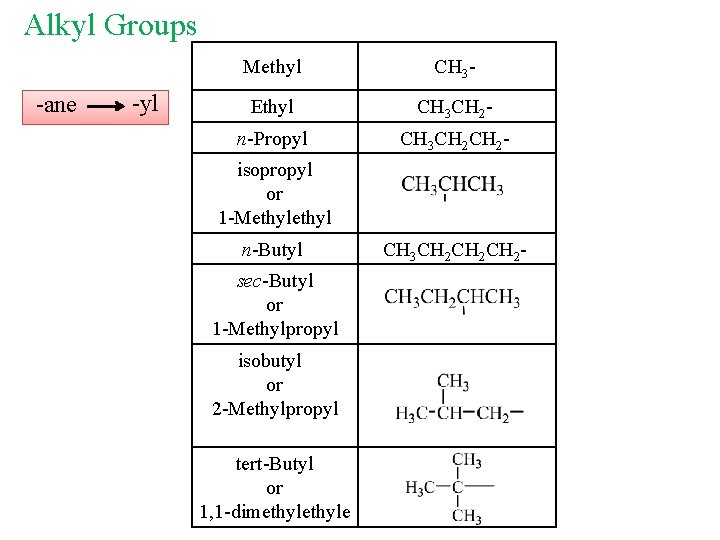
Alkyl Groups -ane -yl Methyl CH 3 - Ethyl CH 3 CH 2 - n-Propyl CH 3 CH 2 - isopropyl or 1 -Methyl n-Butyl sec-Butyl or 1 -Methylpropyl isobutyl or 2 -Methylpropyl tert-Butyl or 1, 1 -dimethyle CH 3 CH 2 CH 2 -

The IUPAC System of Nomenclature 1. Identifying the parent hydrocarbon chain (the longest one) 2. Numbering the chain (starting at the end that a side chain is nearer from ) 3. Listing the side-chains before the of parent chain (in alphatbetical order, giving the number of the carbon atom of the parent chain)
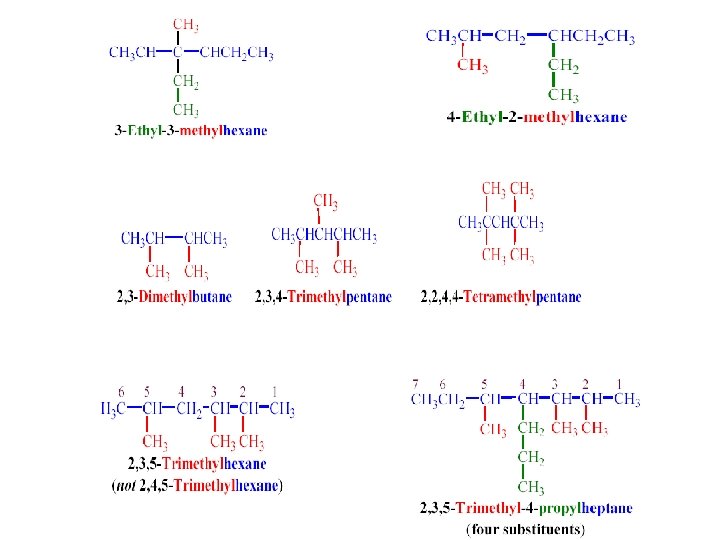

If any other substituents are found on the parent chain, all these substituents are arranged alphabetically. Ø Ø Ø -NO 2 nitro - NH 2 amino -CN cyano - Cl Chloro -Br bromo - I iodo
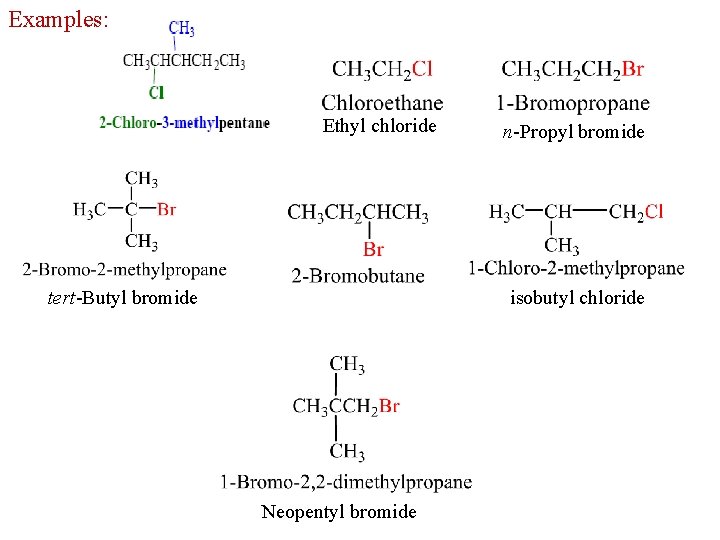
Examples: Ethyl chloride n-Propyl bromide isobutyl chloride tert-Butyl bromide Neopentyl bromide

