Chem 108 Lab Week 3 Sign in Roster

Chem 108: Lab Week 3 Sign in: Roster @ front of lab Turn in Safety Questions if you have not yet done so.

Last Week’s Experiment: Metric Measurement Completed individual forms pp. 12 -15 due today by the end of lab. Include calculations. Staple together with partner’s form and turn in before the end of lab. Most legible report pages on top.

Exp. 1 – Metric Measurement Example of an acceptable set of student data, conversions, and calculations. DO NOT COPY. Use as a guide.
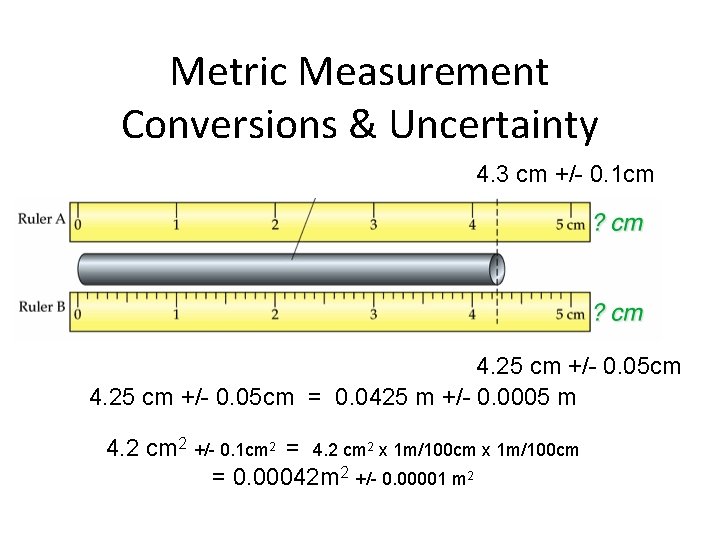
Metric Measurement Conversions & Uncertainty 4. 3 cm +/- 0. 1 cm 4. 25 cm +/- 0. 05 cm = 0. 0425 m +/- 0. 0005 m 4. 2 cm 2 +/- 0. 1 cm 2 = 4. 2 cm 2 x 1 m/100 cm = 0. 00042 m 2 +/- 0. 00001 m 2

CHEM 108 Experiment 2: Density

Comparisons of Relative Properties FUNCTIONS Example: density : mass : volume Functions & variables can be described interchangeably between: 1) 2) 3) 4) 5) 6) 7) Images (Symbols) Words Sounds (Voice) Numbers Tables Graphs Formulas Density Mass Volume

Experiment 2 Measuring Density Ø Goal: To measure diameter and height of a metal cylinder and calculate the respective volume. Ø To plot mass versus volume and determine slope of “trendline(s)”. Ø Using 2 different methods, to measure mass and volume of a liquid, and to determine which method results in higher–precision (most decimal places) Ø To measure mass and volume of a solid using instruments of different precision, and determine which method results in highest–precision Ø Work with same lab partner as the Metric Measurement Experiment Ø Be sure to write partners’ names ON both REPORT FORMS DUE Next Week

Chem 108 Lab: Week 2
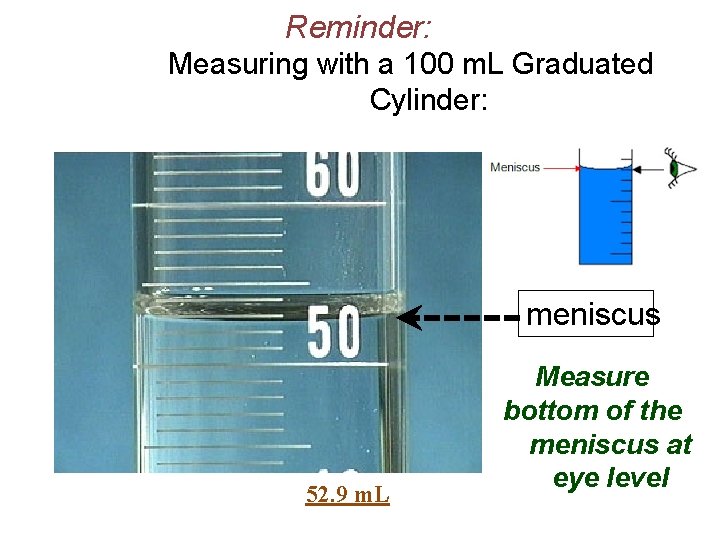
Reminder: Measuring with a 100 m. L Graduated Cylinder: meniscus 52. 9 m. L Measure bottom of the meniscus at eye level

Ø Measurement using a volumetric pipet: meniscus Ø Bottom of the meniscus MUST be at calibration line for accurate measurement
Ø Replace Part C, pg. 19 with handout http: //chemconnections. org/general/chem 108/Measuring%20 Density-graphing. 2017. pdf

