Chem 108 Lab Week 15 Sign in Note

Chem 108: Lab Week 15 Sign in Note your Group # Pick up Papers and Handout
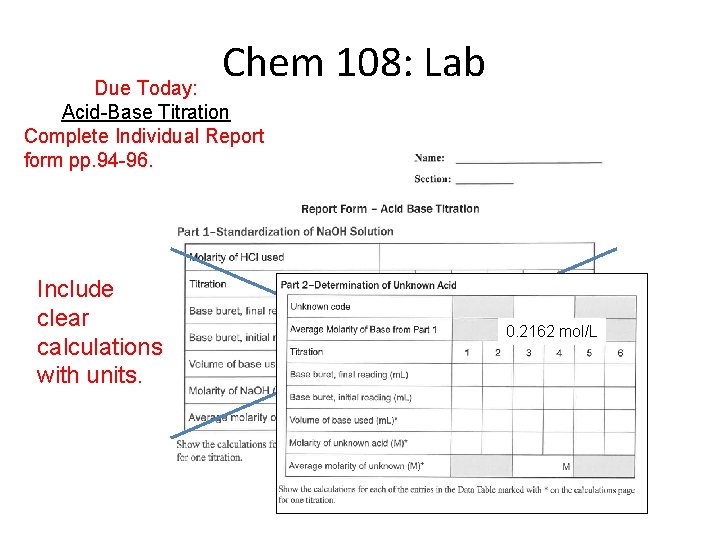
Chem 108: Lab Due Today: Acid-Base Titration Complete Individual Report form pp. 94 -96. Include clear calculations with units. 0. 2162 mol/L
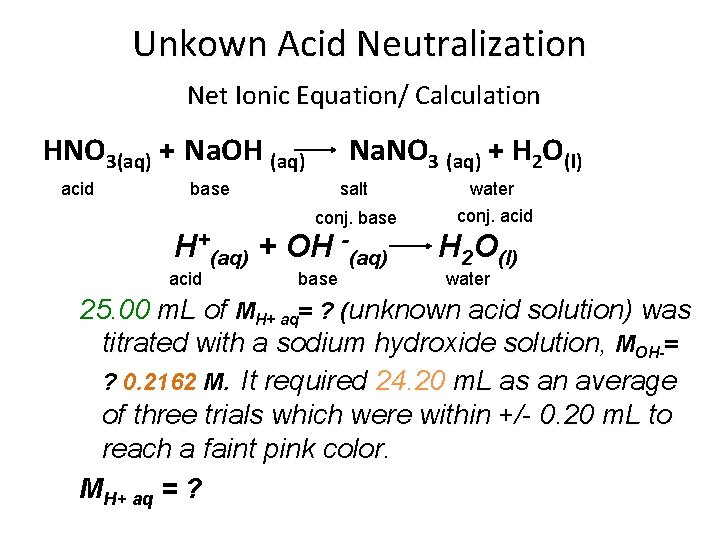
Unkown Acid Neutralization Net Ionic Equation/ Calculation HNO 3(aq) + Na. OH (aq) acid Na. NO 3 (aq) + H 2 O(l) base salt conj. base H+(aq) + OH -(aq) acid base water conj. acid H 2 O(l) water 25. 00 m. L of MH+ aq= ? (unknown acid solution) was titrated with a sodium hydroxide solution, MOH-= ? 0. 2162 M. It required 24. 20 m. L as an average of three trials which were within +/- 0. 20 m. L to reach a faint pink color. MH+ aq = ?

Unkown Acid Neutralization Net Ionic Equation/ Calculation H+(aq) + OH -(aq) acid base H 2 O(l) water 25. 00 m. L of MH+ aq= ? (unknown acid solution) was titrated with a sodium hydroxide solution, MOH-= ? 0. 2162 M. It required 24. 20 m. L as an average of three trials which were within +/- 0. 20 m. L to reach a faint pink color. ? MH+ = = [MOH- x VOH- / VH+ ] [? mol. H+ / ? mol. OH-] x 0. 02420 LOH- x 1 mol. H+ LOH- x 0. 02500 LH+ x 1 mol. OH- 0. 2162 mol. OH- = 0. 2093 MH+

QUESTION A 35. 00 m. L sample of a monoprotic acid of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the unknown acid? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2719 M 0. 1860 M 0. 3720 M

Answer A 35. 00 m. L sample of a monoprotic acid of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the unknown acid? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2719 M 0. 1860 M 0. 3720 M = ? MH+ x 0. 04230 LOH- x 1 mol. H+ LOH- x 0. 03500 LH+ x 1 mol. OH- 0. 2250 mol. OH- H+(aq) + OH -(aq) = [MOH- x H 2 O(l) VOH- / VH+ ] [1 mol. H+ / 1 mol. OH-]

Join with your Group members Read the handout Stoichiometry / Limiting Reactant / Ideal Gas Law http: //chemconnections. org/general/chem 108/Gas-Limiting%20 Reactant-demo. 2018. pdf Acid-Base Reactions & Behavior of Gases
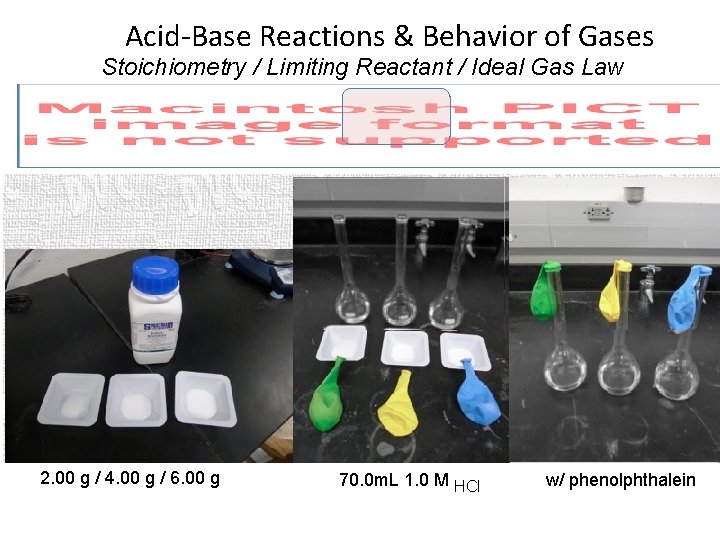
Acid-Base Reactions & Behavior of Gases Stoichiometry / Limiting Reactant / Ideal Gas Law 2. 00 g / 4. 00 g / 6. 00 g 70. 0 m. L 1. 0 M HCl w/ phenolphthalein

