Chem 108 Lab Week 9 Sign in Confer

Chem 108: Lab Week 9 Sign in Confer with partners from last week & week before. DO: Alcohol Distillation To do with Fermentation / Chemical Reactions partner. Turn in Chemical Reactions Report form; (Confer with Molview Group if necessary) Turn in Molview Modeling form with Molview group.

DUE Today Chemical Reactions One report with everyone’s name Laboratory Manual: Report Form pp. 46 -52 DUE Today _____________

DUE Today: One report with everyone’s name Molecular Modeling http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular% 20 Modeling%20 Form%20 Fall%202017. pdf http: //molview. org

Prepare Answers to the following Questions for Next Week

Which of the following bonds is the most polar? A) B) C) D)

In which of the compounds below is the d+ for H the greatest (highest difference in e. n. )? A) B) C) D) CH 4 NH 3 Si. H 4 H 2 O
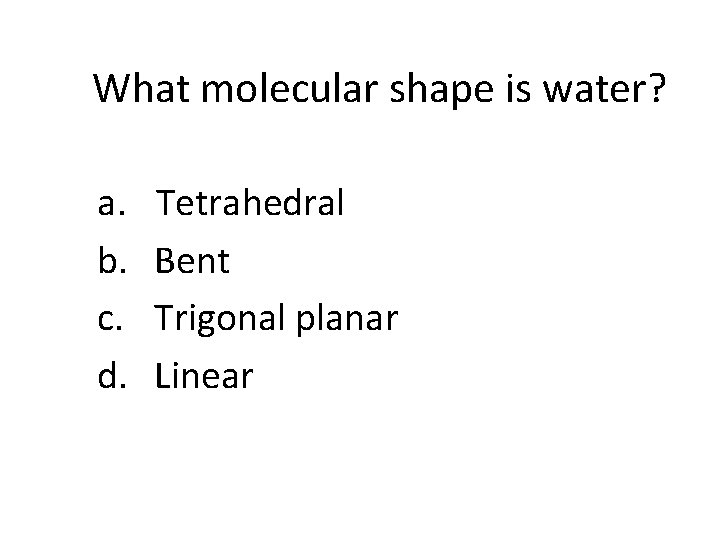
What molecular shape is water? a. b. c. d. Tetrahedral Bent Trigonal planar Linear

What is the electronic geometry of NH 3? a. Linear b. Trigonal Planar c. Tetrahedral d. Trigonal Pyramidal H N H H

Which of these molecules has a linear molecule geometry? a. b. c. d. CO 2 O 3 Both Neither
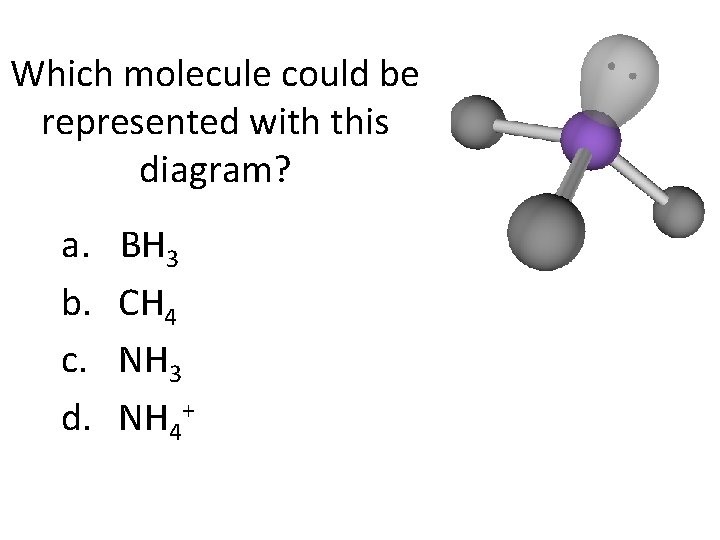
Which molecule could be represented with this diagram? a. b. c. d. BH 3 CH 4 NH 3 NH 4+

What is the molecular geometry of H 2 S? a. Linear b. Tetrahedral c. Trigonal pyramidal d. Bent

Chemical Reactions To DO Today Biological Reactions: Enzyme Catalysts Fermentation / Distillation pp. 63 -47 _____________ http: //www. piney. com/Bab. Ninkasi. html)
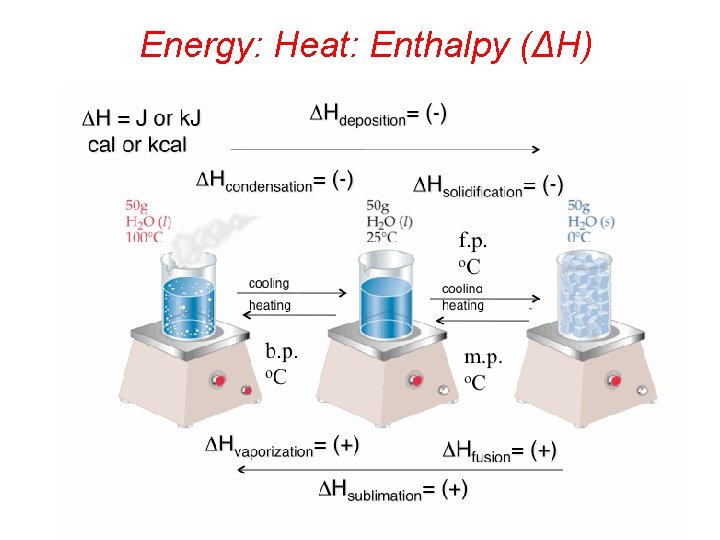
Energy: Heat: Enthalpy (ΔH)


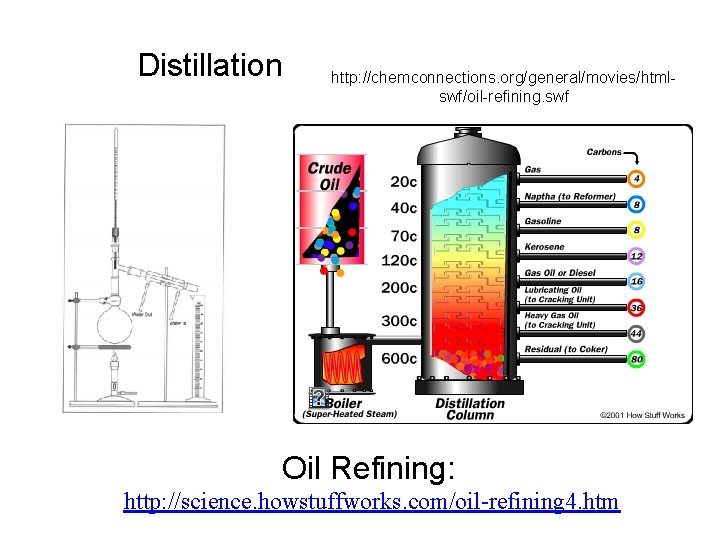
Distillation http: //chemconnections. org/general/movies/htmlswf/oil-refining. swf Oil Refining: http: //science. howstuffworks. com/oil-refining 4. htm

Theoretical & Percent Yield http: //chemconnections. org/general/movies/htmlswf/oil-refining. swf

