Chem 108 Lab Week 8 Experiment Whats My

Chem 108: Lab Week 8 Experiment: What’s My Formula? II Sign in; Sit with Group. Front of Lab C D E F G Work with the reorganized groups from last week’s lab.
Chem 108: Lab Week 8 Experiment: What’s My Formula? II Sign in; Sit with Group. Front of Lab A B C D E F Work with the reorganized groups from last week’s lab.

DUE Today Names, Ions, Formulas Complete Report Form pp. 109 -114 1 form per Individual or 1 per group: With names of only those who contributed on the form.

Bonds: Molecular Shapes: Molecular Modeling http: //molview. org Have completed first & second pages checked Before leaving lab

Molecular Modeling (Individual or Collaborative) Report Form (Replacement pages for Molecular Model Lab pp. 97 -103) http: //chemconnections. org/general/chem 108/Chemistry%20108%20 Molecular%20 Modeling%20 Fo rm%20 Fall%202017. pdf Computers & Internet available in PS 110, if needed http: //molview. org Turn-in individually or one per group Consult Calendar for Due Date

What’s My Formula? Identificaton Unknown Sample Salt 85. 26% 79. 09% 63. 08% Unknown Sample Salt 69. 02% Unknown Sample Salt Experimental Calculation: Comparison to Calculation(s) for a, b, c, d FROM last week: ? ?

What’s My Formula? Your group obtained 2 to 5 unknowns. Complete the experimental procedures and submit one complete report form for each unknown with partner’s names on the data form page & a complete set of calculations for each unknown with % Yield & Theoretical Yield Calculations (replacements for pg. 36) Complete Report Forms DUE Today

Theoretical Mass Calculations for any Reaction Reactants Products grams (Reactant) grams (Product) Moles Molar Mass grams (R) 1 mol (R) grams (R) (Divide) by Molar Mass (R) ? mol (P) ? mol (R) grams (P) ? grams (P) 1 mol (P) (Multiply) "Gatekeepers” from Balanced reaction by Molar Mass (P)

What’s My Formula? % Yield (Example) Heating 10. 00 g of an unknown determined to be sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. What is the Percent Yield? First calculate theoretical yield. (Adaptation of your calculations last week. ) It considers in the calculation that everything went perfectly, and is based on the assumption of 100% accuracy. % Yield is actual; based on reality. Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = ? g (Theoretical) Molar Mass = 105. 99 g/mol
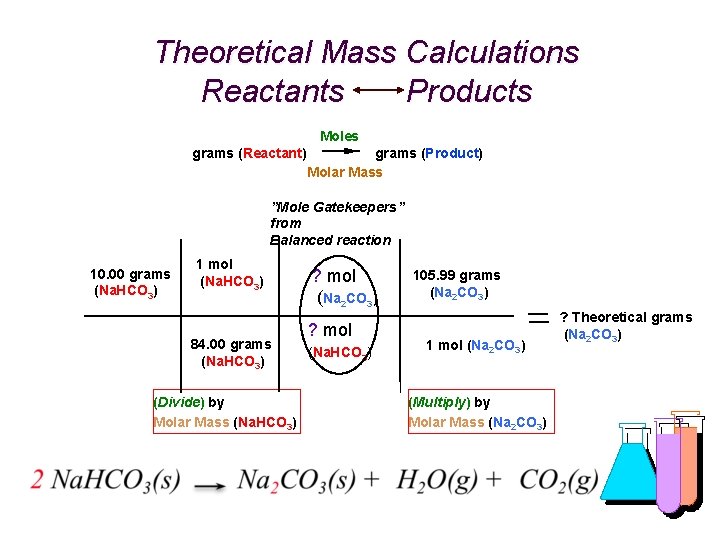
Theoretical Mass Calculations Reactants Products Moles grams (Reactant) grams (Product) Molar Mass ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) ? mol (Na 2 CO 3) ? mol (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) ? Theoretical grams (Na 2 CO 3)
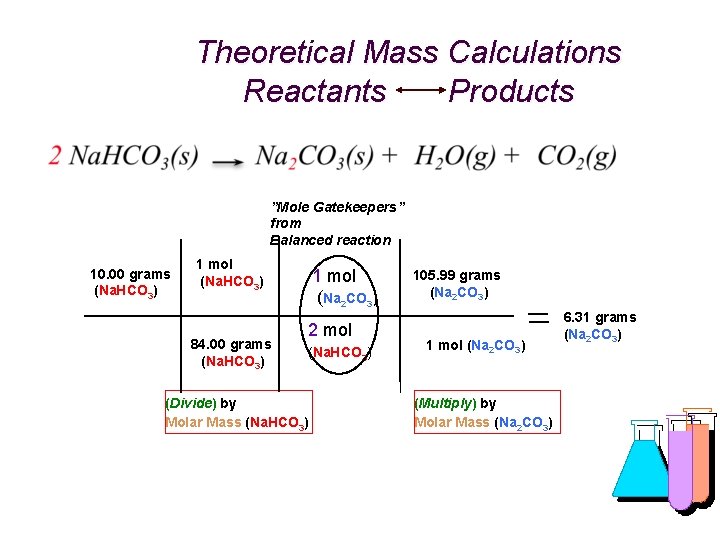
Theoretical Mass Calculations Reactants Products ”Mole Gatekeepers” from Balanced reaction 10. 00 grams (Na. HCO 3) 1 mol (Na. HCO 3) 84. 00 grams (Na. HCO 3) 1 mol (Na 2 CO 3) 2 mol (Na. HCO 3) (Divide) by Molar Mass (Na. HCO 3) 105. 99 grams (Na 2 CO 3) 1 mol (Na 2 CO 3) (Multiply) by Molar Mass (Na 2 CO 3) 6. 31 grams (Na 2 CO 3)