Ø Add 2 Columns to pg. 22 Each partner to measure with both Caliper and Ruler & compare
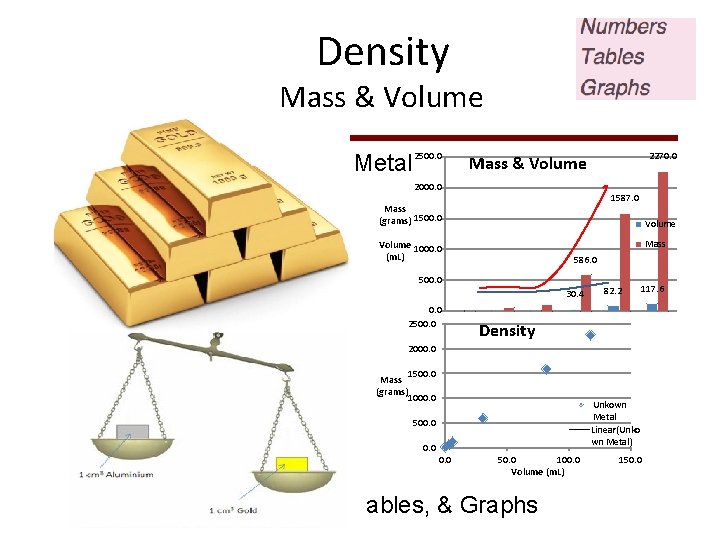
Density Mass & Volume Unknown Metal 2500. 0 2000. 0 Sample Mass (grams)Volume (m. L) 1 10. 0 0. 5 2 55. 0 2. 8 3 113. 5 5. 9 4 586. 0 30. 4 5 1587. 0 82. 2 6 2270. 0 117. 6 1587. 0 Mass (grams) 1500. 0 Volume Mass Volume 1000. 0 (m. L) 586. 0 500. 0 30. 4 0. 0 2500. 0 2000. 0 82. 2 117. 6 Density 1500. 0 Mass (grams) 1000. 0 Unkown Metal Unkown. . . Linear(Unko wn Metal) 500. 0 0. 0 2270. 0 Mass & Volume 50. 0 100. 0 Volume(m. L) Numbers, Tables, & Graphs 150. 0
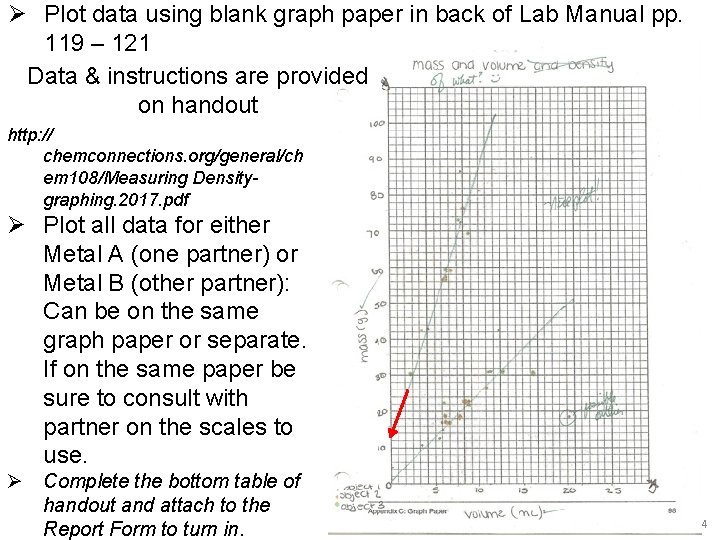
Ø Plot data using blank graph paper in back of Lab Manual pp. 119 – 121 Data & instructions are provided on handout http: // chemconnections. org/general/ch em 108/Measuring Densitygraphing. 2017. pdf Ø Plot all data for either Metal A (one partner) or Metal B (other partner): Can be on the same graph paper or separate. If on the same paper be sure to consult with partner on the scales to use. Ø Complete the bottom table of handout and attach to the Report Form to turn in. 14
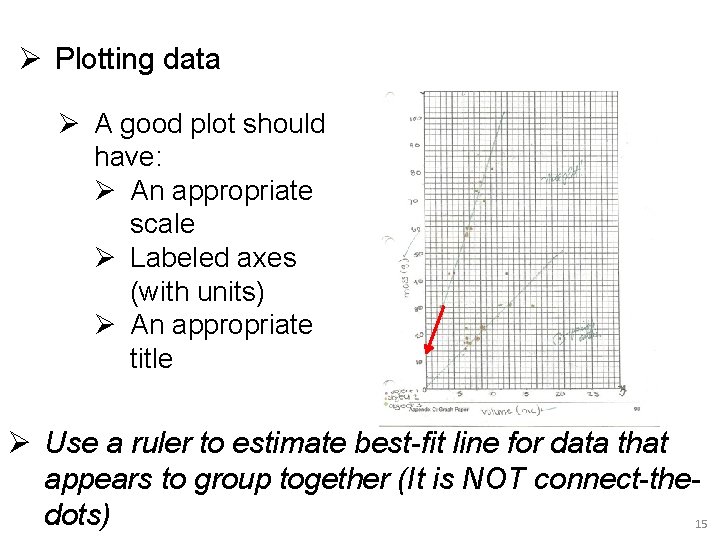
Ø Plotting data Ø A good plot should have: Ø An appropriate scale Ø Labeled axes (with units) Ø An appropriate title Ø Use a ruler to estimate best-fit line for data that appears to group together (It is NOT connect-thedots) 15