Theoretical Yield % Ethanol from Density

Calculations
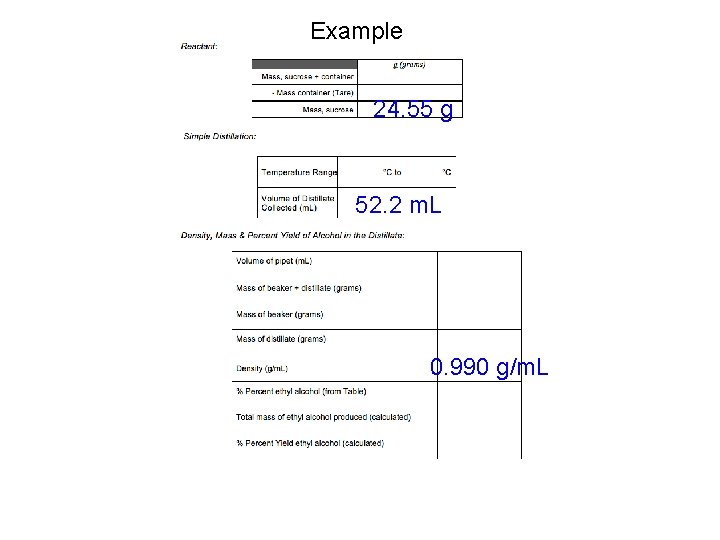
Example 24. 55 g 52. 2 m. L 0. 990 g/m. L
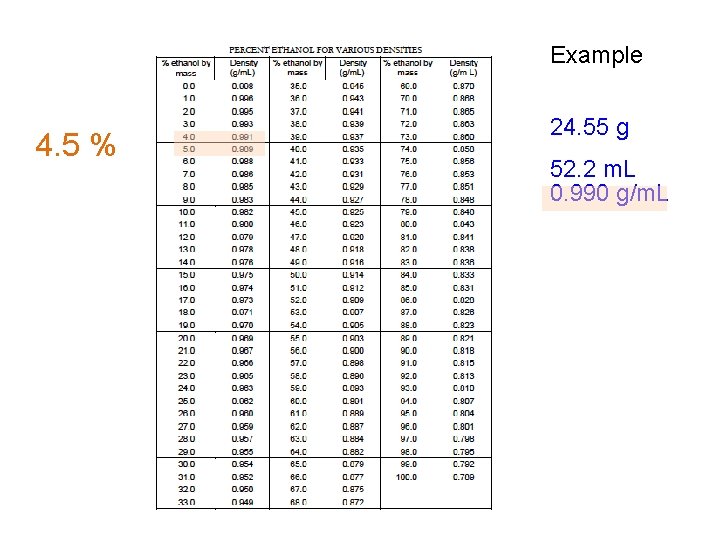
Example 4. 5 % 24. 55 g 52. 2 m. L 0. 990 g/m. L

Theoretical Yield Calculation 24. 55 g ? g (theoretical) 4 Molar mass = 342. 3 g/mol ? mol sucrose = 24. 55 g / 342. 3 g/mol = 0. 07172 mol Molar mass = 46. 07 g/mol ? mol C 2 H 5 OH = 4 x mol sucrose = 0. 2869 mol ? g (theoretical) = mol C 2 H 5 OH x 46. 07 g/mol = 13. 22 g _________ ? g (actual) = [4. 5 % , that is: 4. 5/100] x 52. 2 m. L x 0. 990 g/m. L = 2. 33 g % Yield = g (actual) / g (theoretical) x 100 = 17. 6 %

Theoretical & Percent Yield


Molecular Modeling Computational Chemistry Covalent Bonds: Lewis Structures, Molecular Shapes Dr. Ron Rusay

Molecular Modeling Computational Chemistry Shapes Lewis Structures Polarity, Solubility, Stereochemistry Smell, Teratogenicity Dr. Ron Rusay

https: //www. youtube. com/watch? v=Jq_Ca-HKh 1 g Shapes of Molecules View: What is the shape of a molecule? George Zaidan and Charles Morton http: //chemconnections. org/general/chem 1 08/Molecular%252520 Shapes-Guide. html
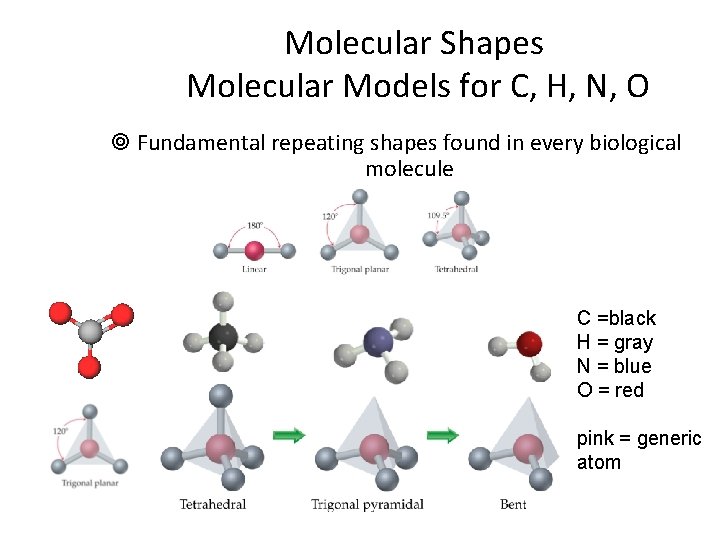
Molecular Shapes Molecular Models for C, H, N, O ¥ Fundamental repeating shapes found in every biological molecule C =black H = gray N = blue O = red pink = generic atom