What’s My Formula? “% Yield” is used to measure the efficiency (similar to “accuracy”) of any reaction in yielding “product(s)” (on the right of an equation) versus the calculated (theoretical) amount of the product based on the amount of “reactant(s)” (from the left of the equation) using the relative number of moles of each in a balanced chemical equation. % Yield = actual grams of product / theoretical (calculated) grams of product x 100 For example, heating 10. 00 g of sodium bicarbonate and actually obtaining 5. 98 g of sodium carbonate. After calculating theoretical yield: Reactant = 10. 00 g Molar Mass = 84. 00 g/mol Product = 6. 31 g (Theoretical) Molar Mass = 105. 99 g/mol % Yield = 5. 98 g (actual) / 6. 31 g (theoretical) x 100 = 94. 6%
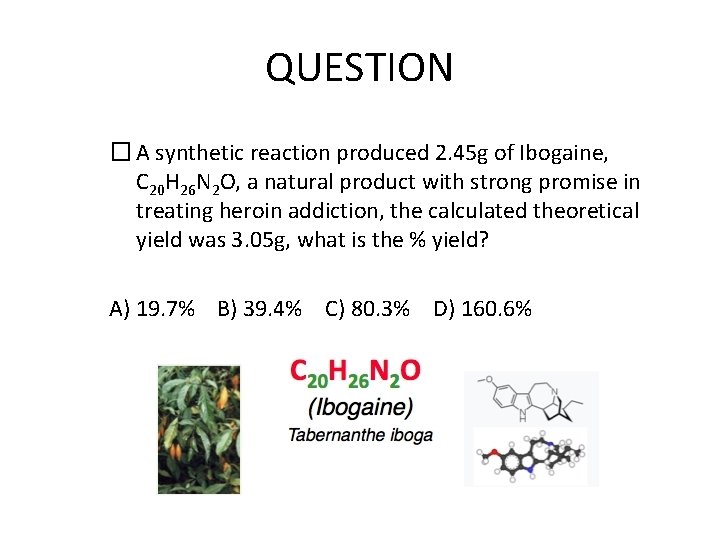
QUESTION � A synthetic reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, the calculated theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6%

ANSWER � If a reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, and theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6% % yield = 2. 45 g / 3. 05 g x 100 = 80. 3%

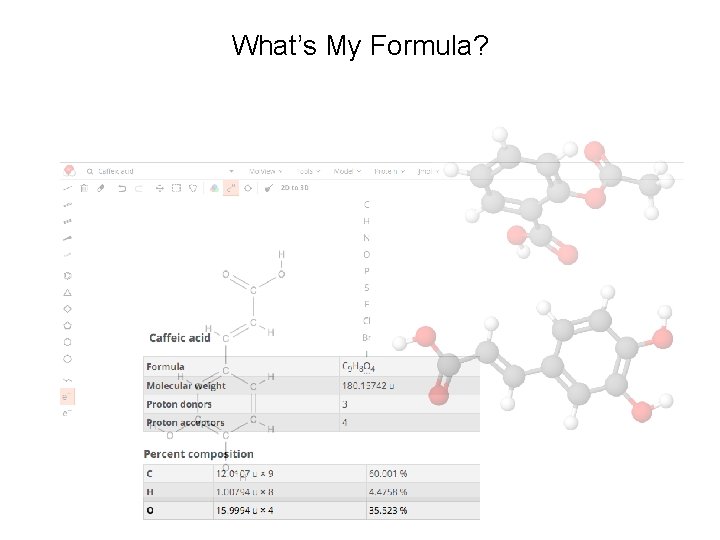
Post Lab: Compounds with the Same Formula [ eg. C 9 H 8 O 4 ] Molar Comparisons of Analgesics Calculate Moles : Doses (mmol/dose) Which analgesic has the most biologically active ingredient based on millimoles per dose (mmol/dose)? 5. 0 g of the active ingredient would produce the following number of doses: Formula Doses mmol/dose Aspirin C 9 H 8 O 4 15. 01. 8 mmol/dose Ibuprofen C 13 H 18 O 2 25. 0 ? Naproxen Sodium C 14 H 13 O 3 Na 22. 7 ? Acetaminophen C 8 H 9 NO 2 5. 0 ? Molar Mass Aspirin = 180. 1 g/mol 5. 0 g / 180. 1 g/mol = 0. 028 mol/15 doses = 1. 8 mmol/dose

Molar Comparisons of Analgesics Calculate Moles : Doses (mmol/dose) Post Lab: Must submit Individually From calendar link DUE Today

What’s My Formula?