Acid-Base Reactions & Behavior of Gases Stoichiometry / Limiting Reactant / Ideal Gas Law Follow the accompanying slides Complete the handout

Acid-Base Reactions & Behavior of Gases Stoichiometry / Limiting Reactant / Ideal Gas Law 1 1 1

Acid-Base Reactions & Behavior of Gases Stoichiometry / Limiting Reactant / Ideal Gas Law Molar Mass = 84. 00 g/mol mol. HCl = 1. 0 mol/L x 1. 00 L/1, 000 m. L x 70. 0 m. L mol = 2. 00 g / 84. 00 g/mol = 4. 00 g / 84. 00 g/mol = 6. 00 g / 84. 00 g/mol



Bring completed Report Forms to Dr. R. to get Mg(s) sample(s). Gas Stoichiometry

• Refer to the Procedure section pp. 54 -56. The following slides correspond to the instructions in the procedure.


What is wrong with this set up? Mg or Zn

Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g)
Mg or Zn

• Refer to the Gas Stoichiometry Report Form, pg. 58 -59 • Experimental data is to be obtained for the reaction of a known mass of magnesium metal: Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) • The volume of hydrogen, pressure and temperature determined and recorded. • Moles of hydrogen is calculated using Ideal Gas Law calculations, then calculating mass of the starting magnesium from the number of moles of hydrogen.
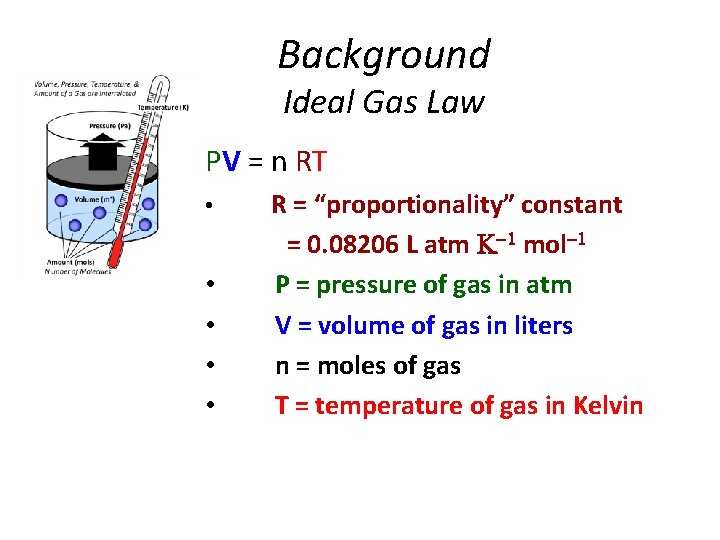
Background Ideal Gas Law PV = n RT • • • R = “proportionality” constant = 0. 08206 L atm mol P = pressure of gas in atm V = volume of gas in liters n = moles of gas T = temperature of gas in Kelvin

Standard Conditions Temperature, Pressure & Moles • “STP” • • For 1 mole of a gas at STP: P = 1 atmosphere T = C (273. 15 K) The molar volume of an ideal gas is 22. 42 liters at STP
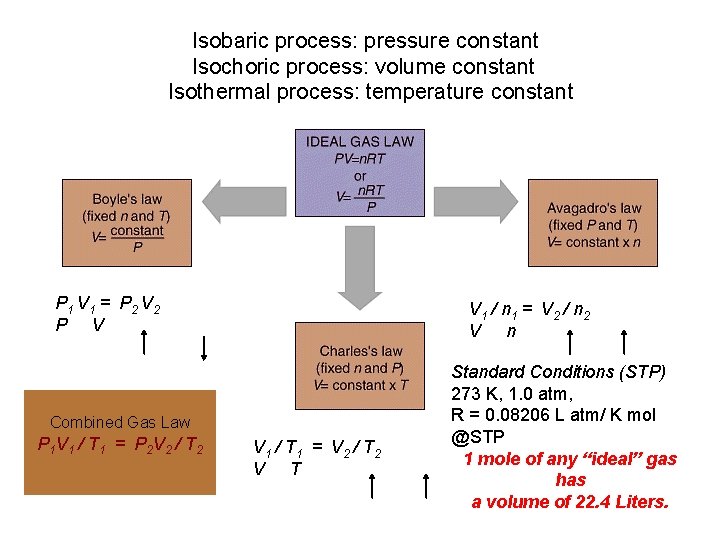
Isobaric process: pressure constant Isochoric process: volume constant Isothermal process: temperature constant P 1 V 1 = P 2 V 2 P V V 1 / n 1 = V 2 / n 2 V n Combined Gas Law P 1 V 1 / T 1 = P 2 V 2 / T 2 V 1 / T 1 = V 2 / T 2 V T Standard Conditions (STP) 273 K, 1. 0 atm, R = 0. 08206 L atm/ K mol @STP 1 mole of any “ideal” gas has a volume of 22. 4 Liters.
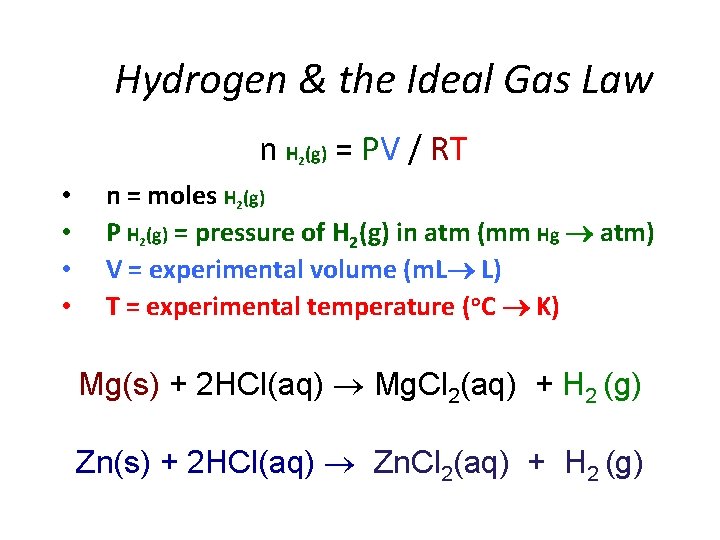
Hydrogen & the Ideal Gas Law n H (g) = PV / RT 2 • • n = moles H 2(g) P H 2(g) = pressure of H 2(g) in atm (mm Hg atm) V = experimental volume (m. L L) T = experimental temperature (o. C K) Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g)