Professor Gilbert Newton Lewis (circa 1940) G. N. Lewis Photo Bancroft Library, University of California/LBNL Image Library Footnote: G. N. Lewis, despite his insight and contributions to chemistry, was never awarded the Nobel prize. Notes from Lewis’s notebook and his “Lewis” structure.
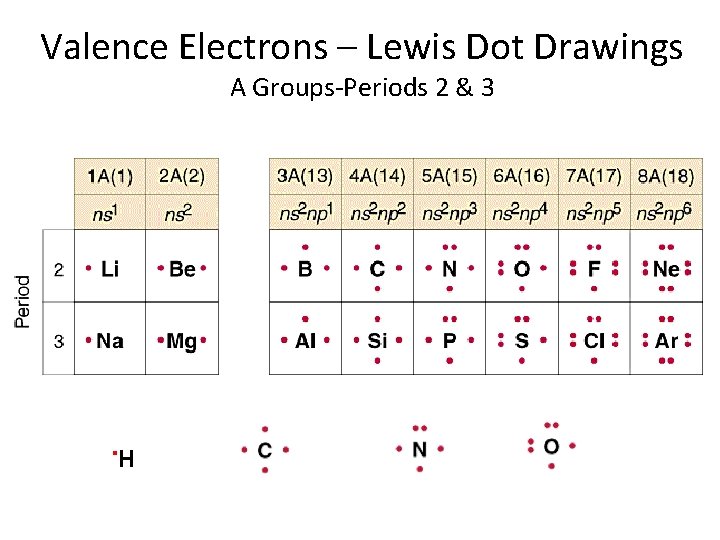
Valence Electrons – Lewis Dot Drawings A Groups-Periods 2 & 3 . H
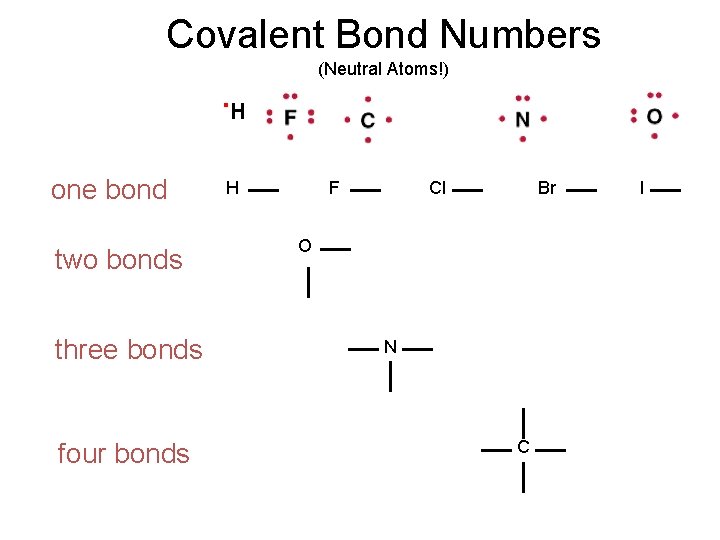
Covalent Bond Numbers (Neutral Atoms!) . H one bond two bonds three bonds four bonds H F Cl Br O N C I
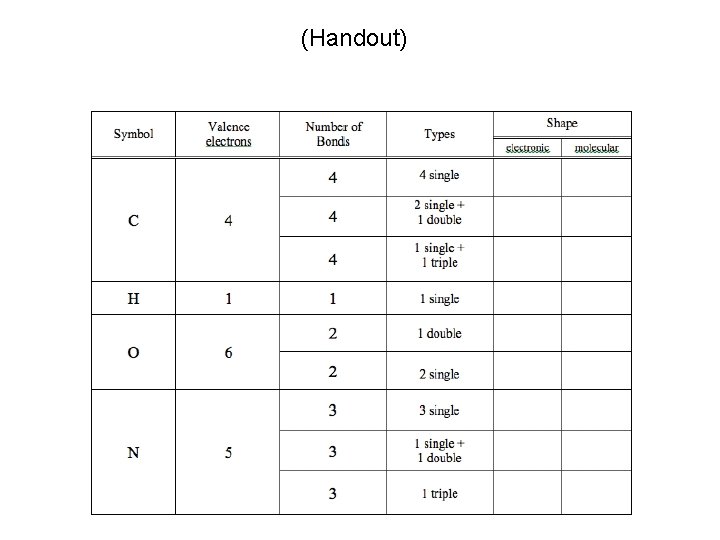
(Handout) Due End of Lab Next Week
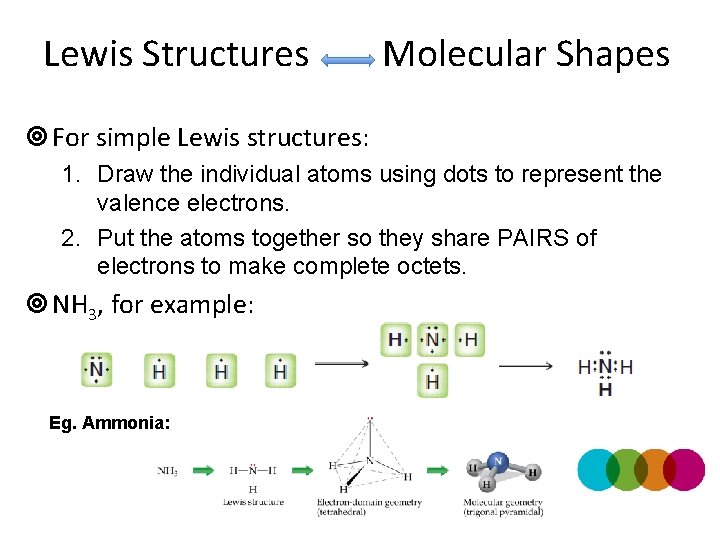
Lewis Structures Molecular Shapes ¥ For simple Lewis structures: 1. Draw the individual atoms using dots to represent the valence electrons. 2. Put the atoms together so they share PAIRS of electrons to make complete octets. ¥ NH 3, for example: Eg. Ammonia:
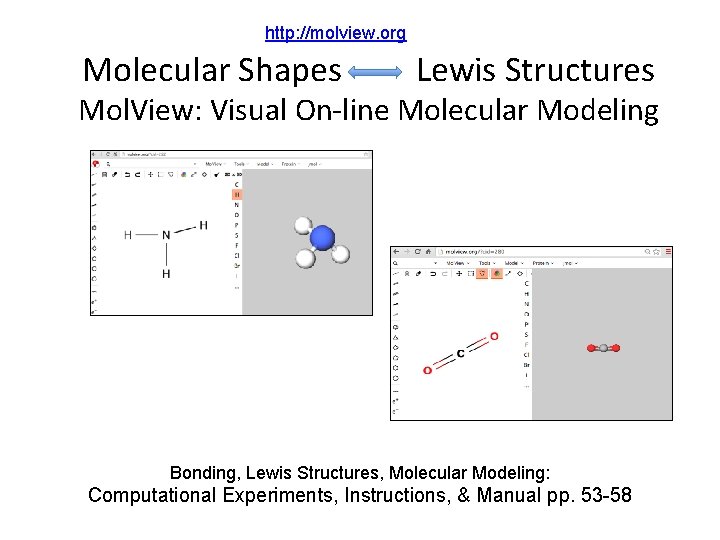
http: //molview. org Molecular Shapes Lewis Structures Mol. View: Visual On-line Molecular Modeling Bonding, Lewis Structures, Molecular Modeling: Computational Experiments, Instructions, & Manual pp. 53 -58
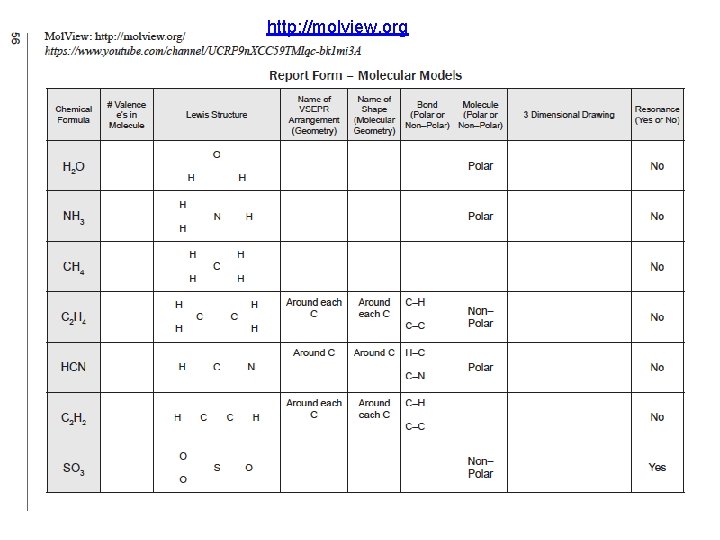
http: //molview. org (Course/ Lab Manual pp. 53 -58) Organize Today with your Moleview Group Due End of Lab Next Week

Orbital (Electronic) Molecular Geometry Important Linear in Trigonal Planar Organic Compounds Trigonal Planar Bent Bond Angle # of lone pairs 0 0 1 Tetrahedral 0 Tetrahedral Trigonal Pyramidal 1 Tetrahedral Bent 2 See again Trigonal Bipyramidal Chem 120 and possibly. Trigonal Bipyramidal in Chem 109 Trigonal Bipyramidal 0 Seesaw 1 T-shape 2 Linear 3 Octahedral 0 Octahedral Square Pyramidal 1 Octahedral Square Planar 2
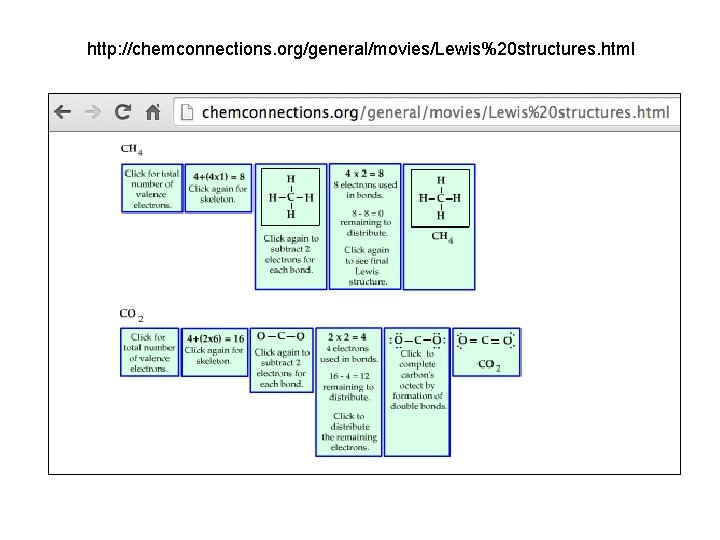
http: //chemconnections. org/general/movies/Lewis%20 structures. html

Covalent Compounds • Share valence electrons. • 1 pair = 1 bond; maximum # of atom-atom bonds = 3. • Octet rule (“duet” for hydrogen) • Lewis structure examples: Notice the charges: In one case they balance, can you name the compound? In the other they do not. It has a “Formal” charge. Can you name the polyatomic ion?
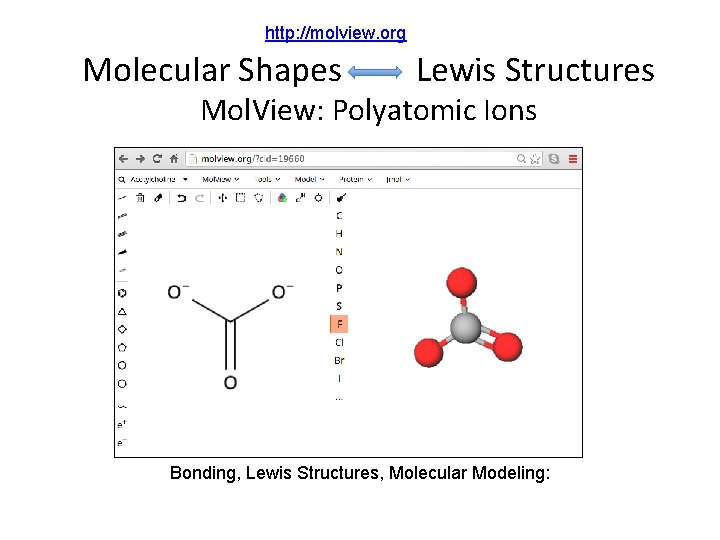
http: //molview. org Molecular Shapes Lewis Structures Mol. View: Polyatomic Ions Bonding, Lewis Structures, Molecular Modeling:

Resonance ¥ Occurs when more than one valid Lewis structure can be written for a particular molecule. [Adjacent free electrons, double or triple bonds. ] ¥These are resonance structures. The actual structure is an average of all of the resonance structures.
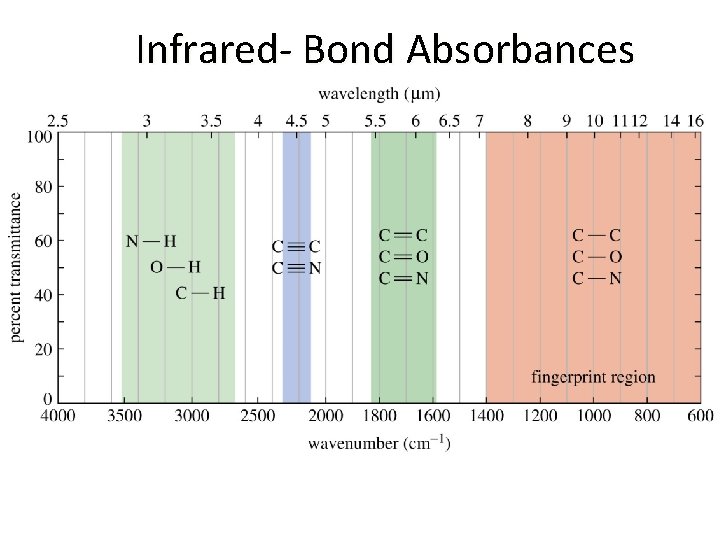
Infrared- Bond Absorbances
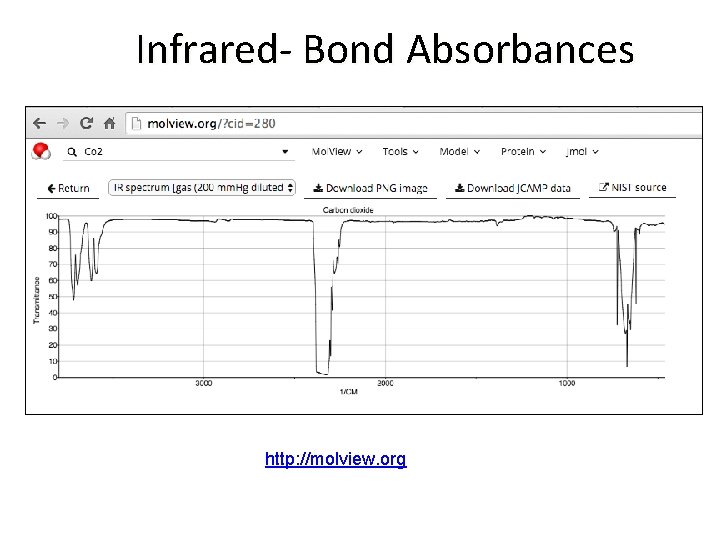
Infrared- Bond Absorbances http: //molview. org

Infrared- Bond Absorbances https: //www. co 2. earth/
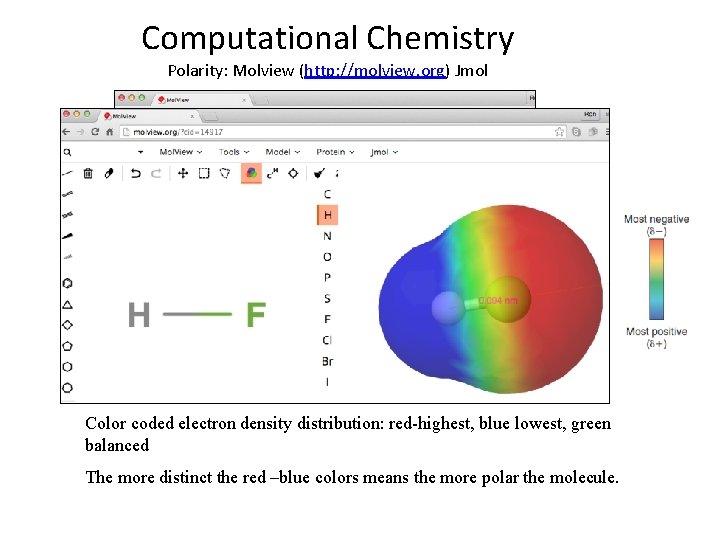
Computational Chemistry Polarity: Molview (http: //molview. org) Jmol Color coded electron density distribution: red-highest, blue lowest, green balanced The more distinct the red –blue colors means the more polar the molecule.
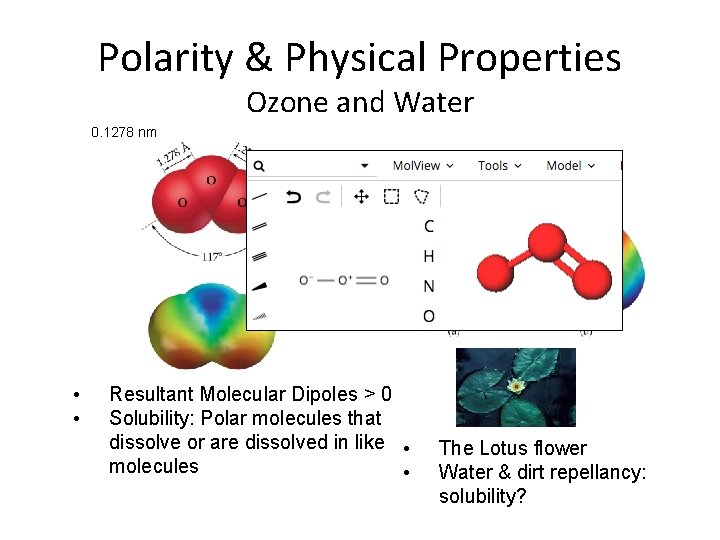
Polarity & Physical Properties Ozone and Water 0. 1278 nm • • Resultant Molecular Dipoles > 0 Solubility: Polar molecules that dissolve or are dissolved in like • molecules • The Lotus flower Water & dirt repellancy: solubility?
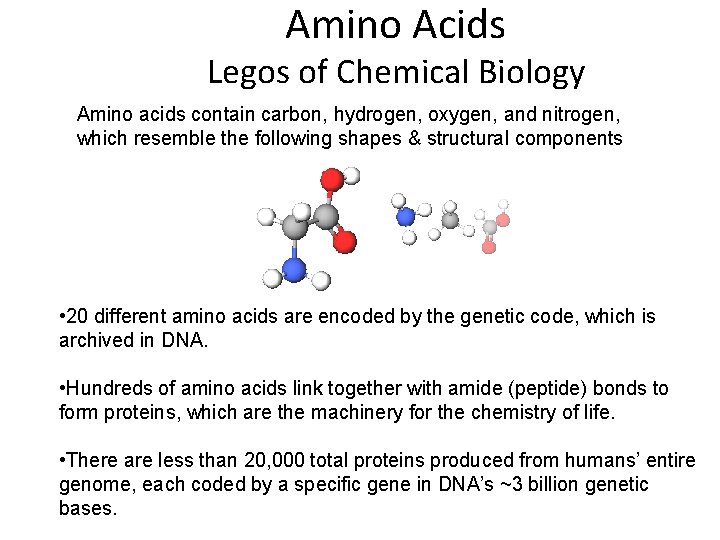
Amino Acids Legos of Chemical Biology Amino acids contain carbon, hydrogen, oxygen, and nitrogen, which resemble the following shapes & structural components • 20 different amino acids are encoded by the genetic code, which is archived in DNA. • Hundreds of amino acids link together with amide (peptide) bonds to form proteins, which are the machinery for the chemistry of life. • There are less than 20, 000 total proteins produced from humans’ entire genome, each coded by a specific gene in DNA’s ~3 billion genetic bases.