Post Lab: What’s My Formula? Comparisons of Analgesics (mmol/dose) Which analgesic has the most biologically active ingredient based on millimoles per dose (mmol/dose)? 5. 0 g of each would produce the following number of doses: Formula Doses Aspirin C 9 H 8 O 4 15. 4 Ibuprofen C 13 H 18 O 2 25 Naproxen Sodium C 14 H 13 O 3 Na Acetaminophen C 8 H 9 NO 2 5 mmol/dose 22. 7 Molar Mass Aspirin = 180. 1 g/mol 5. 0 g / 180. 1 g/mol = 0. 028 mol = 28 mmol
Chem 108: Lab Week 8 Sign in Pick up papers Sit with group partners from last week’s lab G D B H A E C

Molecular Modeling Computational Chemistry Covalent Bonds: Lewis Structures, Molecular Shapes Dr. Ron Rusay

Molecular Modeling Computational Chemistry Shapes Lewis Structures Polarity, Solubility, Stereochemistry Smell, Teratogenicity Dr. Ron Rusay

https: //www. youtube. com/watch? v=Jq_Ca-HKh 1 g Shapes of Molecules View: What is the shape of a molecule? George Zaidan and Charles Morton http: //chemconnections. org/general/chem 1 08/Molecular%252520 Shapes-Guide. html
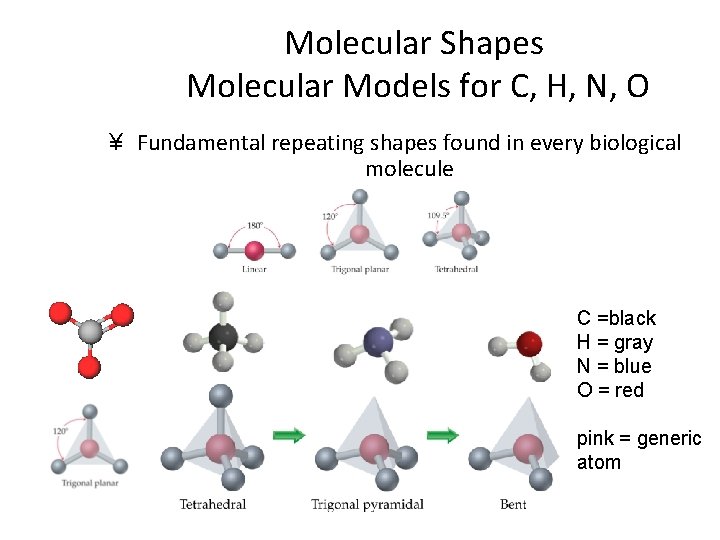
Molecular Shapes Molecular Models for C, H, N, O ¥ Fundamental repeating shapes found in every biological molecule C =black H = gray N = blue O = red pink = generic atom