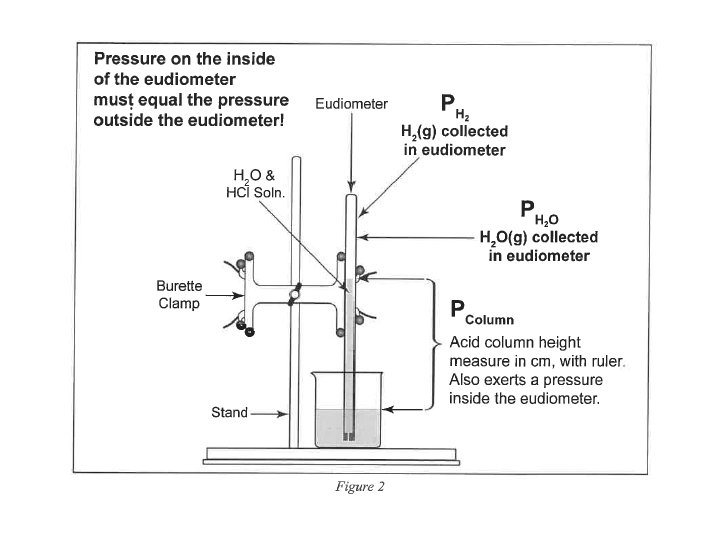
Total Pressure: Sum of the Partial Pressures • For a mixture of gases, the total pressure is the sum of the pressures of each gas in the mixture. PTotal = P 1 + P 2 + P 3 +. . . PTotal n Total n. Total = n 1 + n 2 + n 3 +. . .

• P H 2(g) = P Total (barometric) - P H 2 O (g) [TABLE] - P HCl (g) = HCl Height (mm) ÷ 12. 95 ______ HCl Height (mm) x 0. 0772 ______ Density Hg is 12. 95 times > density HCl(aq) 0. 772 mm Hg/cm of acid solution

Ideal Gas Law: Moles / Avogadro’s Law n H (g) = PV / RT 2 • • • n = moles H 2(g) P H 2(g) = pressure of H 2(g) in atm (mm Hg atm) P H 2(g) = P Total (barometric) - P H 2 O (g) [TABLE] - P HCl (g) V = experimental volume (m. L L) T = experimental temperature (o. C K) R = 0. 082057338 L atm K− 1 mol− 1 (constant) Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g)

• Refer to Report Form Part I: (Example uses Zinc. ) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) Mole Calculations: • Stoichiometry Calculation • Ideal Gas Law Calculations • Comparison (% Error)
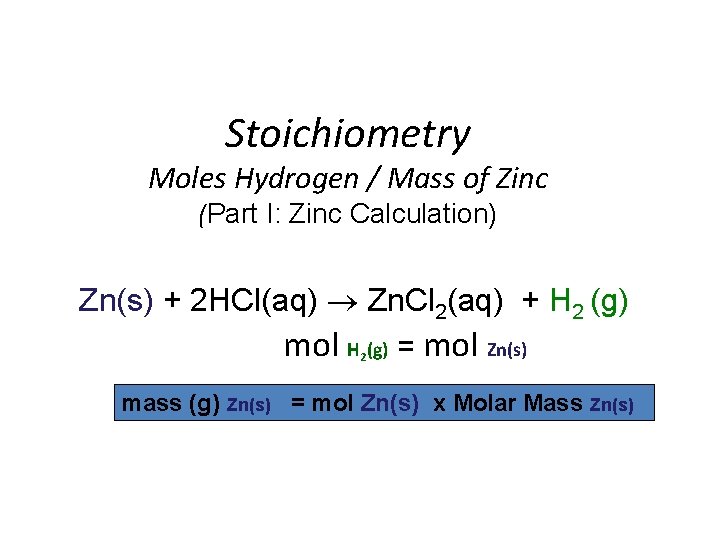
Stoichiometry Moles Hydrogen / Mass of Zinc (Part I: Zinc Calculation) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) mol H (g) = mol Zn(s) 2 mass (g) Zn(s) = mol Zn(s) x Molar Mass Zn(s)

Zinc Example Calculation • Complete Report Form pg. 58 Part I: Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) Mole Calculations: • • • Stoichiometry Calculation Ideal Gas Law Calculations Comparison (% Error)

Moles : Ideal Gas Law (Part I: Zinc Calculation Example) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 • • • n = moles H 2(g) P H 2(g) = pressure of H 2(g) in atm (mm Hg atm) P H 2(g) = P Total (barometric) - P H 2 O (g) [TABLE] - P HCl (g) V = experimental volume (m. L L) T = experimental temperature (o. C K) R = 0. 082057338 L atm K− 1 mol− 1

Moles : Ideal Gas Law (Part I: Zinc Calculation Example) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 V = experimental volume (m. L L) R = 0. 082057338 L atm K− 1 mol− 1

Moles : Ideal Gas Law (Part I: Zinc Calculation Example) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 V = experimental volume (m. L L) T = experimental temperature (o. C K) R = 0. 082057338 L atm K− 1 mol− 1