Homework 1 - Indicate whether the following pairs of structures are (1) the same, (2) structural isomers, or (3) entirely unrelated.

2 - Give IUPC names for the following compounds. 3 - Write structural formulas for the following compounds. A. 2, 3 -Dimethyl-4 -ethylhexane. B. 2, 4 -Dimethyl-5 -ethyl-4 -t-butyleheptane. C. 3, 4 -Dimethyl-5 -ethyl-6 -isopropylnonane.

Physical properties of alkanes A Physical States and Solubilities C 1 -C 4 colorless gases C 5 -C 17 liquids with characteristic odor C 20 and more odorless waxy materials Alkanes are nonpolar compounds. Thus alkanes are soluble in the nonpolar solvents such as carbon tetrachloride (CCl 4) and benzene (C 6 H 6), but they are insoluble in polar solvents such as water.

B Boiling Points The boiling points of the normal alkanes increase with increasing molecular weight. Branching of the alkane chain lowers the boiling point. Example: C Melting Points Generally, melting point increases as molecular weight increases, but with no particular pattern.
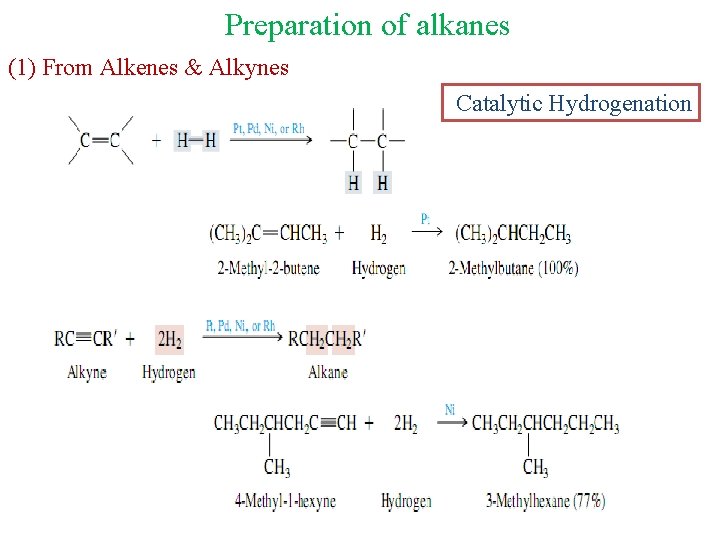
Preparation of alkanes (1) From Alkenes & Alkynes Catalytic Hydrogenation
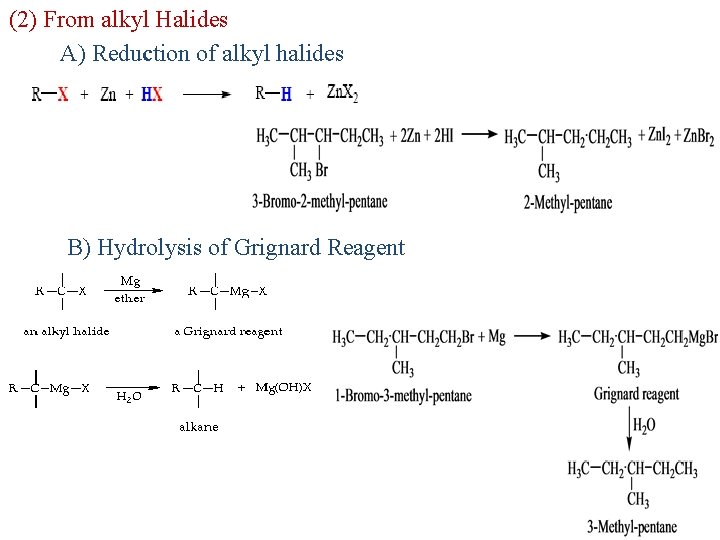
2 (2) From alkyl Halides A) Reduction of alkyl halides B) Hydrolysis of Grignard Reagent
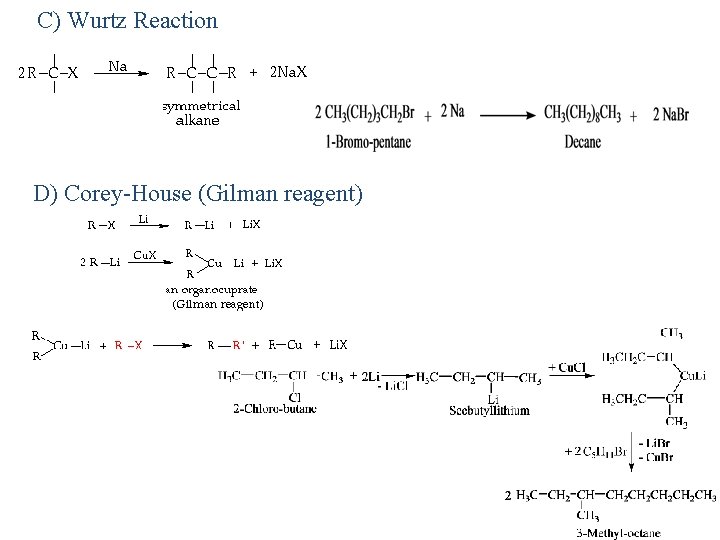
C) Wurtz Reaction D) Corey-House (Gilman reagent)