Amino Acids Proteins Indispensible Biopolymers Acetylcholinesterase (ACE) ACE, an enzyme, which catalyzes a key reaction in a repetitive biochemical cycle that is crucial to neurological and physiological functions in humans…. and insects among others. 4, 496 atoms; 4, 404 bonds 574 amino acid residues

Biological Reactions eg. Fermentation https: //www. youtube. com/watch? v=v. W 99 JEa. DAp. Y http: //www. piney. com/Bab. Ninkasi. html

https: //www. youtube. com/watch? v=OKRJ 73 Ro 5 jw https: //www. youtube. com/watch? v=r. L 14 u. Lb. SLLc

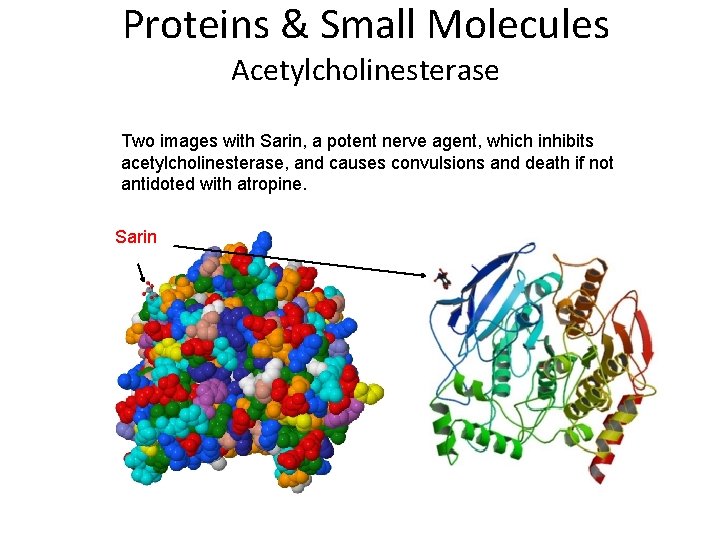
Proteins & Small Molecules Acetylcholinesterase Two images with Sarin, a potent nerve agent, which inhibits acetylcholinesterase, and causes convulsions and death if not antidoted with atropine. Sarin
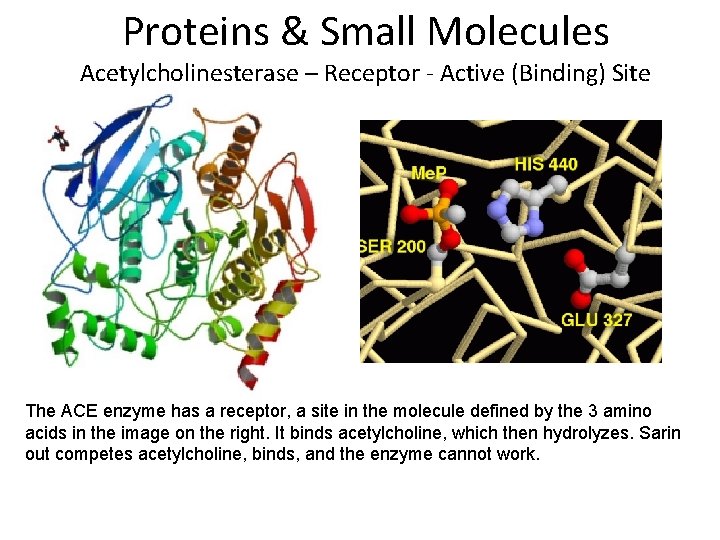
Proteins & Small Molecules Acetylcholinesterase – Receptor - Active (Binding) Site The ACE enzyme has a receptor, a site in the molecule defined by the 3 amino acids in the image on the right. It binds acetylcholine, which then hydrolyzes. Sarin out competes acetylcholine, binds, and the enzyme cannot work.

Proteins & Small Molecules Acetylcholinesterase: Docking The 3 amino acids that bind acetylcholine prefers to bind Sarin, which does not unbind and the enzyme does not work.

Proteins & Small Molecules Acetylcholinesterase-Active Site-Docking The normal function of acetylcholinesterase is with acetylcholine. This general process is similar to the way we smell and relates to many, many physiological and pharmacological processes. http: //chemconnections. org/general/movies/richard. mpg

Molecular Modeling Computational Chemistry Polarity, Stereochemistry Smell Biomimcry Birth Defects Dr. Ron Rusay
Detecting stuff we cannot see: the Sense of Smell https: //www. youtube. com/watch? v=sn. Jn. O 6 Opj. Cs
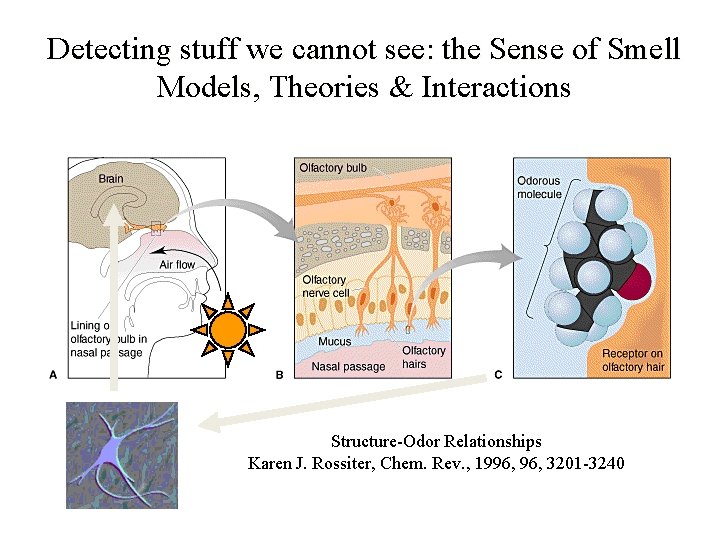
Detecting stuff we cannot see: the Sense of Smell Models, Theories & Interactions Structure-Odor Relationships Karen J. Rossiter, Chem. Rev. , 1996, 3201 -3240

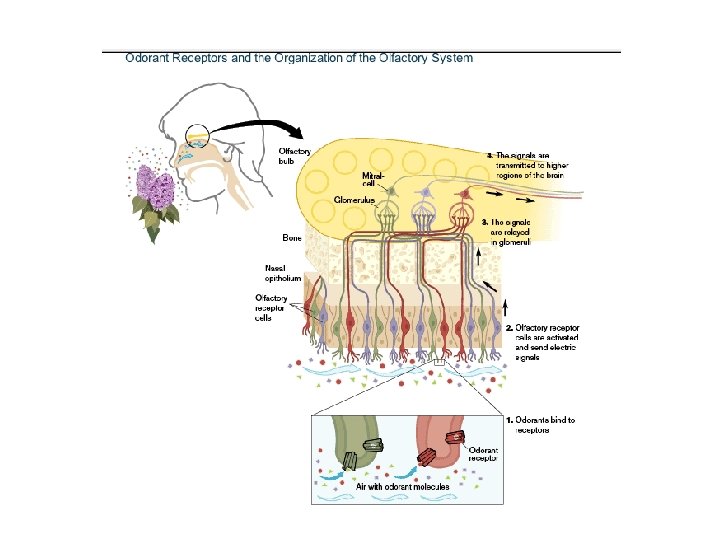
Historical view of a few smell receptors. 4 October 2004 The Nobel Assembly at Karolinska Institutet has today decided to award The Nobel Prize in Physiology or Medicine for 2004 jointly to Richard Axel and Linda B. Buck for their discoveries of "odorant receptors and the organization of the olfactory system"
What a smell looks like https: //www. youtube. com/watch? v=58 U 52 l. DTuvk&list=PLgawtc. OBBjr 9 I-NDo. UXHm. TQr_VN 465 G 2&index=3

Inside the extraordinary nose of a search-and-rescue dog https: //www. youtube. com/watch? v=FLH 36 ML 8 IEU
Dogs Can Smell Cancer - Secret Life of Dogs - BBC https: //www. youtube. com/watch? v=e 0 UK 6 kk. S 0_M

The “Lotus Effect” Biomimicry Wax ¥ Water not only dissolves some dirt, but attracts and removes it like a snowball rolling downhill. ¥ Lotus petals have micrometer-scale roughness, resulting in water contact angles up to 170°

The “Lotus Effect” Biomimicry ¥ Isotactic polypropylene (i-PP) melted between two glass slides and subsequent crystallization provided a smooth surface. Atomic force microscopy tests indicated that the surface had root mean square (rms) roughness of 10 nm. ¥ A) The water drop on the resulting surface had a contact angle of 104° ± 2 ¥ B) the water drop on a superhydrophobic i-PP coating surface has a contact angle of 160°. Science, 299, (2003), pp. 1377 -1380, H. Yldrm Erbil, A. Levent Demirel, Yonca Avc, Olcay Mert http: //www. sciencemag. org/cgi/content/full/299/5611/1377/DC 1

Polarity & Physical Properties Chloroform (polar) vs. Carbon tetrachloride (non-) An electrically charged rod attracts a stream of chloroform but has no effect on a stream of carbon tetrachloride.