Moles : Ideal Gas Law (Part I: Zinc Calculation Example) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 V = experimental volume (m. L L) T = experimental temperature (o. C K) P H 2(g) = pressure of H 2(g) in atm (mm Hg atm) P H 2(g) = P Total (barometric) - P H 2 O (g) [TABLE] - P HCl (g) R = 0. 082057338 L atm K− 1 mol− 1

Moles : Ideal Gas Law Part I: Hydrogen Calculation, (Refer to Form’s Data) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 • • • n = moles H 2(g) P H 2(g) = pressure of H 2(g) in atm (mm Hg atm) P H 2(g) = 29. 98 inches Hg (barometric) - 19. 8 mm Hg H 2 O (g) [TABLE] - P HCl (g) R = 0. 082057338 L atm K− 1 mol− 1 19. 2 cm 10. 0 cm

• P H 2(g) = P Total (barometric) - P H 2 O (g) [TABLE] - P HCl (g) = 19. 2 cm Hg - 10. 0 cm Hg = 92 mm Hg HCl Height (mm) ÷ 12. 95 = 7. 1 mm Hg ______ P HCl (g) = 19. 2 cm Hg 10. 0 cm Hg = 92 mm Hg HCl Height (mm) x 0. 0772 = 7. 1 mm Hg _____ 0. 772 mm Hg/cm of acid solution Density Hg is 12. 95 times > density HCl(aq)

P H 2(g) = 761. 5 mm Hg (barometric) - 19. 8 mm Hg H 2 O (g) - 7. 1 mm Hg HCl (g) = 734. 6 mm Hg / = 0. 9666 atm 760. 0 mm Hg / 1. 000 atm

Moles : Ideal Gas Law (Part I: Hydrogen Calculation) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) n H (g) = PV / RT 2 • • n = moles H 2(g) P H 2(g) = 0. 9666 atm V = 0. 0815 L T = 295. 1 K R = 0. 08206 L atm K− 1 mol− 1 n H (g) = 0. 00325 moles Zn(s) 2 2

% Error Theoretical Mass Zinc vs. Experimental (Part I: Calculation) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) mass (g) Zn(s) = mol Zn(s) x Molar Mass Zn(s) = 0. 00325 moles Zn(s) x 65. 37 g/mol Zn(s) experimental grams Zn(s) - theoretical grams Zn(s) x 100 % Error = theoretical grams Zn(s) _____________________________________________________--________ = = 0. 213 g - 0. 21 g x 100 __________________________ 0. 21 g 1. 4 %

Molar Mass of any Gas (Hydrogen for example) • PV = n RT • n = g of gas/ MM gas [MM gas = g/mol] • PV = (g of gas/ MM gas)RT • MM gas = g of gas/V (RT/P) Density of gas • MM gas = g of gas/V (RT/P) • MM gas = density of gas (RT/P)

(Part II) Magnesium Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) Mole Calculations: • • • Stoichiometry Calculation Ideal Gas Law Calculations Comparison (% Error) Get equipment from stockroom and complete data acquisition for Part II. Have individual Report Forms checked before leaving lab today.


Handouts • Select a partner and get 2 handouts each, which replace Lab Manual pp. 53 -60. • Read the Background section.
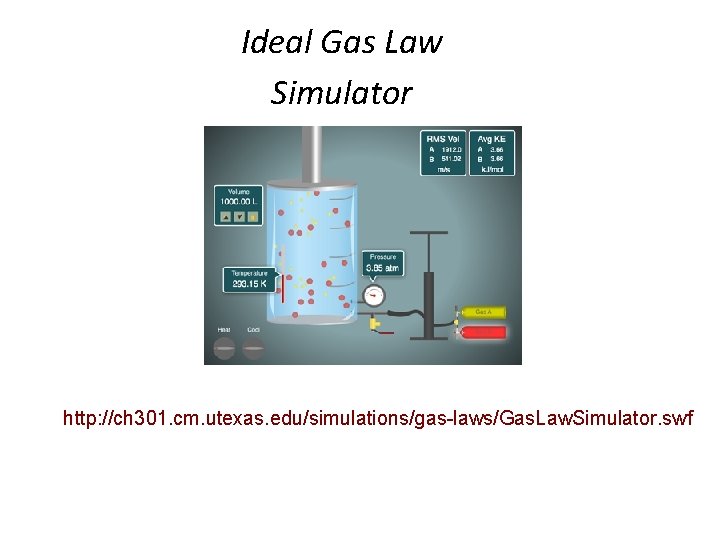
Ideal Gas Law Simulator http: //ch 301. cm. utexas. edu/simulations/gas-laws/Gas. Law. Simulator. swf

Gases & Airbags Use of Chemical Reactions and Physical Properties http: //chemconnections. org/general/movies/airbags. MOV


Real Gases Dr. Ron Rusay
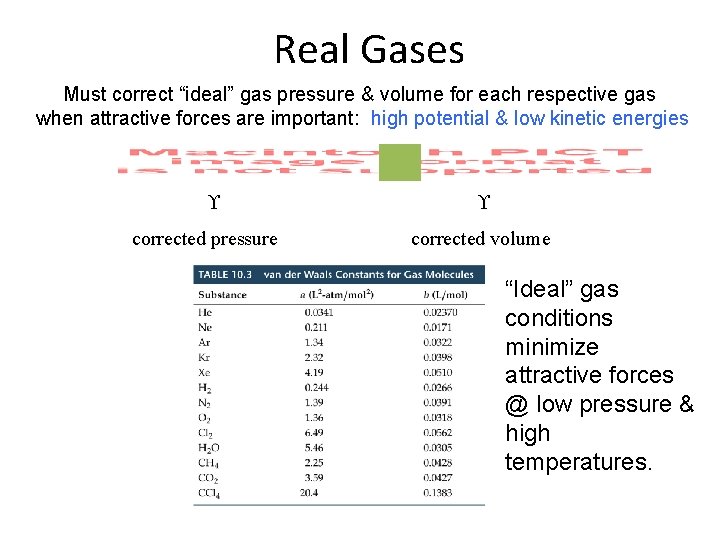
Real Gases Must correct “ideal” gas pressure & volume for each respective gas when attractive forces are important: high potential & low kinetic energies corrected pressure corrected volume “Ideal” gas conditions minimize attractive forces @ low pressure & high temperatures.