Professor Gilbert Newton Lewis (circa 1940) G. N. Lewis Photo Bancroft Library, University of California/LBNL Image Library Footnote: G. N. Lewis, despite his insight and contributions to chemistry, was never awarded the Nobel prize. Notes from Lewis’s notebook and his “Lewis” structure.
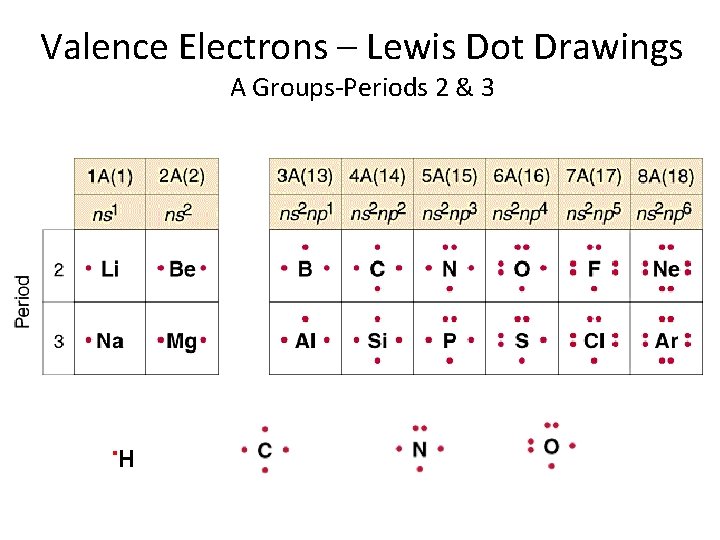
Valence Electrons – Lewis Dot Drawings A Groups-Periods 2 & 3 . H
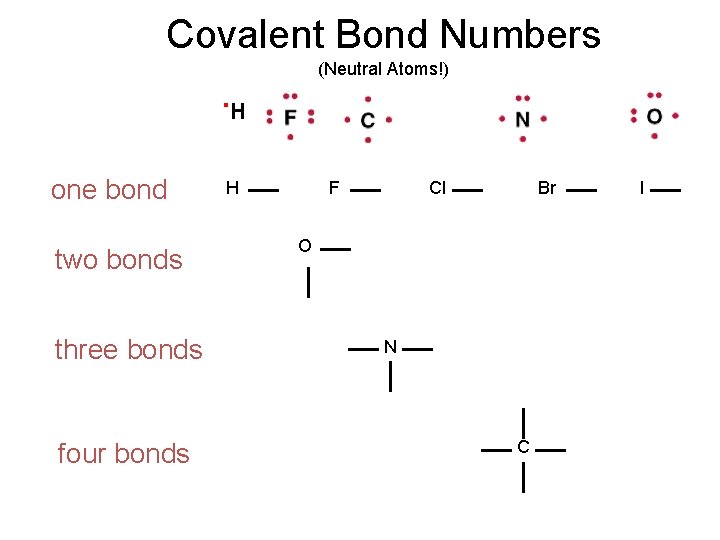
Covalent Bond Numbers (Neutral Atoms!) . H one bond two bonds three bonds four bonds H F Cl Br O N C I
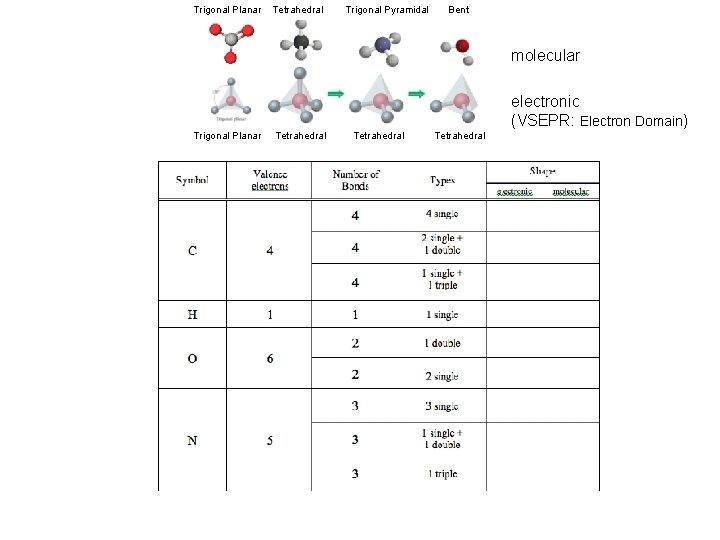
Trigonal Planar Tetrahedral Trigonal Pyramidal Bent (Handout) molecular electronic (VSEPR: Electron Domain) Trigonal Planar Tetrahedral Due End of Lab Next Week
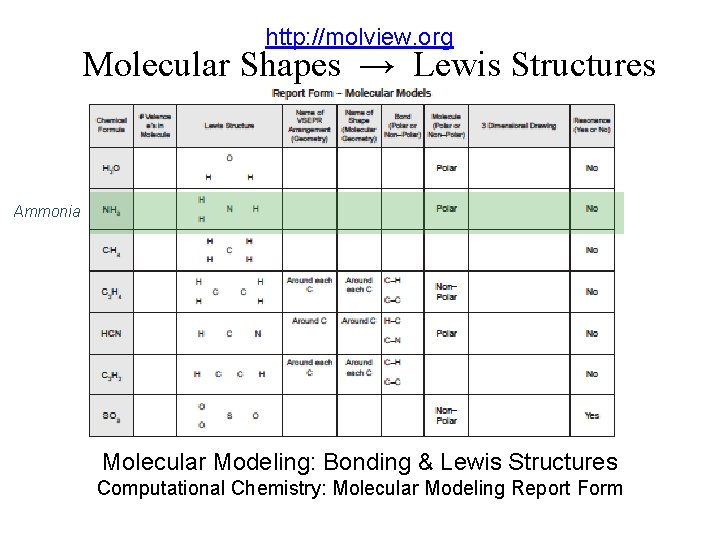