Polarity & Physical Properties Solubility Generally likes dissolves like: ¥ Polar compounds dissolve other polar compounds & ionic compounds. Eg. ethanol and water, sodium chloride and water, sugar and water ¥ Nonpolar compounds are soluble in other nonpolar compounds. Eg. carbon tetrachloride and oil, diesel and gasoline
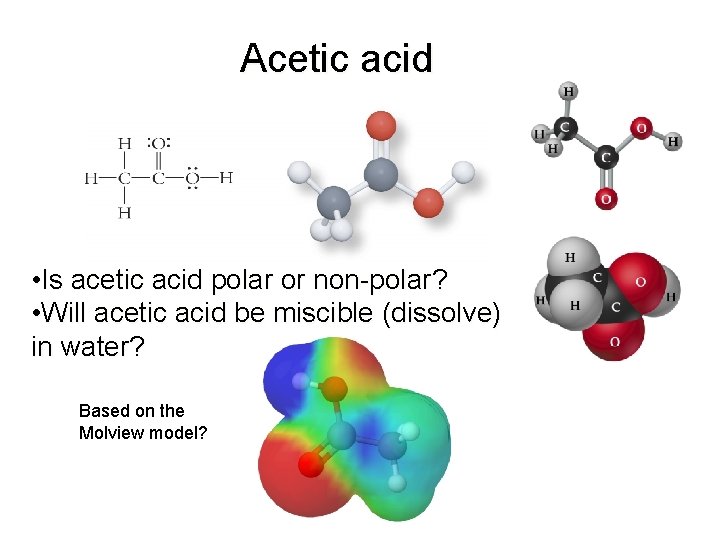
Acetic acid • Is acetic acid polar or non-polar? • Will acetic acid be miscible (dissolve) in water? Based on the Molview model?
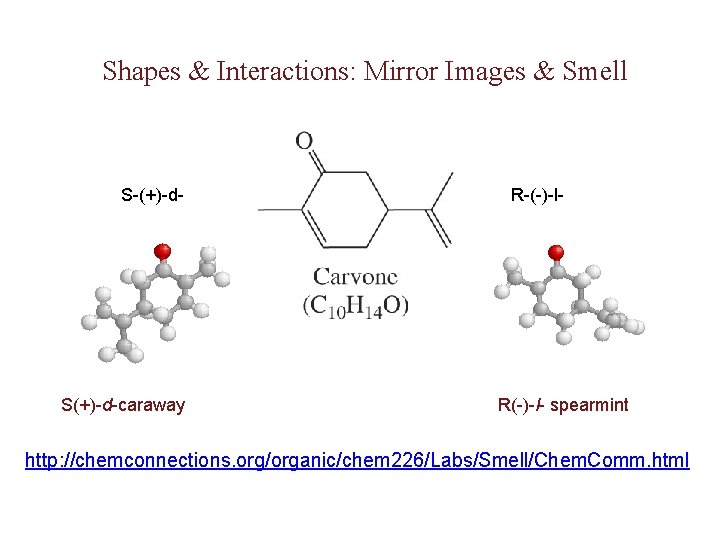
Shapes & Interactions: Mirror Images & Smell S-(+)-d- S(+)-d-caraway R-(-)-l- R(-)- l- spearmint http: //chemconnections. org/organic/chem 226/Labs/Smell/Chem. Comm. html
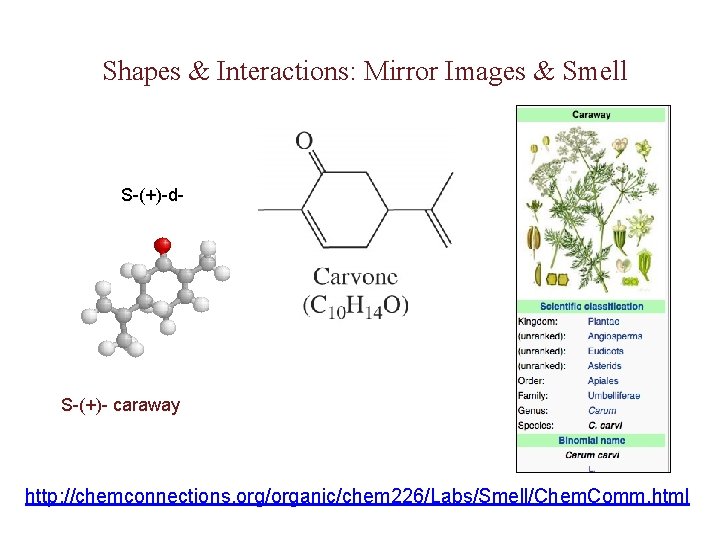
Shapes & Interactions: Mirror Images & Smell S-(+)-d- S-(+)- caraway http: //chemconnections. org/organic/chem 226/Labs/Smell/Chem. Comm. html
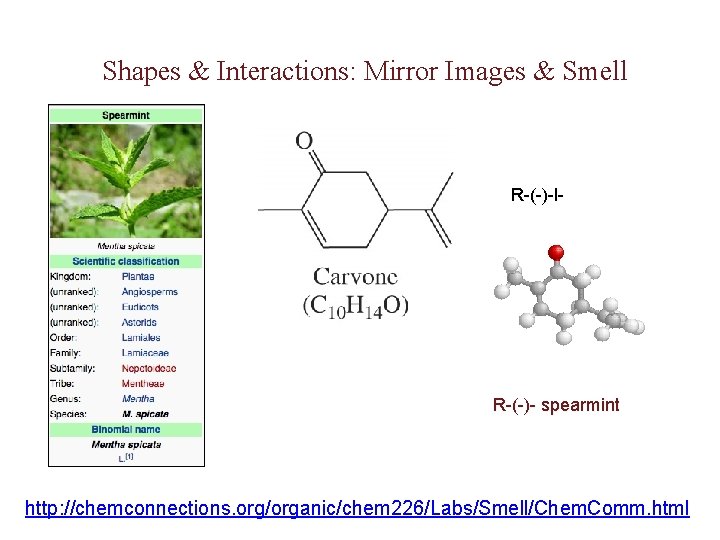
Shapes & Interactions: Mirror Images & Smell R-(-)-l- R-(-)- spearmint http: //chemconnections. org/organic/chem 226/Labs/Smell/Chem. Comm. html

Isomerism • Molecules which have the same molecular formula, but differ in the arrangement of their atoms, are called isomers. • Constitutional (or structural) isomers differ in their bonding sequence. • Stereoisomers differ only in the arrangement of the atoms in space.


