Chem 108 Lab Week 8 Sign in Pick
Chem 108: Lab Week 8 Sign in Pick up papers Sit with group partners from last week’s lab G D B H A E C

Exp. #5: What’s My Formula? Your A-H group has obtained 2 to 4 unknowns. Complete the experimental procedure and submit one complete group report form today with everyone’s name on the front page and including data form page & calculations for each unknown & % Yield (replacements for pg. 36) Complete Report Form DUE Today Mass Evaporating Dish + Product Mass Product (Experimental) Molar Mass Product / Molar Mass Reactant x Gatekeepers x 100 = Theoretical % Mass Product (Exp) / Mass Reactant x 100 = Calculations: % Yield = actual grams of product / theoretical (calculated) grams of produc x 100

Calculations: % Yield = actual grams of product / theoretical (calculated) grams of produc x 100

Exp. #5: What’s My Formula? Identificaton Unknown Sample Salt Unknown Sample Salt Experimental Calculation: Comparison to Calculation(s) for a, b, c, d FROM last week: ? ?

Theoretical Mass Calculations for any Reaction Reactants Products grams (Reactant) grams (Product) Moles Molar Mass grams (R) 1 mol (R) grams (R) (Divide) by Molar Mass (R) ? mol (P) ? mol (R) grams (P) ? grams (P) 1 mol (P) (Multiply) "Gatekeepers” from Balanced reaction by Molar Mass (P)

Exp. #5: What’s My Formula? % Yield Example Heating 10. 00 g of an unknown determined to be sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. What is the Percent Yield? First calculate theoretical yield. (Adaptation of your calculations last week. ) Consider the calculation assumes everything was perfect & based on 100% accuracy. Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = ? g (Theoretical) Molar Mass = 105. 99 g/mol
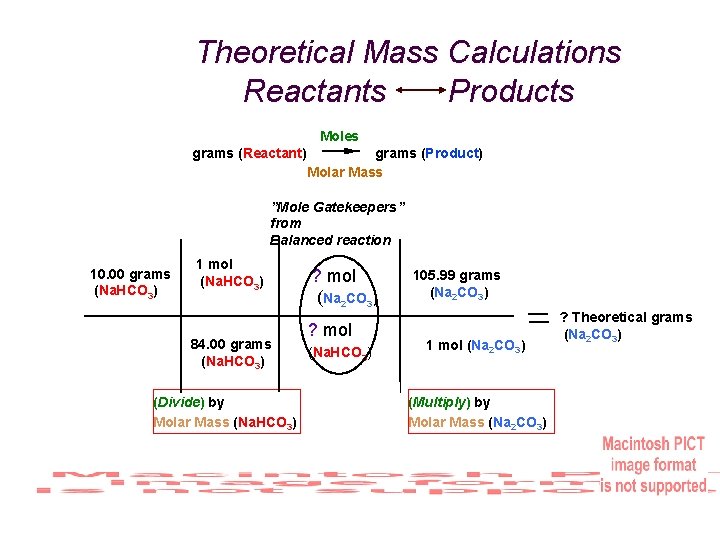
Theoretical Mass Calculations Reactants Products Moles grams (Reactant) grams (Product) Molar Mass ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) ? mol (Na 2 CO 3) ? mol (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) ? Theoretical grams (Na 2 CO 3)
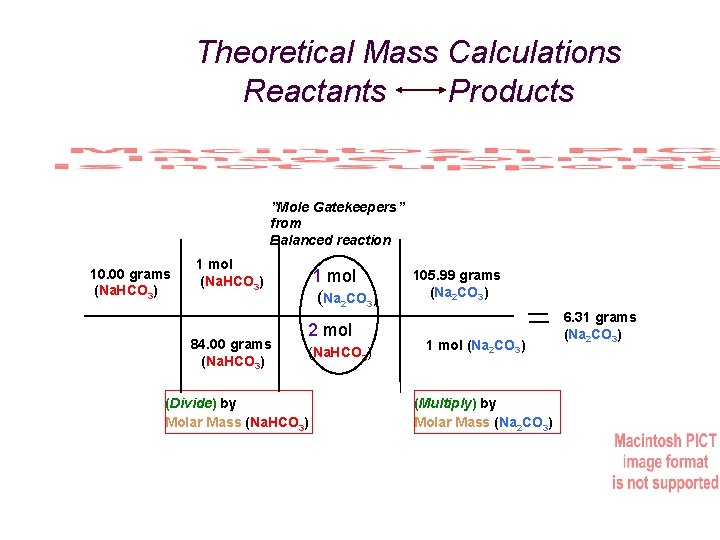
Theoretical Mass Calculations Reactants Products ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) 1 mol (Na 2 CO 3) 2 mol (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) 6. 31 grams (Na 2 CO 3)