http: //molview. org Molecular Shapes → Lewis Structures Ammonia Molecular Modeling: Bonding & Lewis Structures Computational Chemistry: Molecular Modeling Report Form
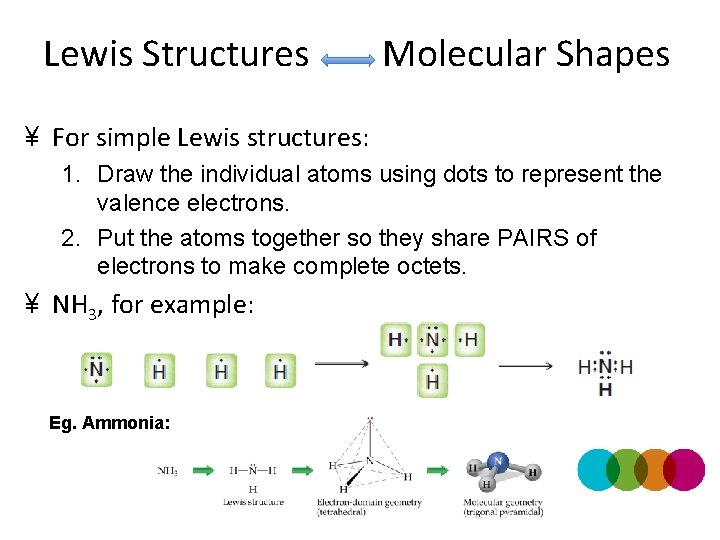
Lewis Structures Molecular Shapes ¥ For simple Lewis structures: 1. Draw the individual atoms using dots to represent the valence electrons. 2. Put the atoms together so they share PAIRS of electrons to make complete octets. ¥ NH 3, for example: Eg. Ammonia:
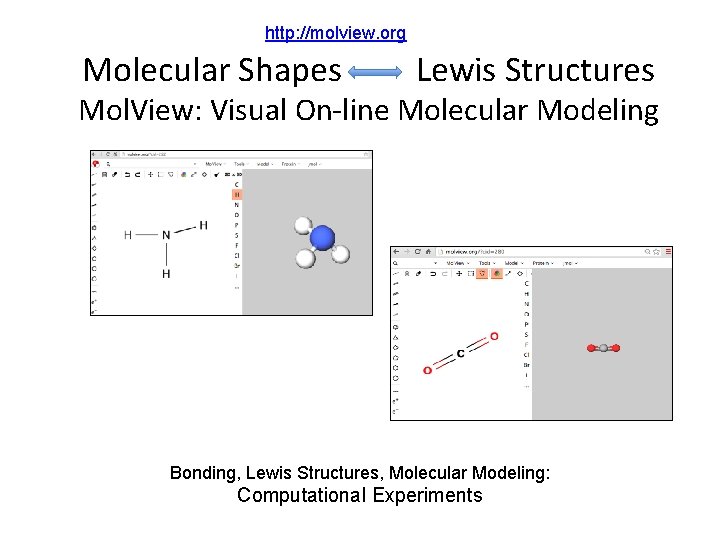
http: //molview. org Molecular Shapes Lewis Structures Mol. View: Visual On-line Molecular Modeling Bonding, Lewis Structures, Molecular Modeling: Computational Experiments
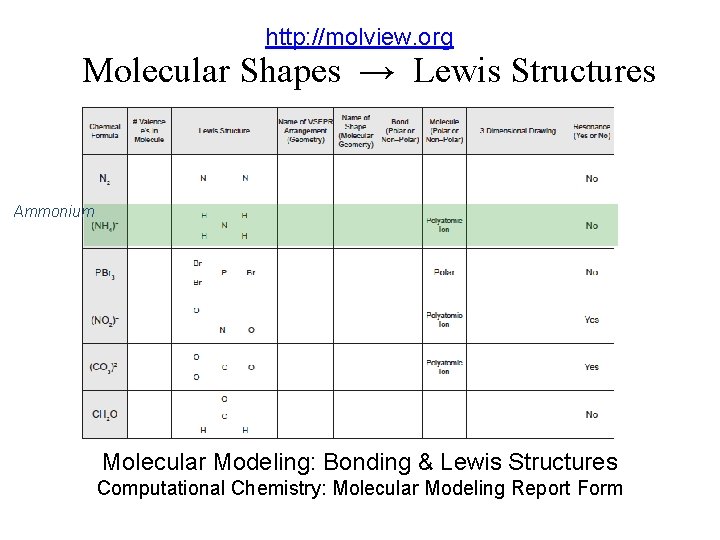
http: //molview. org Molecular Shapes → Lewis Structures Ammonium Molecular Modeling: Bonding & Lewis Structures Computational Chemistry: Molecular Modeling Report Form