Real Gases Volume vs. Temperature @ constant P



Atmospheric Pollutants

QUESTION What time of day is it in LA? (There is no marine layer. ) A) 8: 00 AM B) 4: 00 PM

ANSWER What time of day is it in LA? A) 8: 00 AM B) 4: 00 PM
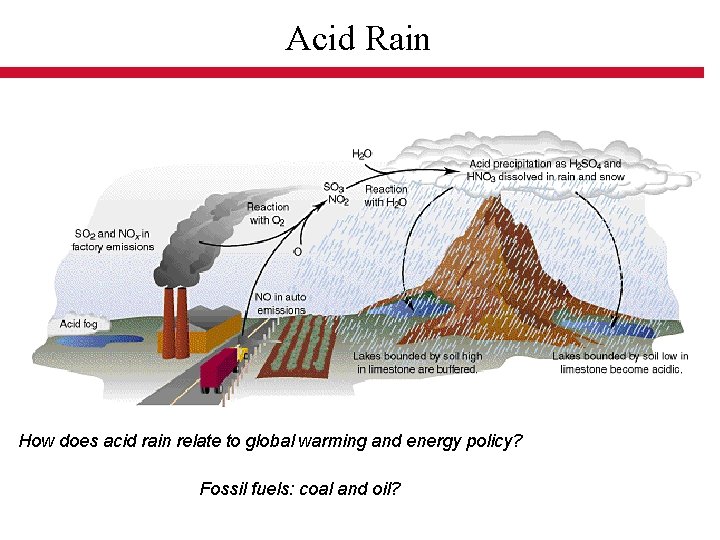
Acid Rain How does acid rain relate to global warming and energy policy? Fossil fuels: coal and oil?
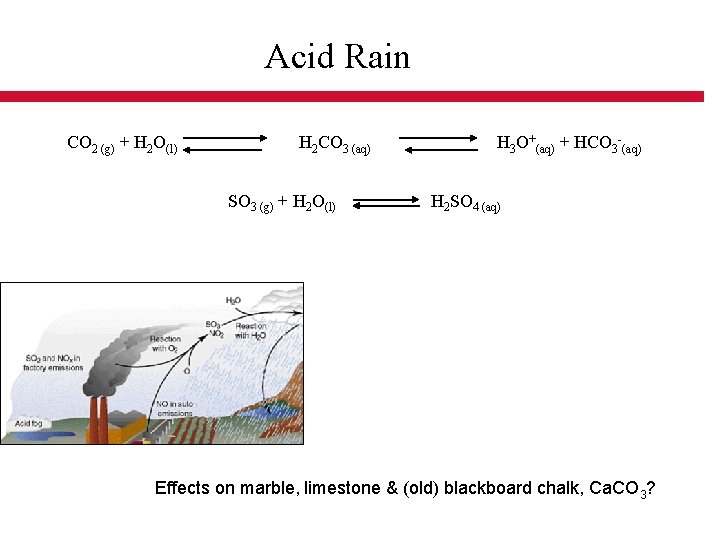
Acid Rain CO 2 (g) + H 2 O(l) H 2 CO 3 (aq) H 3 O+(aq) + HCO 3 -(aq) SO 3 (g) + H 2 O(l) H 2 SO 4 (aq) Effects on marble, limestone & (old) blackboard chalk, Ca. CO 3?