Stereoisomerism Enantiomers are chiral: i. e. They are nonsuperimposable mirror images. ¥ Enantiomers are “optical isomers. ” eg. (+) and (-) carvone ¥ Most physical and chemical properties of enantiomers are identical. ¥ Therefore, enantiomers are very difficult to separate eg. Tartaric acid… ask Louis Pasteur. ¥ Enantiomers can have very different physiological effects: eg. (+) and (-) carvone ¥
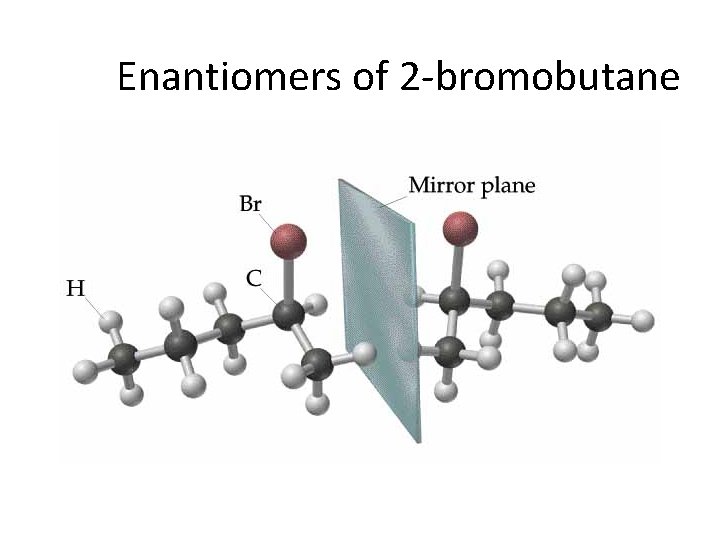
Enantiomers of 2 -bromobutane
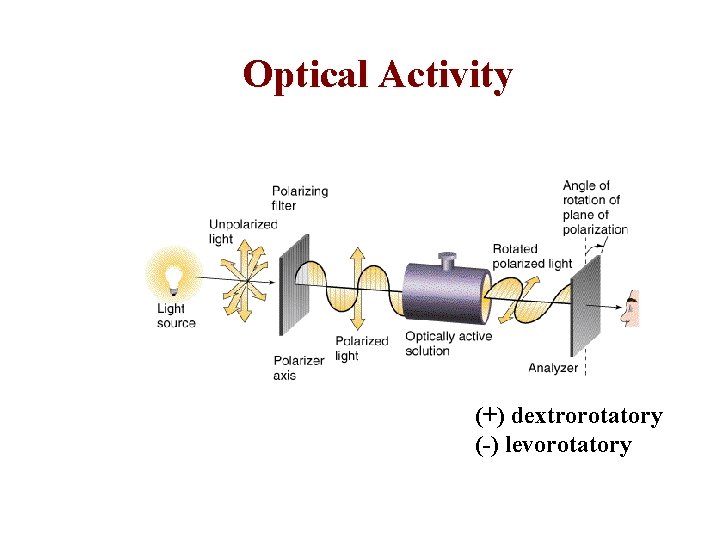
Optical Activity (+) dextrorotatory (-) levorotatory
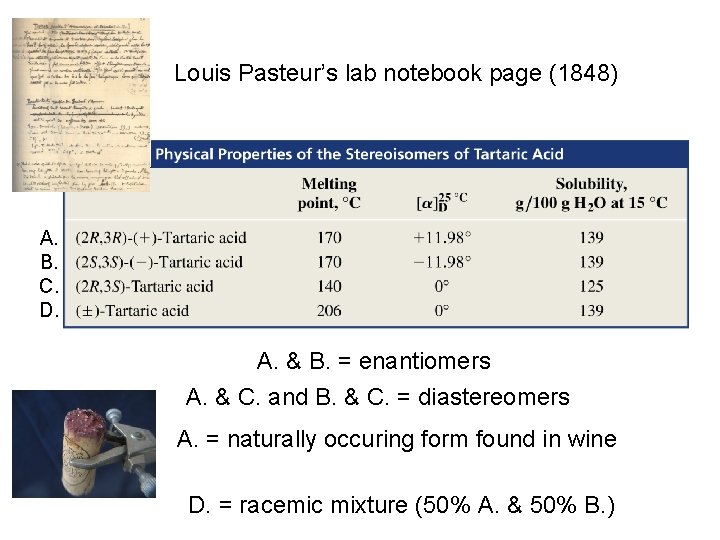
Louis Pasteur’s lab notebook page (1848) A. B. C. D. A. & B. = enantiomers A. & C. and B. & C. = diastereomers A. = naturally occuring form found in wine D. = racemic mixture (50% A. & 50% B. )

d, l-Carvone: Mint or Caraway http: //chemconnections. org/organic/chem 226/jmol-html/d-carvone. html http: //chemconnections. llnl. gov/Organic/Chem 226/jmol-html/l-carvone. html The mirror image of an enantiomer will rotate the plane of polarized light by the same amount in the opposite direction. Eg (+) d-carvone +62 o (caraway) and (-) l-carvone -62 o (spearmint)…. What about a 50: 50 (racemic) mixture?

Stereochemistry • Stereochemistry: v. The study of the three-dimensional structures of molecules, particularly stereoisomers • Structural isomers: v. Have the same molecular formula, same types of bonds, but different bonding sequences, “connectivity” • Stereoisomers: v. Have the same molecular formula, same bonding sequences, but different spatial arrangements & relative orientations
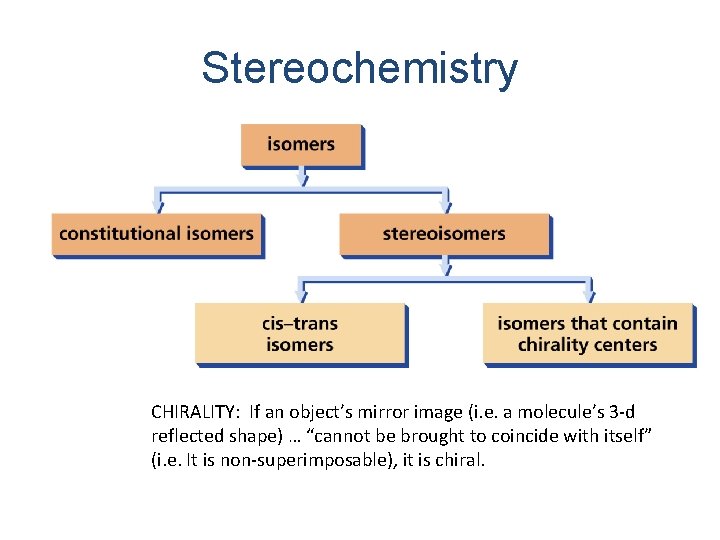
Stereochemistry CHIRALITY: If an object’s mirror image (i. e. a molecule’s 3 -d reflected shape) … “cannot be brought to coincide with itself” (i. e. It is non-superimposable), it is chiral.
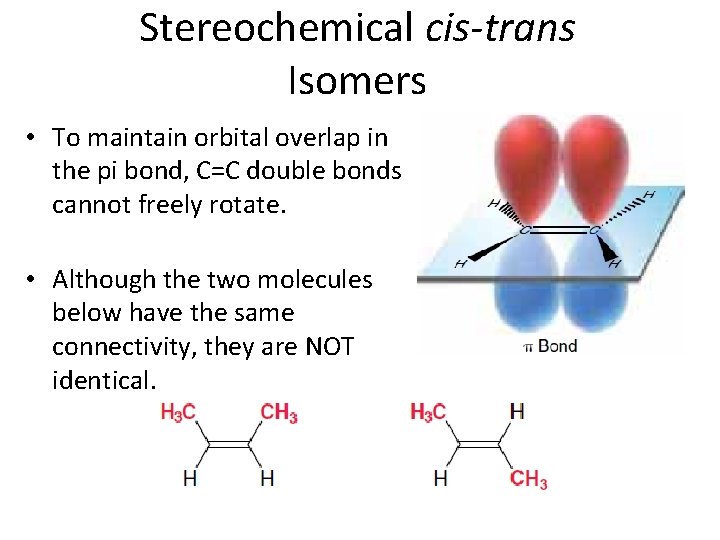
Stereochemical cis-trans Isomers • To maintain orbital overlap in the pi bond, C=C double bonds cannot freely rotate. • Although the two molecules below have the same connectivity, they are NOT identical.