http: //molview. org Molecular Shapes → Lewis Structures Mol. View: Visual On-line Molecular Modeling Ammonium Molecular Modeling: Bonding & Lewis Structures Computational Chemistry: Molecular Modeling Report Form
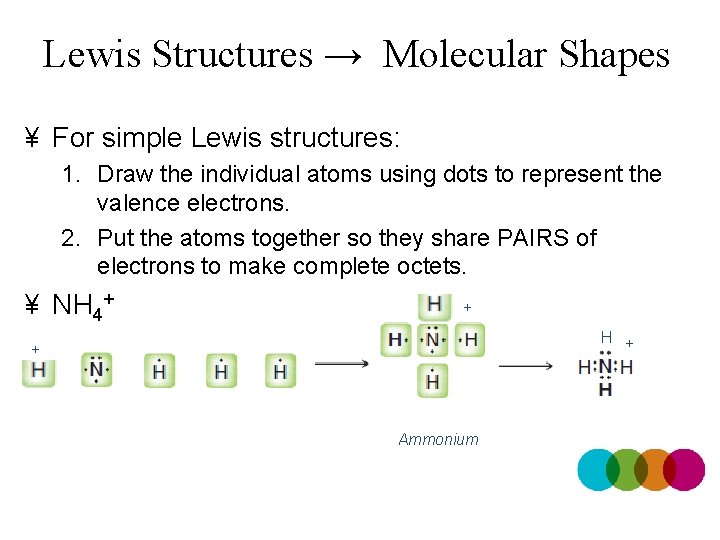
Lewis Structures → Molecular Shapes ¥ For simple Lewis structures: 1. Draw the individual atoms using dots to represent the valence electrons. 2. Put the atoms together so they share PAIRS of electrons to make complete octets. ¥ NH 4+ + H + + Ammonium

Orbital (Electronic) Molecular Geometry Important Linear in Trigonal Planar Organic Compounds Trigonal Planar Bent Bond Angle # of lone pairs 0 0 1 Tetrahedral 0 Tetrahedral Trigonal Pyramidal 1 Tetrahedral Bent 2 See again Trigonal Bipyramidal Chem 120 and possibly. Trigonal Bipyramidal in Chem 109 Trigonal Bipyramidal 0 Seesaw 1 T-shape 2 Linear 3 Octahedral 0 Octahedral Square Pyramidal 1 Octahedral Square Planar 2
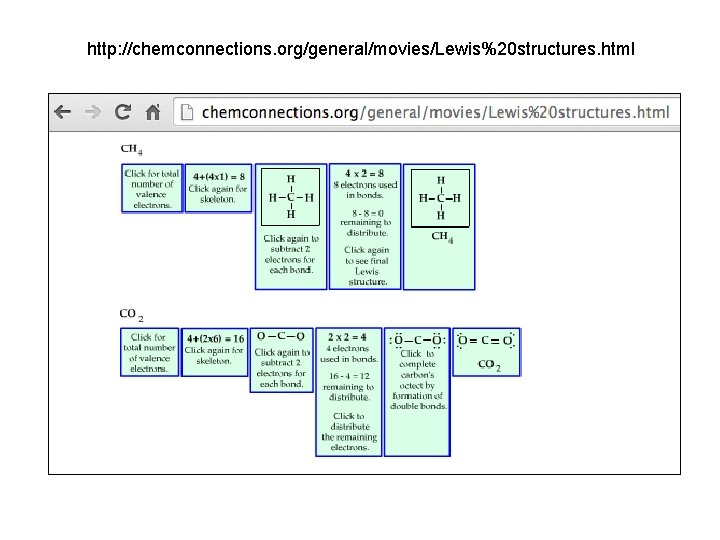
http: //chemconnections. org/general/movies/Lewis%20 structures. html