Experiment 2 – Measuring Density Ø “Extrapolate your lines through the y (vertical) axis” Ø This means draw your line all the way to the y-axis Error / Accuracy 4 experimental Value = Average sets of data True =1. 0 g/m. L Exp. = 1. 0 g/m. L True =1. 0 g/m. L Exp. = 1. 1 g/m. L - + - True =1. 0 g/m. L Exp. = 1. 0 g/m. L + - + True =1. 0 g/m. L Exp. = 1. 2 g/m. L
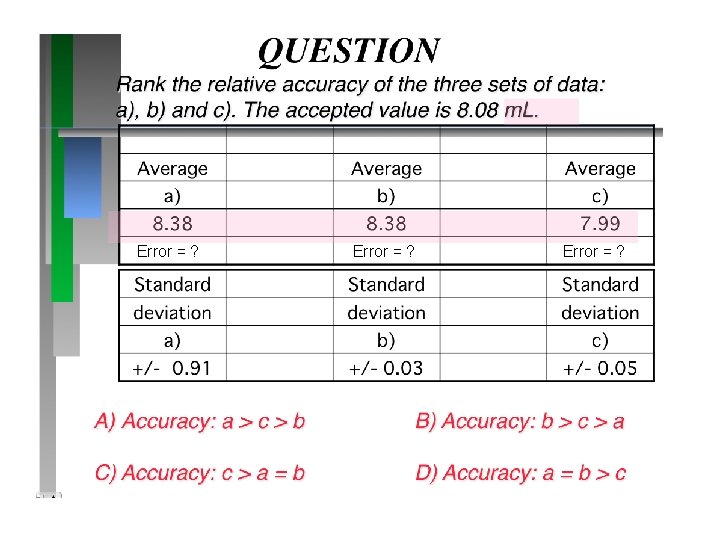
Error = ?
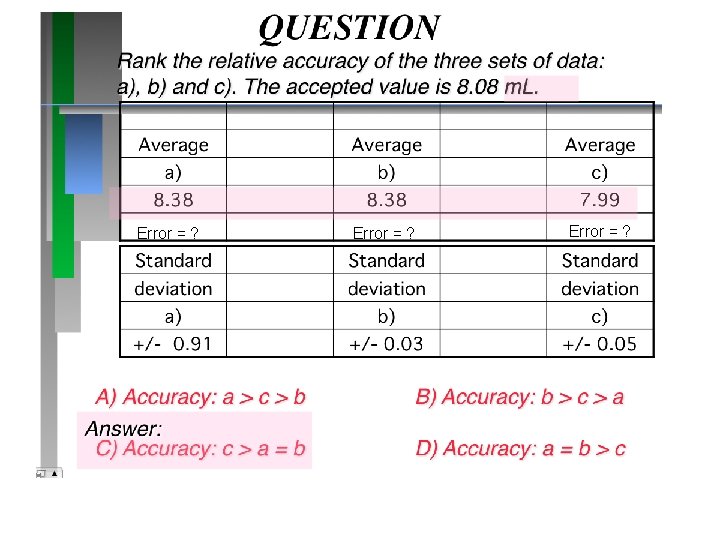
Error = ?