Atmospheric Pollutants Acid Rain Protection

Atmospheric Pollutants?
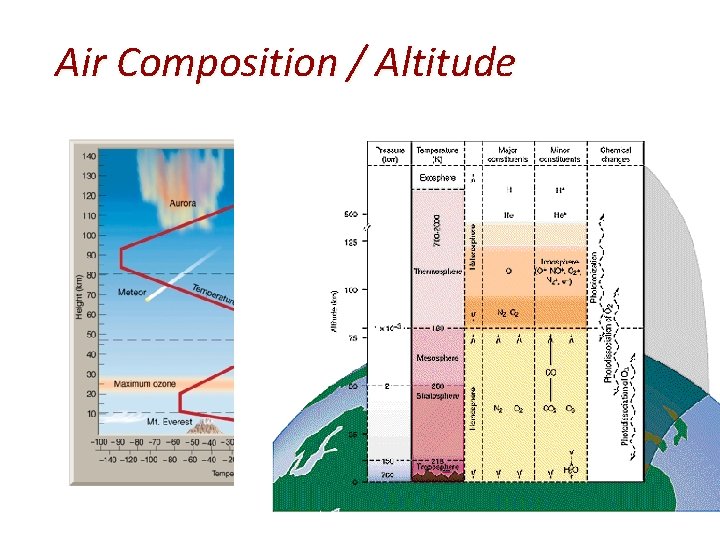
Air Composition / Altitude
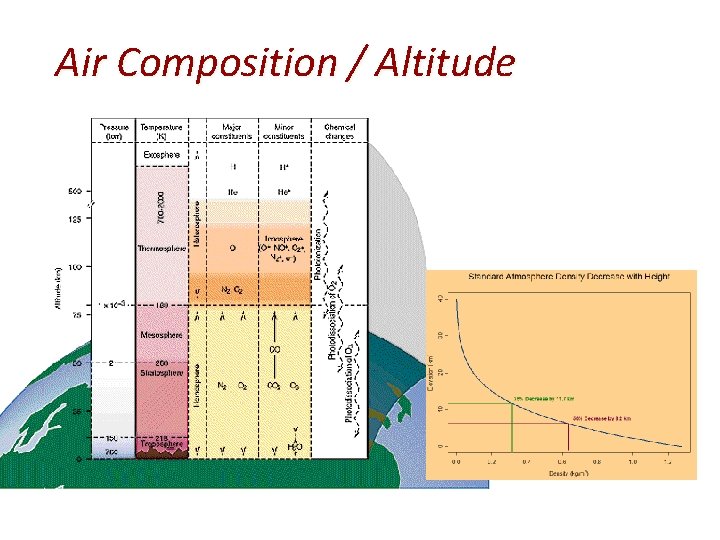
Air Composition / Altitude
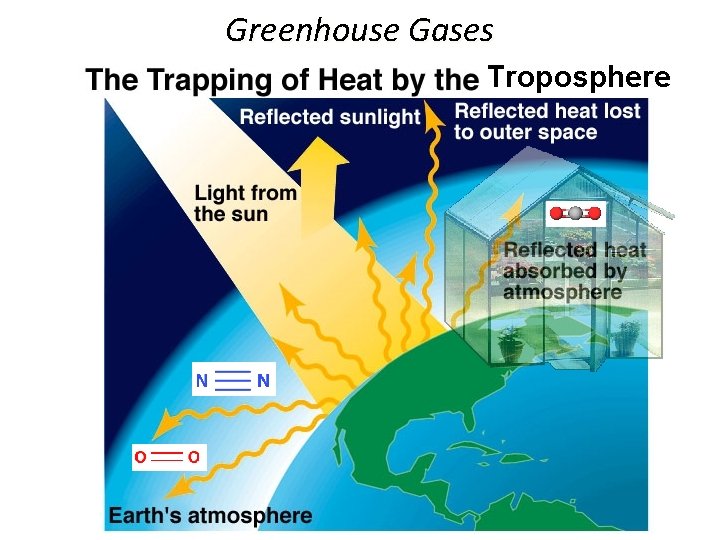
Greenhouse Gases Troposphere
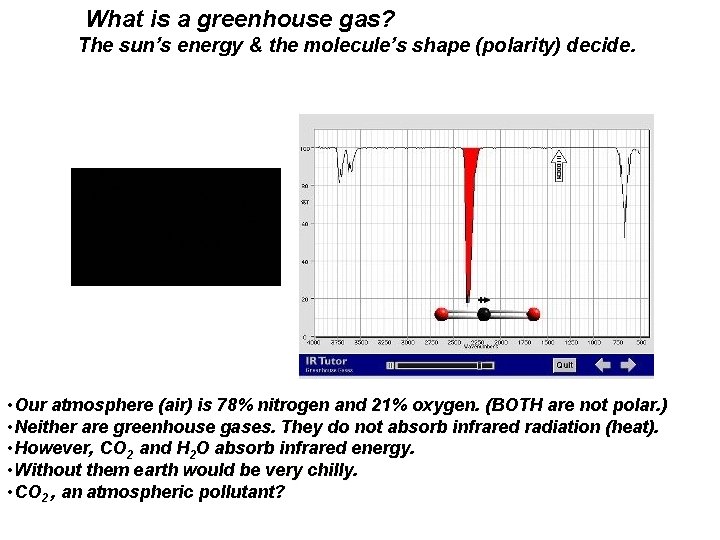
What is a greenhouse gas? The sun’s energy & the molecule’s shape (polarity) decide. • Our atmosphere (air) is 78% nitrogen and 21% oxygen. (BOTH are not polar. ) • Neither are greenhouse gases. They do not absorb infrared radiation (heat). • However, CO 2 and H 2 O absorb infrared energy. • Without them earth would be very chilly. • CO 2 , an atmospheric pollutant?

What is a greenhouse gas? The sun’s energy & a molecule’s shape & polarity decide. ……. and a pollutant? • Our atmosphere (air) is 78% nitrogen and 21% oxygen. (BOTH are not polar. ) • Neither are greenhouse gases. They do not absorb infrared radiation (heat). • However, H 2 O and CO 2 can absorb infrared energy. Without them earth would be very chilly. • ………. CO 2 , CH 4 atmospheric pollutants? • Governments decide. http: //zebu. uoregon. edu/1998/es 202/l 13. html

Energy (Carbon) (CO 2) Global Warming (Carbon) Economy (Jobs) https: //www. co 2. earth/ (CO 2)

Your Future?
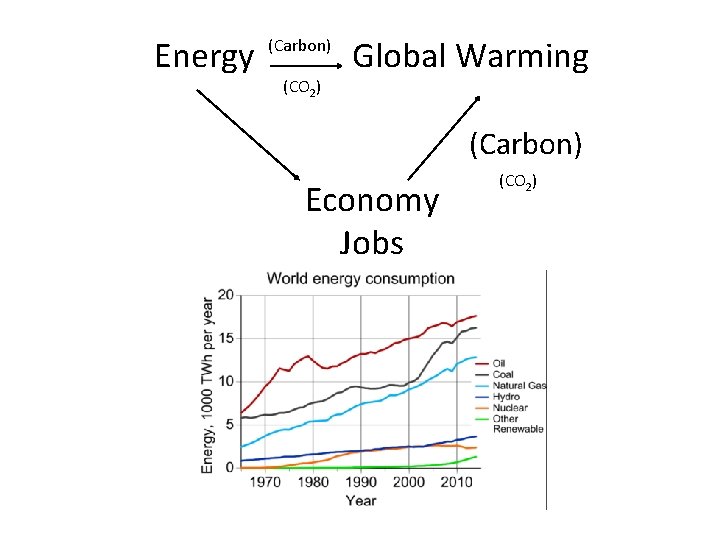
Energy (Carbon) (CO 2) Global Warming (Carbon) Economy Jobs (CO 2)