Triglycerides & Unsaturated Fatty Acids Saturated & Unsaturated: cis- vs. trans-

Chirality & Carbon Atoms Each carbon atom with four different substituents are chiral. http: //chemconnections. org/general/movies/Chirality. mov
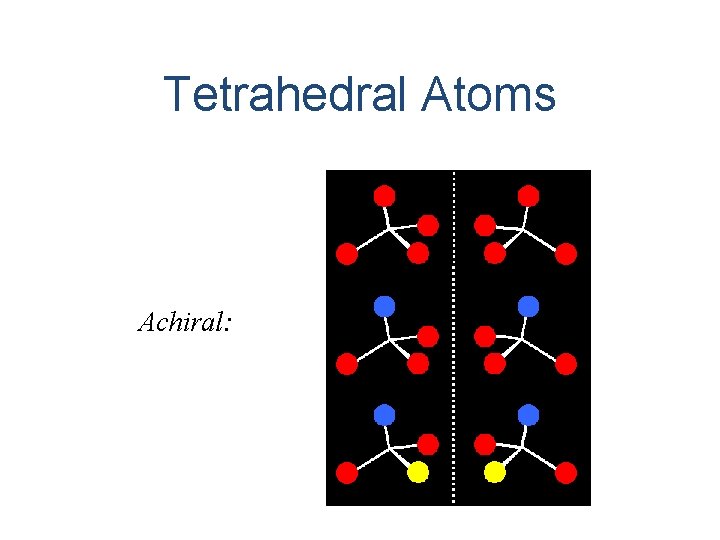
Tetrahedral Atoms Achiral:
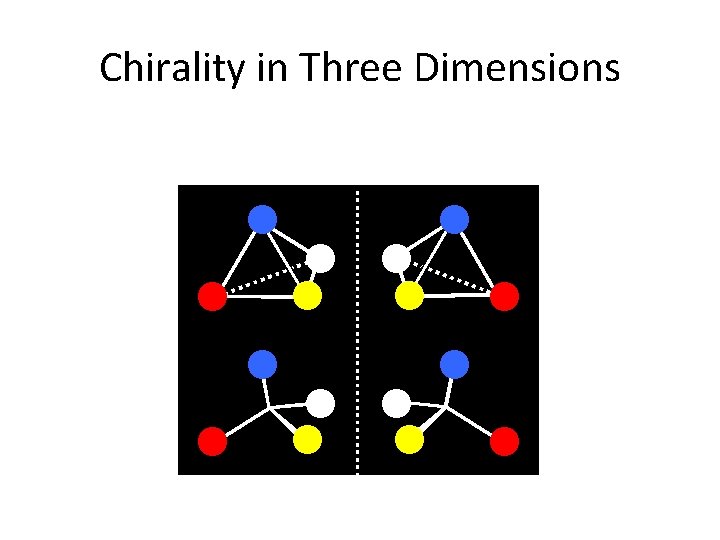
Chirality in Three Dimensions
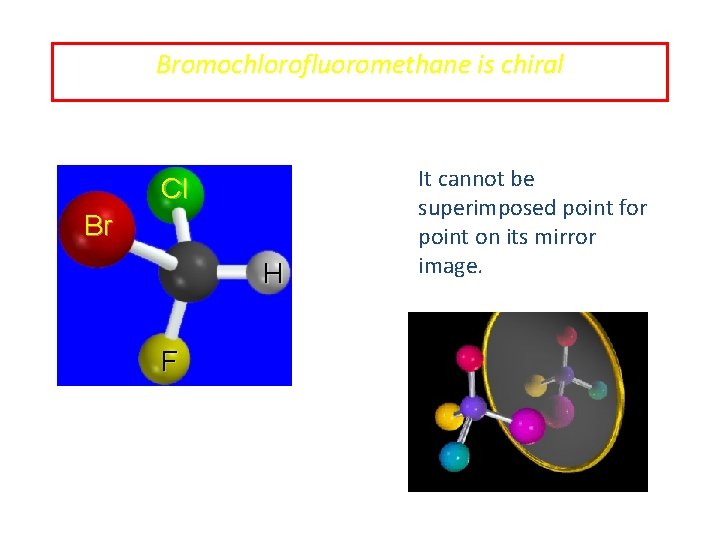
Bromochlorofluoromethane is chiral Cl Br H F It cannot be superimposed point for point on its mirror image.
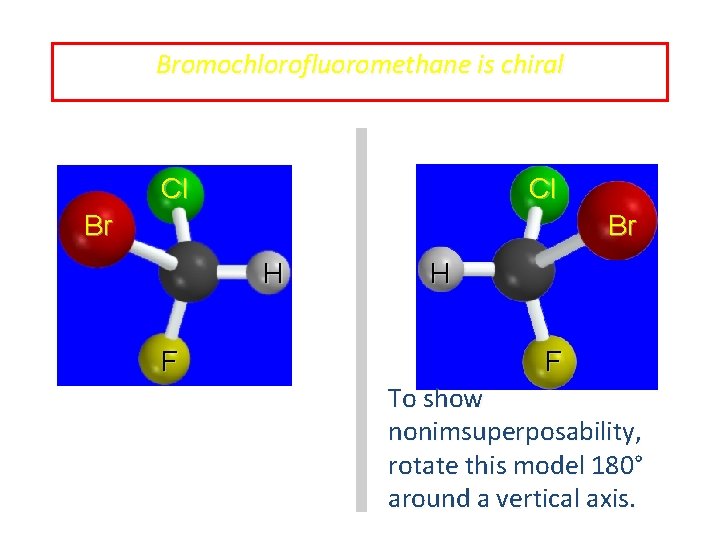
Bromochlorofluoromethane is chiral Cl Cl Br Br H F To show nonimsuperposability, rotate this model 180° around a vertical axis.

Chirality
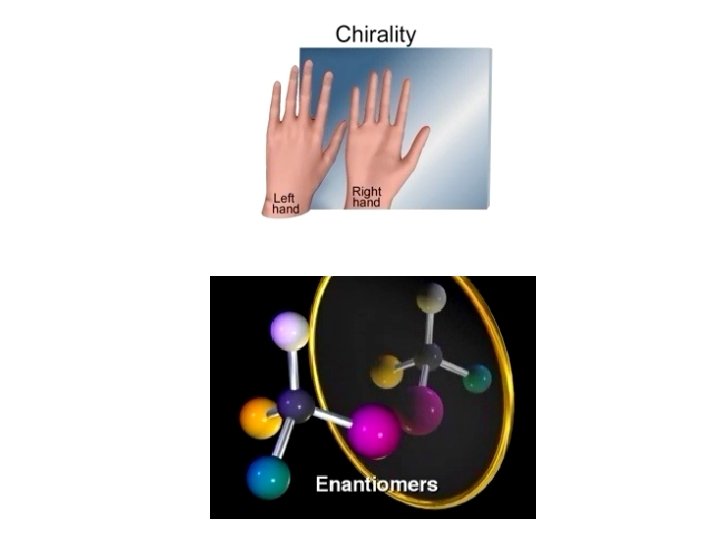

Stereoisomerism Enantiomers are chiral: i. e. They are non-superimposable mirror images. ¥ Most physical and chemical properties of enantiomers are identical. ¥ Therefore, enantiomers are very difficult to separate eg. Tartaric acid… Louis Pasteur: ¥ • Enantiomers can have very different physiological effects: eg. (+) and (-) carvone, Advil (ibuprofen) …… (thalidomide)
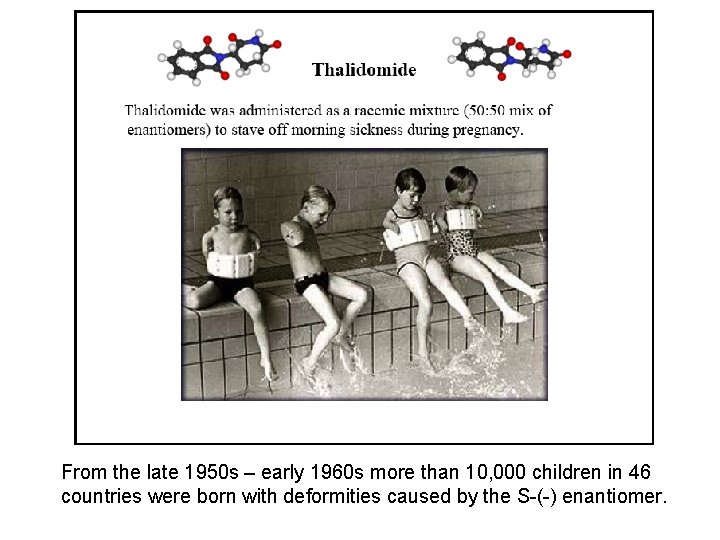
From the late 1950 s – early 1960 s more than 10, 000 children in 46 countries were born with deformities caused by the S-(-) enantiomer.
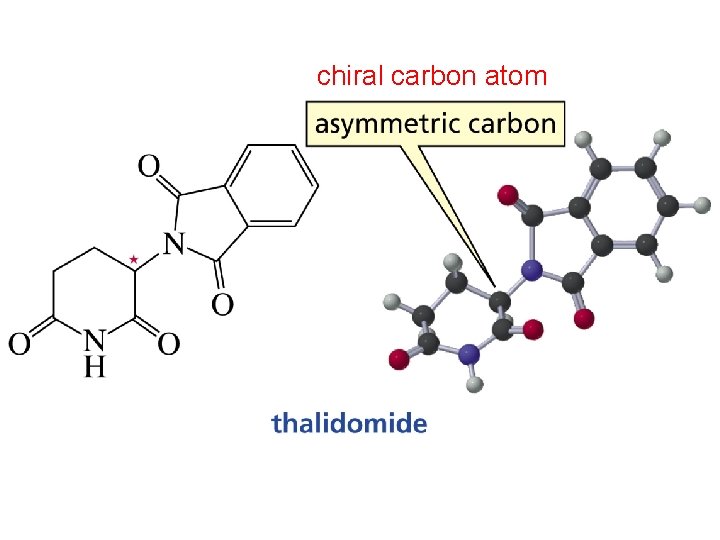
chiral carbon atom
- Slides: 91