Reactions of alkanes (1) Halogenation (2) Combustion

Selectivity in Halogenation Reactions
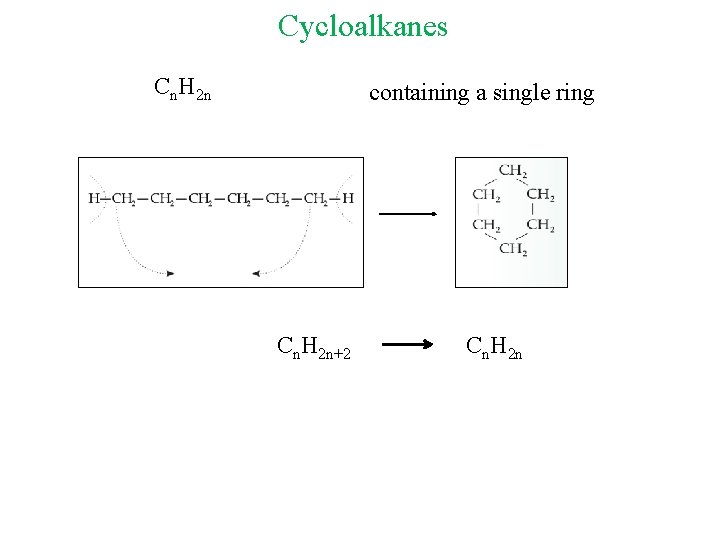
Cycloalkanes Cn. H 2 n containing a single ring Cn. H 2 n+2 Cn. H 2 n
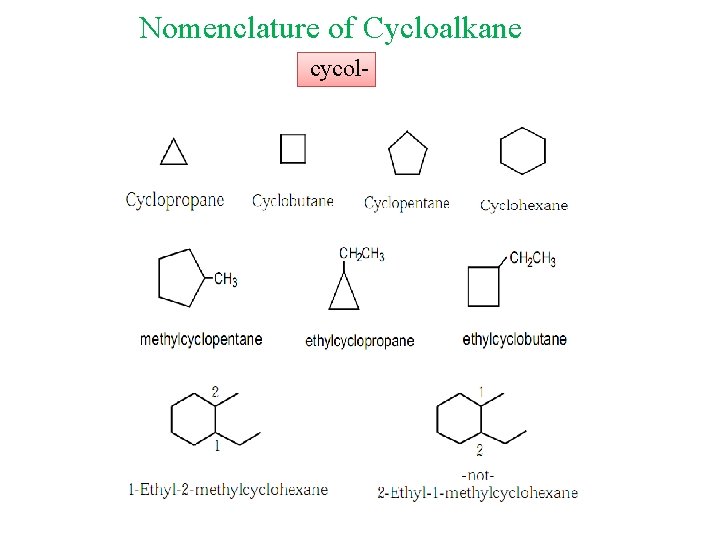
Nomenclature of Cycloalkane cycol-