Experiment 2 – Measuring Density Ø “Extrapolate your lines through the y (vertical) axis” Ø This means draw your line all the way to the y-axis Error / Accuracy & Precision True =1. 0 g/m. L Exp. = 1. 0 +/- 0. 1 g/m. L True =1. 0 g/m. L Exp. = 1. 1 +/- 0. 1 g/m. L - + - True =1. 0 g/m. L Exp. = 1. 0 +/- 0. 2 g/m. L + - + True =1. 0 g/m. L Exp. = 1. 2 +/- 0. 2 g/m. L
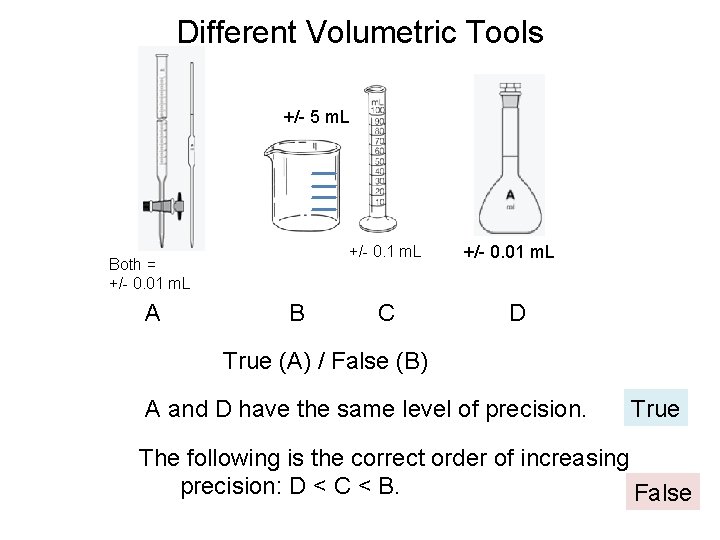
Different Volumetric Tools +/- 5 m. L Both = +/- 0. 01 m. L A +/- 0. 1 m. L +/- 0. 01 m. L B C D True (A) / False (B) A and D have the same level of precision. True The following is the correct order of increasing precision: D < C < B. False
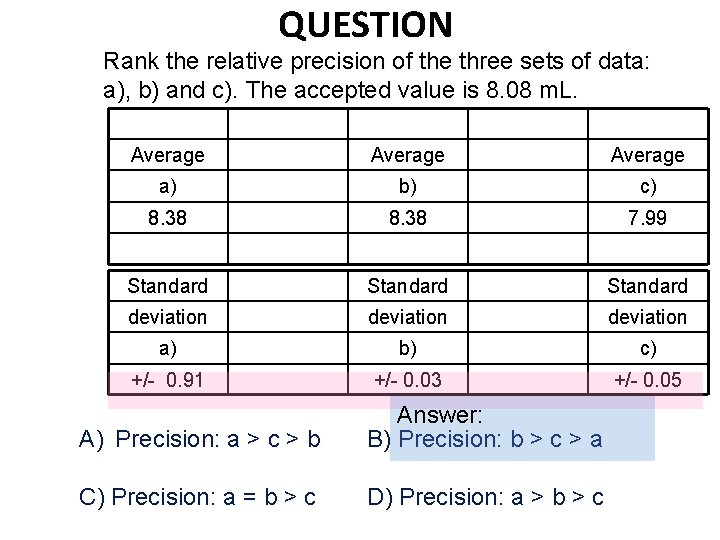
QUESTION Rank the relative precision of the three sets of data: a), b) and c). The accepted value is 8. 08 m. L. Average a) b) c) 8. 38 7. 99 Standard deviation a) b) c) +/- 0. 91 +/- 0. 03 +/- 0. 05 A) Precision: a > c > b Answer: B) Precision: b > c > a C) Precision: a = b > c D) Precision: a > b > c

Equation of a line: y = mx + b

Next Week • Check Calendar for assignments • Complete Density calculations, graphs & Report Form pp. 20 -25 (One form for each lab partner to be turned in; stapled together clearest written form first. ) DUE Next Lab Period • (GQ) On-line Density & Buoyancy Guiding Questions (individually done) DUE Next Lab Period https: //phet. colorado. edu/sims/density-andbuoyancy/density_en. html

• (GQ) On-line Density & Buoyancy Guiding DUE Next Lab Period http: //chemconnections. org/general/chem 108/Density%20&%20 Bouyancy. html

• (GQ) On-line Density & Buoyancy Guiding DUE Next Lab Period https: //phet. colorado. edu/sims/density-andbuoyancy/density_en. html Come up with partner with lab forms completed with names. Dr. R will provide unknown liquid & unknown metal, caliper & ruler.

Come up with partner with lab forms completed with names. Dr. R will provide unknown liquid & unknown metals, caliper & ruler. Read instructions carefully & discuss with partner before beginning. Return empty liquid vials & metal cylinders when finished. Liquid is salt solution that can be poured down drain. Caliper

Ø 2 Columns added to pg. 22 Each partner to measure with both Caliper and Ruler & compare Using the caliper Come up with partner with lab forms completed with names. Dr. R will provide unknown liquid & unknown metals, caliper & ruler. Read instructions carefully & discuss with partner before beginning.
- Slides: 27