Chem 108 Lab Week 8 Sign in Pick

Chem 108: Lab Week 8 Sign in; Pick up papers, Sit with Group from last week. DUE Today Nomenclature What’s My Formula? Electronegativity Worksheet (pre-lab)
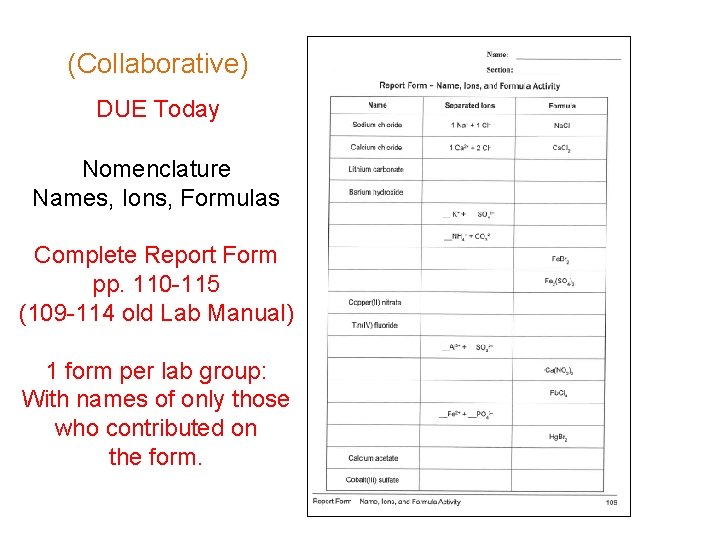
(Collaborative) DUE Today Nomenclature Names, Ions, Formulas Complete Report Form pp. 110 -115 (109 -114 old Lab Manual) 1 form per lab group: With names of only those who contributed on the form.

What’s My Formula? Individual Completed: pg. 36, plus pp 37 & 38 (Unknown ID and clear calculations)

Theoretical Mass Calculations for any Reaction Reactants Products grams (Reactant) grams (Product) Moles Molar Mass grams (R) 1 mol (R) grams (R) (Divide) by Molar Mass (R) ? mol (P) ? mol (R) grams (P) ? grams (P) 1 mol (P) (Multiply) "Gatekeepers” from Balanced reaction by Molar Mass (P)

What’s My Formula? % Yield (Example) Heating 10. 00 g of an unknown determined to be sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. What is the Percent Yield? First calculate theoretical yield. (Adaptation of your calculations last week. ) It considers in the calculation that everything went perfectly, and is based on the assumption of 100% accuracy. % Yield is actual; based on reality. Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = ? g (Theoretical) Molar Mass = 105. 99 g/mol
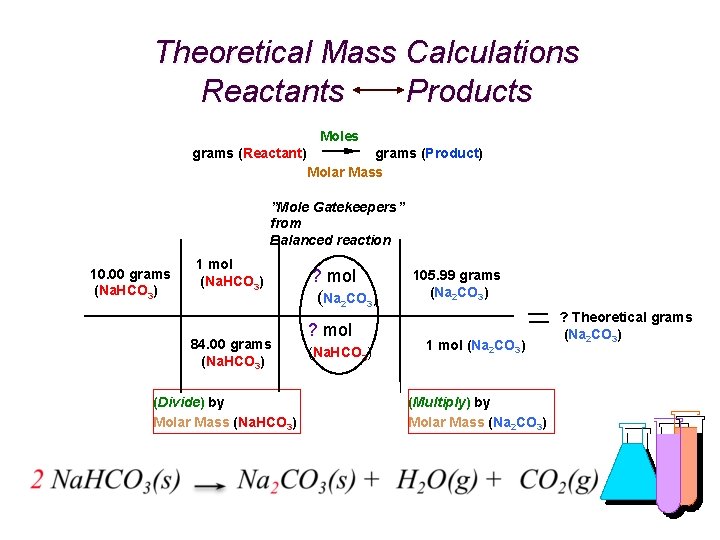
Theoretical Mass Calculations Reactants Products Moles grams (Reactant) grams (Product) Molar Mass ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) ? mol (Na 2 CO 3) ? mol (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) ? Theoretical grams (Na 2 CO 3)
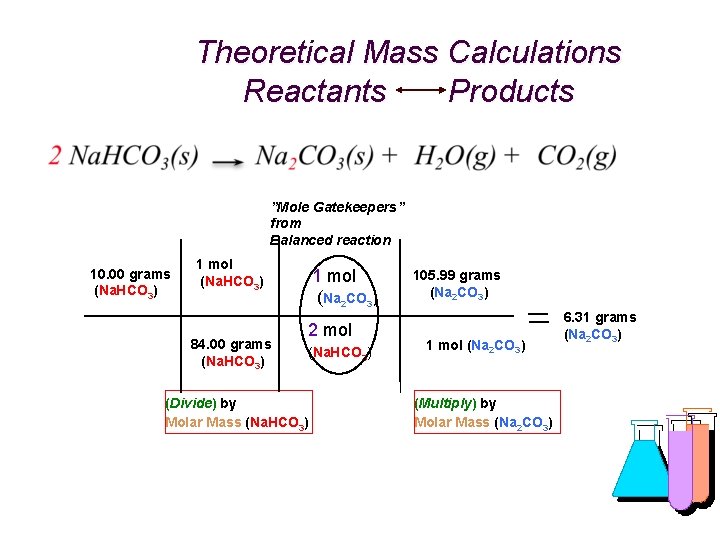
Theoretical Mass Calculations Reactants Products ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) 1 mol (Na 2 CO 3) 2 mol (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) 6. 31 grams (Na 2 CO 3)