Exp. #5: What’s My Formula? “% Yield” is used to measure the efficiency (similar to “accuracy”) of any reaction in yielding “product(s)” (on the right of an equation) versus the calculated (theoretical) amount of the product based on the amount of “reactant(s)” (from the left of the equation) using the relative number of moles of each in a balanced chemical equation. % Yield = actual grams of product / theoretical (calculated) grams of product x 100 For example, heating 10. 00 g of sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. After calculating theoretical yield: Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = 6. 31 g (Theoretical) Molar Mass = 105. 99 g/mol % Yield = 5. 98 g (actual) / 6. 31 g (theoretical) x 100 = 94. 6%

QUESTION A synthetic reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, the calculated theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6%

ANSWER If a reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, and theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6% % yield = 2. 45 g / 3. 05 g x 100 = 80. 3%

What’s My Formula? Post Lab: Compounds with the Same Formula [ eg. C 9 H 8 O 4 ] Aspirin 4 -Hydroxyphenylpyruvic acid Dihydroxycinnamic acids: Caffeic acid (3, 4 -dihydroxycinnamic acid) Umbellic acid (2, 4 -dihydroxycinnamic acid) 2, 3 -Dihydroxycinnamic acid 2, 5 -Dihydroxycinnamic acid 3, 5 -Dihydroxycinnamic acid

Post Lab: Compounds with the Same Formula [ eg. C 9 H 8 O 4 ] Molar Comparisons of Analgesics Calculate Moles : Doses (mmol/dose) Which analgesic has the most biologically active ingredient based on millimoles per dose (mmol/dose)? 5. 0 g of each would produce the following number of doses: Formula Doses Aspirin C 9 H 8 O 4 15. Ibuprofen C 13 H 18 O 2 25 Naproxen Sodium C 14 H 13 O 3 Na 22. 7 Acetaminophen C 8 H 9 NO 2 5 mmol/dose 28 mmol ? ? ? Molar Mass Aspirin = 180. 1 g/mol 5. 0 g / 180. 1 g/mol = 0. 028 mol = 28 mmol

Post Lab: Molar Comparisons of Analgesics Submit Individually Calculate Moles : Doses (mmol/dose)

i-clicker Quiz (Moles / Molar Masses / Nomenclature) Moles / Molar Mass and Molecular Formulas i-clicker QUIZ QUESTIONS http: //chemconnections. org/general/chem 108/Moles-Molar Mass Quiz. html Submit individually on-line Consult Calendar for DUE DATE

Chemical Reactions Combination (Synthesis) Decomposition Single Displacement Double Displacement _____________ Combustion: Oxidation-Reduction Biological Reactions: Enzyme Catalysts Fermentation Set-up Today http: //www. piney. com/Bab. Ninkasi. html)

Biological Reactions Fermentation https: //www. youtube. com/watch? v=v. W 99 JEa. DAp. Y http: //www. piney. com/Bab. Ninkasi. html


Chemical Reactions Biological Reactions: Enzyme Catalysts Fermentation Handout + Lab Manual pp. 61 -67 Set-up Today Have Dr. R. initial pg. 66 before leaving lab

Molecular Modeling (Individual or Collaborative) Report Form (Replacement pages for Molecular Model Lab pp. 97 -103) http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular%20 Modeling%20 Fo rm%20 Fall%202017. pdf Computers & Internet available in PS 110, if needed http: //molview. org Turn-in individually or one per group Consult Calendar for Due Date

- Slides: 21