Covalent Compounds • Share valence electrons. • 1 pair = 1 bond; maximum # of atom-atom bonds = 3. • Octet rule (“duet” for hydrogen) • Lewis structure examples: Notice the charges: In one case they balance, can you name the compound? In the other they do not. It has a “Formal” charge. Can you name the polyatomic ion?
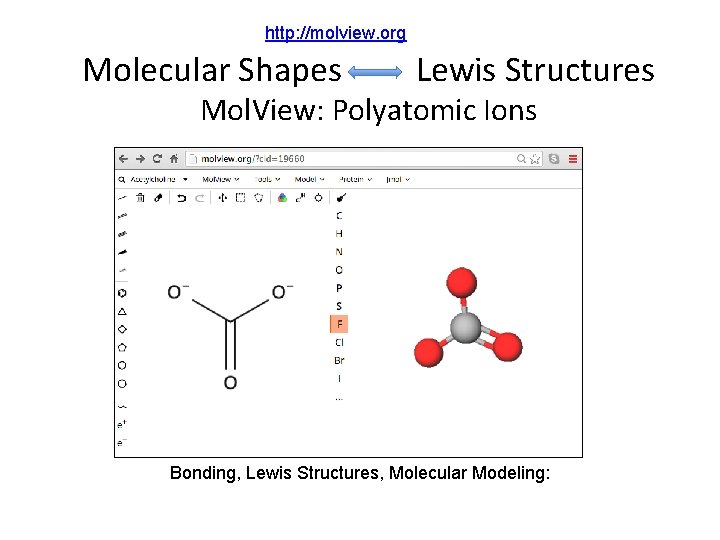
http: //molview. org Molecular Shapes Lewis Structures Mol. View: Polyatomic Ions Bonding, Lewis Structures, Molecular Modeling:
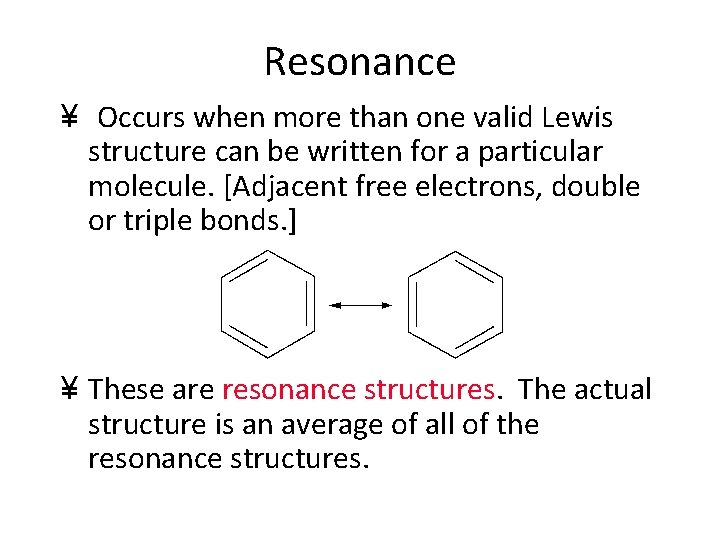
Resonance ¥ Occurs when more than one valid Lewis structure can be written for a particular molecule. [Adjacent free electrons, double or triple bonds. ] ¥ These are resonance structures. The actual structure is an average of all of the resonance structures.
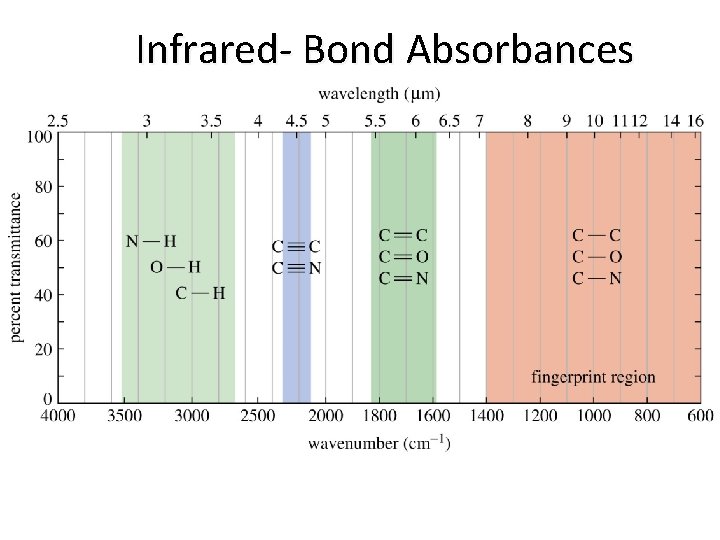
Infrared- Bond Absorbances
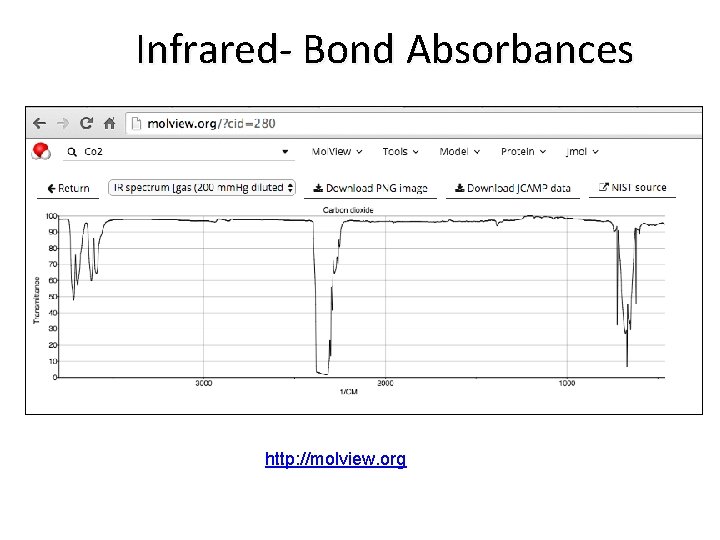
Infrared- Bond Absorbances http: //molview. org