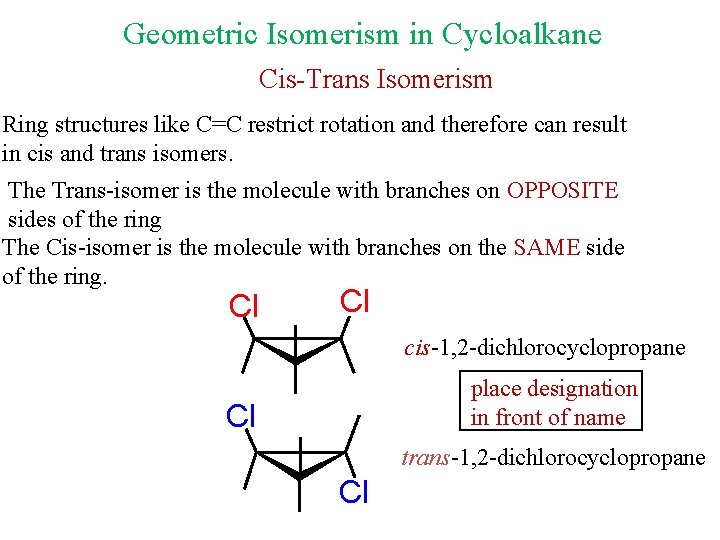
Geometric Isomerism in Cycloalkane Cis-Trans Isomerism Ring structures like C=C restrict rotation and therefore can result in cis and trans isomers. The Trans-isomer is the molecule with branches on OPPOSITE sides of the ring The Cis-isomer is the molecule with branches on the SAME side of the ring. Cl Cl cis-1, 2 -dichlorocyclopropane place designation in front of name Cl trans-1, 2 -dichlorocyclopropane Cl

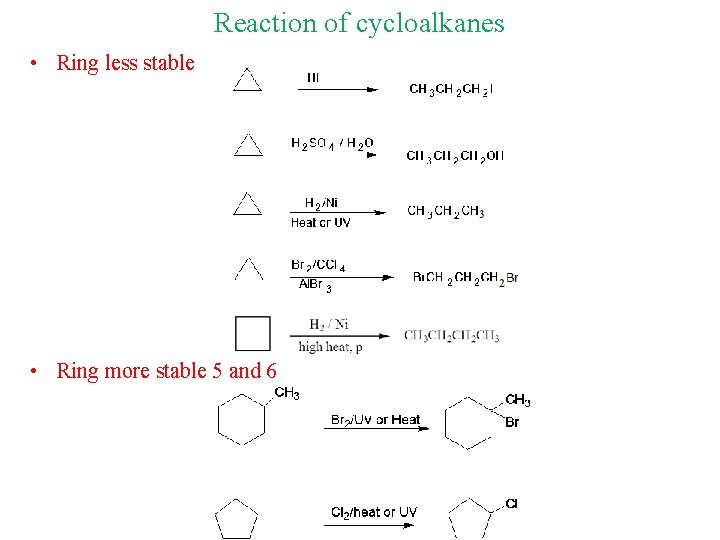
Reaction of cycloalkanes • Ring less stable • Ring more stable 5 and 6

Homework 1 - Arrange the following compounds in order of increasing boiling points : n-hexane; 2, 2 -dimethylbutane; 2 -methylpentane. 2 - Write the condensed structural formula for each of named compounds, and give the correct name for each. a) 1, 4 -Dimethylcyclobutane b) 2, 2 -Dichloro-5 -methylcyclohexane c) Cis-1, 3 -Dimethylcyclopropane d) 1, 1 -Dibromo-3 -methylcyclopentane (note: There are no cis and trans in this case. Why)
- Slides: 27