Energy (Carbon) (CO 2) Global Warming (Carbon) Economy Jobs https: //www. co 2. earth/ (CO 2)
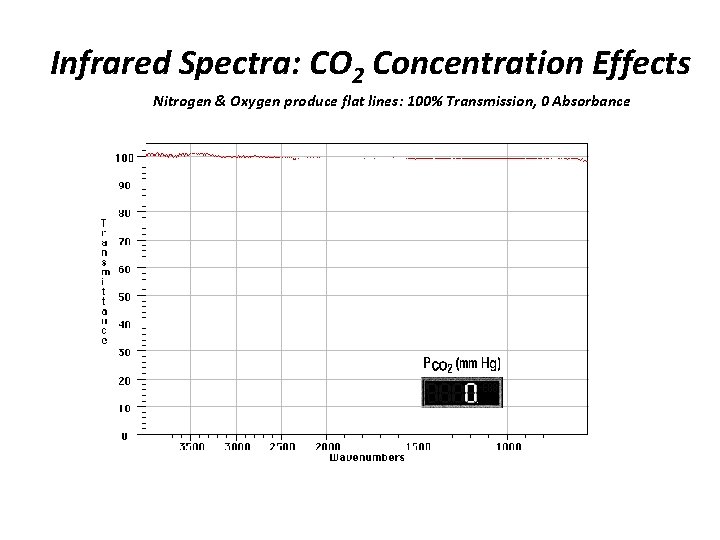
Infrared Spectra: CO 2 Concentration Effects Nitrogen & Oxygen produce flat lines: 100% Transmission, 0 Absorbance

Bonus Assignment http: //chemconnections. org/Global%20 Warming/Global%20 warming%20& %20 Carbon%20 Footprint. pdf Global Warming, Your Carbon Footprint & Your Future Capstone Writing Project http: //chemconnections. org/Global%20 Warming/

Bonus Assignment https: //chem. libretexts. org/Libre. Texts/Diablo_Valley_College/DVC_Chem_106%3 A_Rusay/ Vocabulary_III#global%25252 Bwarming Global Warming, Your Carbon Footprint & Your Future http: //chemconnections. org/Global%20 Warming/

Global Warming, Your Carbon Footprint & Your Future SEE: http: //chemconnections. org/IFTVET-2016/IFVET-Global%20 Warming. htm


Quiz Questions

QUESTION The density of an unknown atmospheric gas pollutant was experimentally determined to be 1. 964 g/ L @ 0 o. C and 760 torr. • What is the molar mass of the gas? • What might the gas be? A) CO B) SO 2 C) H 2 O D) CO 2

ANSWER 1. 964 g/ L @ 0 o. C and 760 torr. R = 0. 08206 L atm mol o. C K torr atm MM gas = density of gas (RT/P) MM gas = 1. 964 g/ L x 0. 08206 L atm mol x 273 K/ 760 torr x 760 torr/ 1 atm MM MM gas = 44. 0 g/mol gas= D) CO 2

QUESTION Freon-12, CF 2 Cl 2, a “safe” compressible gas, was widely used from 1935 -1994 as a refrigerant in refrigerators, freezers, and air conditioning systems. However, it had been shown to be a greenhouse gas and to catalytically destroy the ozone layer in a ratio of >14, 000: 1. It was phased out and banned. 200. ml of Freon-12 was collected by syringe. It weighed 0. 927 grams, had a temperature of 30. 0°C (303. 1 K), and a pressure of 730 mm of Hg (. What is the experimental molar mass of Freon-12? • • • 12. 1 g/mol 84 g/mol 92. 7 g/mol 115 g/mol 121. g/mol R = 0. 082 L atm K− 1 mol− 1

ANSWER E) 121 g/mol is the molar mass of Freon-12. MM gas = g of gas/V (RT/P)

QUESTION 0. 0820 grams of a volatile compound in the gas phase, which smells like fresh raspberries, was trapped in a syringe. It had a volume of 12. 2 m. L at 1. 00 atmosphere of pressure and 25. 0°C. What is the molar mass of this pleasant smelling compound ? A) B) C) D) 13. 8 g/mol 164 g/mol 40. 9 g/mol 224 g/mol

ANSWER B) 164 g/mol Using PV = n. RT : 0. 0122 L for V, 298 K for T, 0. 08206 for R and solving for n = 1. MOLAR MASS (grams in one mole) can be calculated. MM gas = density of gas (RT/P) MM gas = 0. 0820 g/ L x 0. 00122 L x 0. 0821 atm mol x 298 K/ 1 atm
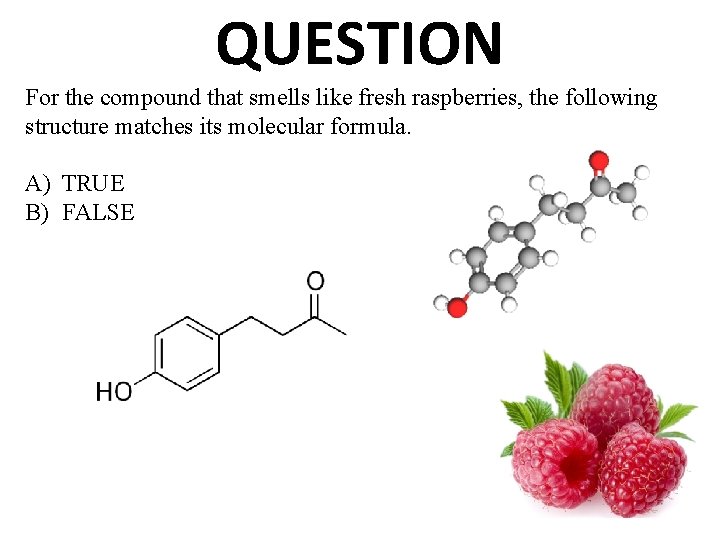
QUESTION For the compound that smells like fresh raspberries, the following structure matches its molecular formula. A) TRUE B) FALSE
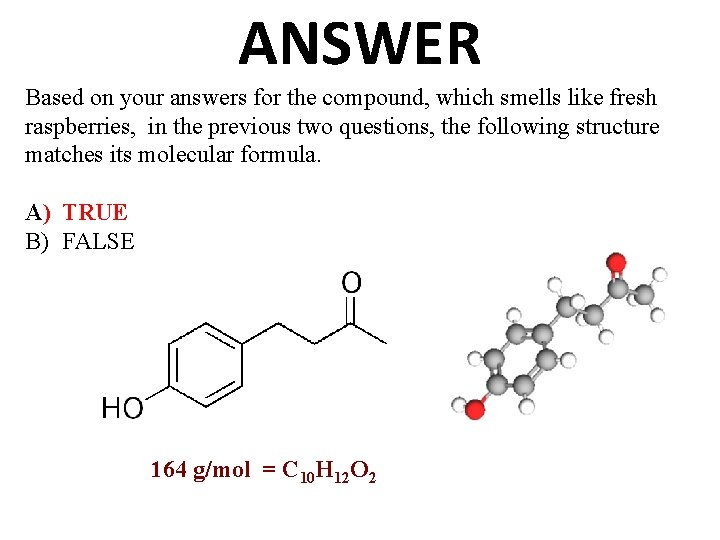
ANSWER Based on your answers for the compound, which smells like fresh raspberries, in the previous two questions, the following structure matches its molecular formula. A) TRUE B) FALSE 164 g/mol = C 10 H 12 O 2