What’s My Formula? “% Yield” is used to measure the efficiency (similar to “accuracy”) of any reaction in yielding “product(s)” (on the right of an equation) versus the calculated (theoretical) amount of the product based on the amount of “reactant(s)” (from the left of the equation) using the relative number of moles of each in a balanced chemical equation. % Yield = actual grams of product / theoretical (calculated) grams of product x 100 For example, heating 10. 00 g of sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. After calculating theoretical yield: Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = 6. 31 g (Theoretical) Molar Mass = 105. 99 g/mol % Yield = 5. 98 g (actual) / 6. 31 g (theoretical) x 100 = 94. 6%
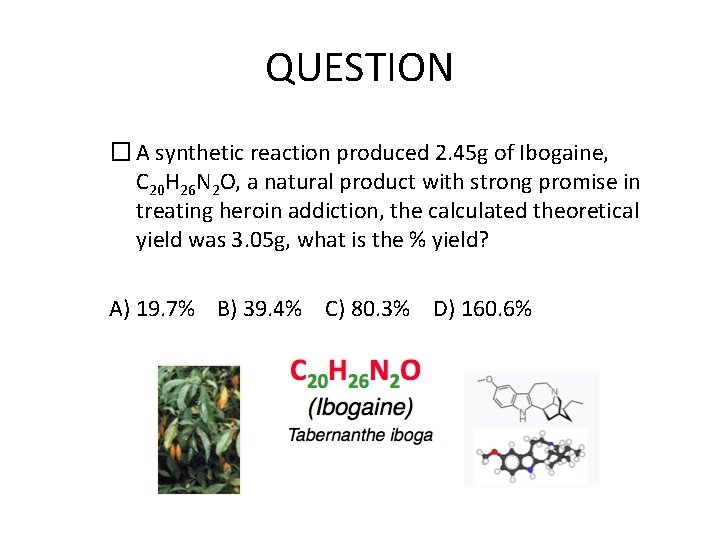
QUESTION � A synthetic reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, the calculated theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6%

ANSWER � If a reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, and theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6% % yield = 2. 45 g / 3. 05 g x 100 = 80. 3%

Molar Comparisons of Analgesics Calculate Moles : Doses (mmol/dose) Post Lab: Must submit Individually from calendar link DUE Friday, 20 -Mar
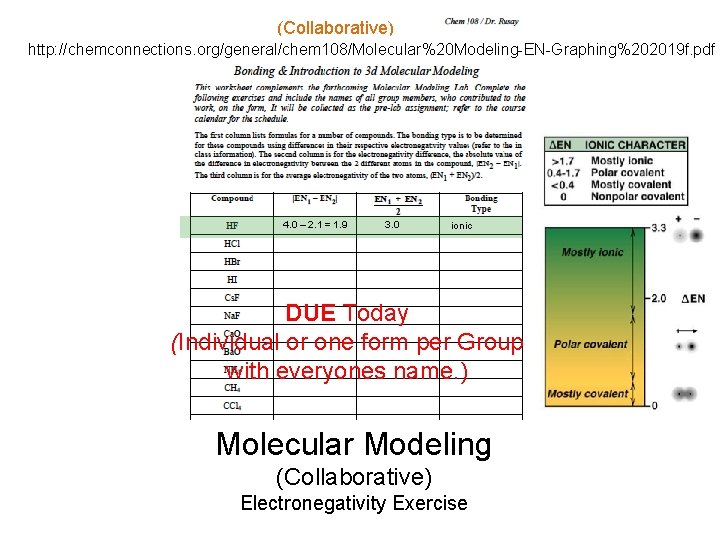
(Collaborative) Bonding. H? http: //chemconnections. org/general/chem 108/Molecular%20 Modeling-EN-Graphing%202019 f. pdf and F? 4. 0 – 2. 1 = 1. 9 3. 0 ionic DUE Today (Individual or one form per Group with everyones name. ) Molecular Modeling (Collaborative) Electronegativity Exercise

Molecular Modeling (Collaborative Effort / Individual Report Forms) Report Form (Replaces Molecular Model Lab pp. 102 -104) http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular%20 Modeling%2 0 Form%20 Fall%202019. pdf http: //chemconnections. org/general/chem 108/Molecular%20 Modelingintro%20%26%20 table%202019 f. pdf Begin in Lab this week as a Group & Turn-in individually Due 23 -Oct
- Slides: 13