Infrared- Bond Absorbances https: //www. co 2. earth/
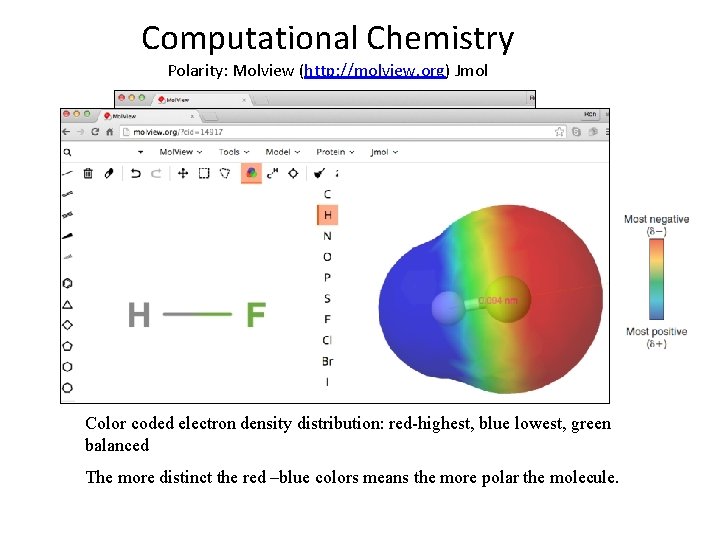
Computational Chemistry Polarity: Molview (http: //molview. org) Jmol Color coded electron density distribution: red-highest, blue lowest, green balanced The more distinct the red –blue colors means the more polar the molecule.
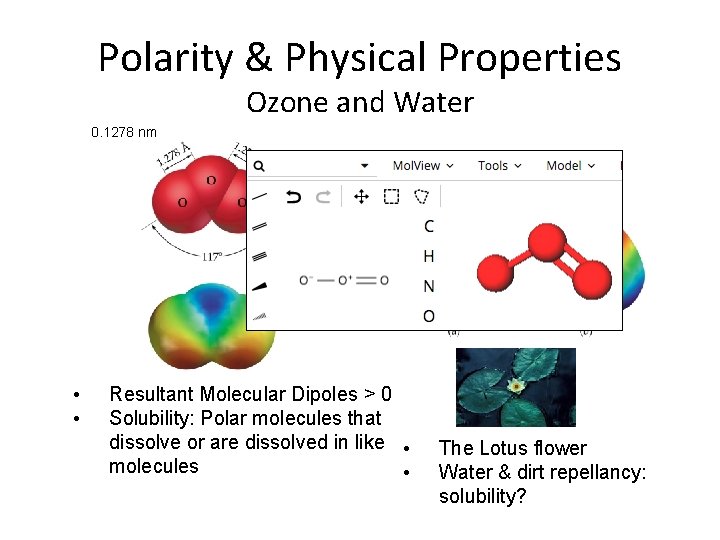
Polarity & Physical Properties Ozone and Water 0. 1278 nm • • Resultant Molecular Dipoles > 0 Solubility: Polar molecules that dissolve or are dissolved in like • molecules • The Lotus flower Water & dirt repellancy: solubility?
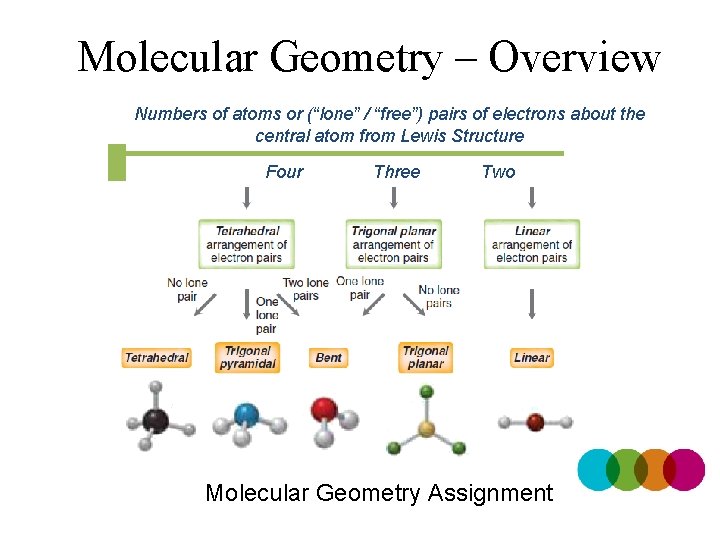
Molecular Geometry – Overview Numbers of atoms or (“lone” / “free”) pairs of electrons about the central atom from Lewis Structure Four Three Two Molecular Geometry Assignment
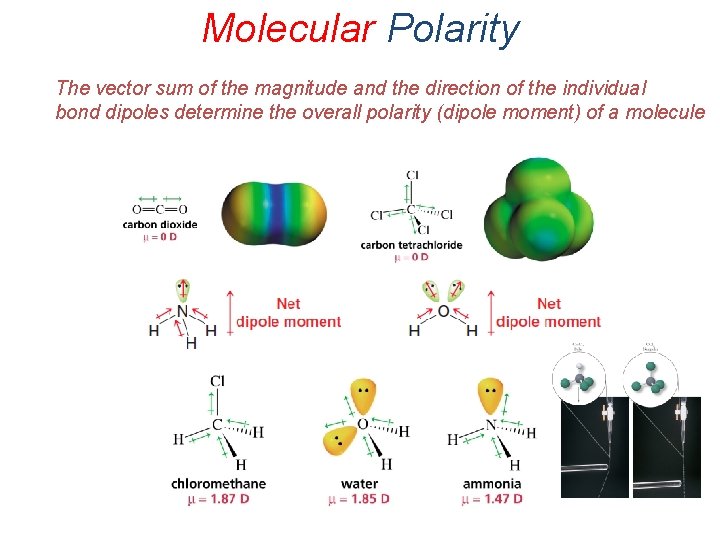
Molecular Polarity The vector sum of the magnitude and the direction of the individual bond dipoles determine the overall polarity (dipole moment) of a molecule
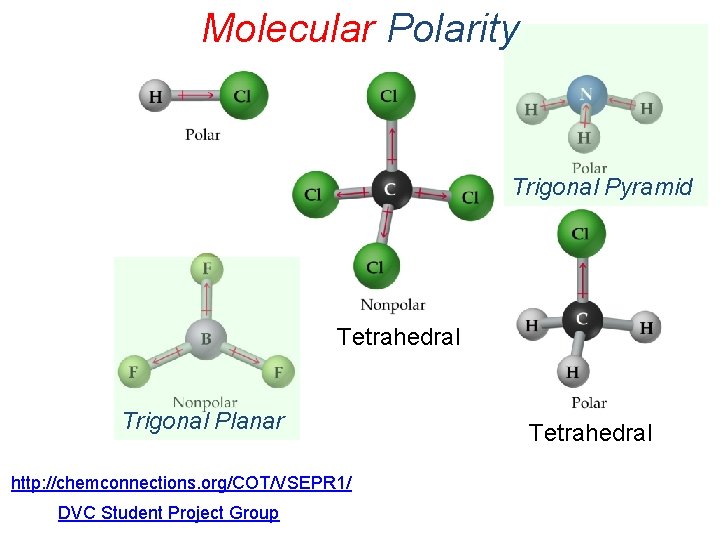
Molecular Polarity Trigonal Pyramid Tetrahedral Trigonal Planar http: //chemconnections. org/COT/VSEPR 1/ DVC Student Project Group Tetrahedral
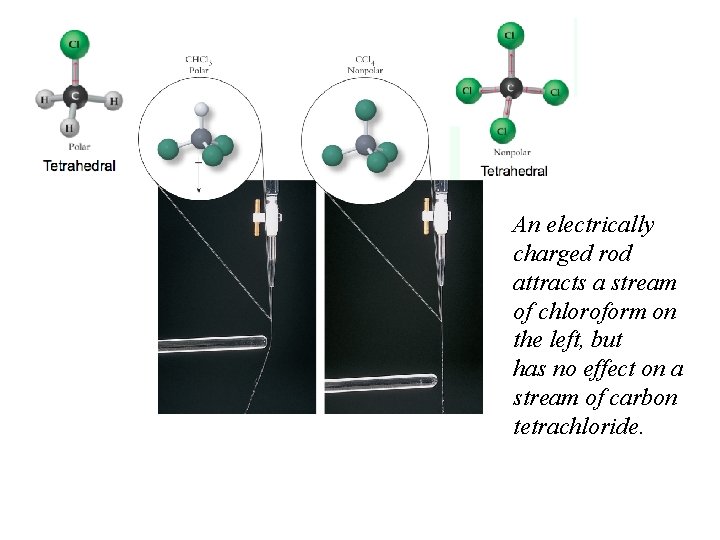
An electrically charged rod attracts a stream of chloroform on the left, but has no effect on a stream of carbon tetrachloride.

What’s My Formula? Post Lab: Compounds with the Same Formula [ eg. C 9 H 8 O 4 ] Aspirin 4 -Hydroxyphenylpyruvic acid Dihydroxycinnamic acids: Caffeic acid (3, 4 -dihydroxycinnamic acid) Umbellic acid (2, 4 -dihydroxycinnamic acid) 2, 3 -Dihydroxycinnamic acid 2, 5 -Dihydroxycinnamic acid 3, 5 -Dihydroxycinnamic acid
- Slides: 49