QUESTION Which sequence represents the gases in order of increasing density at STP? A) Fluorine < Carbon monoxide < Chlorine < Argon B) Carbon monoxide < Fluorine < Argon < Chlorine C) Argon < Carbon monoxide < Chlorine < Fluorine D) Fluorine < Chlorine < Carbon monoxide < Argon

ANSWER Which sequence represents the gases in order of increasing density at STP? A) Fluorine < Carbon monoxide < Chlorine < Argon B) Carbon monoxide < Fluorine < Argon < Chlorine C) Argon < Carbon monoxide < Chlorine < Fluorine D) Fluorine < Chlorine < Carbon monoxide < Argon

QUESTION Real gases exhibit their most “ideal” behavior at which relative conditions? A) B) C) D) Low temperatures and low pressures High temperatures and high pressures High temperatures and low pressures Low temperatures and high pressures

ANSWER C) High temperatures and low pressures At these conditions gas molecules are farthest apart and exert their least influence on each other (High kinetic energy and low potential energy) thereby permitting their behavior to more closely follow mathematical formulas, “laws’.


% Mg: Calculations Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2 (g) Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2 (g) total moles H 2(g) = moles Mg(s) + moles Zn(s) mass (g) Zn(s) = mass sample (g) – ? mass Mg(s) (g) (mass sample (g) – ? mass Mg(s) ( ___________________ total moles H 2(g) = + Molar Mass Mg(s) Molar Mass Zn(s) Solve ? grams Mg(s) % Mg(s)


Related Quiz Questions Experimentally Determining Moles of Hydrogen

QUESTION The density of an unknown atmospheric gas pollutant was experimentally determined to be 1. 964 g/ L @ 0 o. C and 760 torr. • What is the molar mass of the gas? • What might the gas be? A) CO B) SO 2 C) H 2 O D) CO 2

QUESTION Freon-12, CF 2 Cl 2, a “safe” compressible gas, was widely used from 1935 -1994 as a refrigerant in refrigerators, freezers, and air conditioning systems. However, it had been shown to be a greenhouse gas and to catalytically destroy the ozone layer in a ratio of >14, 000: 1. It was phased out and banned. 200. ml of Freon-12 was collected by syringe. It weighed 0. 927 grams, had a temperature of 30. 0°C (303. 1 K), and a pressure of 730 mm of Hg (. What is the experimental molar mass of Freon-12? • • • 12. 1 g/mol 84 g/mol 92. 7 g/mol 115 g/mol 121. g/mol R = 0. 082 L atm K− 1 mol− 1

QUESTION 0. 0820 grams of a volatile compound in the gas phase, which smells like fresh raspberries, was trapped in a syringe. It had a volume of 12. 2 m. L at 1. 00 atmosphere of pressure and 25. 0°C. What is the molar mass of this pleasant smelling compound ? A) B) C) D) 13. 8 g/mol 164 g/mol 40. 9 g/mol 224 g/mol
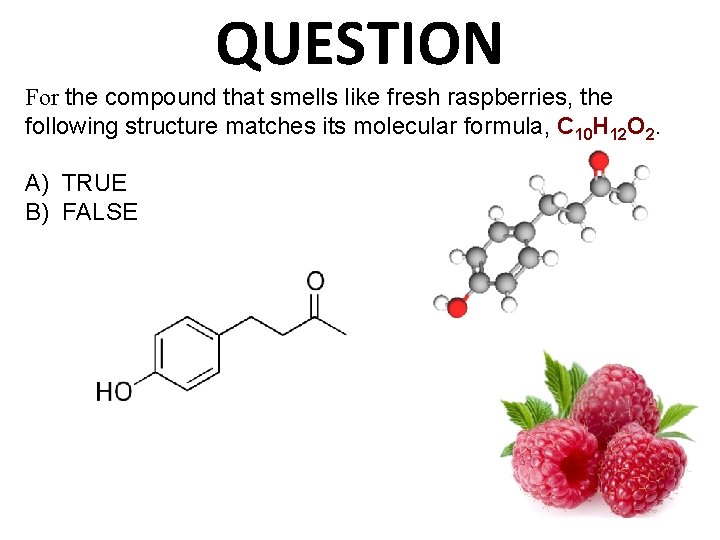
QUESTION For the compound that smells like fresh raspberries, the following structure matches its molecular formula, C 10 H 12 O 2. A) TRUE B) FALSE

QUESTION Which sequence represents the gases in order of increasing density at STP? A) Fluorine < Carbon monoxide < Chlorine < Argon B) Carbon monoxide < Fluorine < Argon < Chlorine C) Argon < Carbon monoxide < Chlorine < Fluorine D) Fluorine < Chlorine < Carbon monoxide < Argon

QUESTION Real gases exhibit their most “ideal” behavior at which relative conditions? A) B) C) D) Low temperatures and low pressures High temperatures and high pressures High temperatures and low pressures Low temperatures and high pressures
- Slides: 95