Chem 108 Lab Week 13 Sign in Pick

Chem 108: Lab Week 13 Sign in / Pick up Papers Turn in Global Warming Questions Scantron

Chem 108: Lab Standardization completed last week Individual Titrations of Unknown To Do Today

Neutralization Reactions H+(aq) + OH -(aq) H 2 O(l) There is no difference in the stoichiometry of the reaction of acetic acid & HCl with Na. OH, but there is a difference in p. H.

Methods for Measuring the p. H of an Aqueous Solution (a) p. H paper (b) Electrodes of a p. H meter

Neutralizations / Titrations Chem 108 titration: phenolphthalein indicator Chem 120/121 Titration Curves

Aqueous Reactions: Neutralization p. H @ the end point http: //chemconnections. org/general/movies/p. Hof. Salt. Solutions. swf HCl(aq) + Na. OH (aq) acid base salt CH 3 COOH(aq) + Na. OH (aq) acid Na. Cl (aq) + H 2 O(l) base p. H = 7 @ end pt. water CH 3 COONa (aq) + H 2 O(l) p. H > 7 @ end pt. salt water
http: //chemconnections. org/general/chem 108/Acids. Bases%20 Guide. html
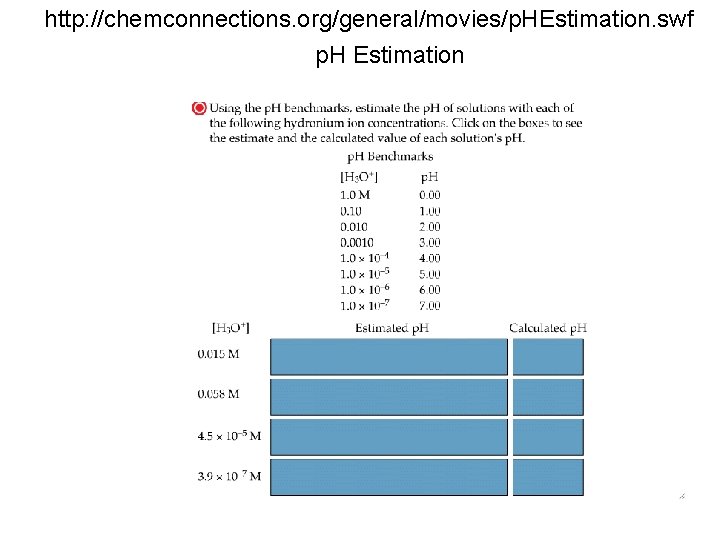
http: //chemconnections. org/general/movies/p. HEstimation. swf p. H Estimation
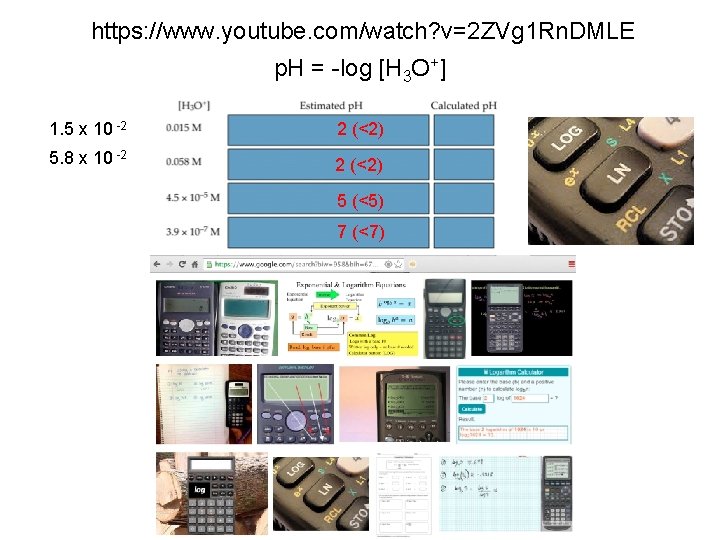
https: //www. youtube. com/watch? v=2 ZVg 1 Rn. DMLE p. H = -log [H 3 O+] 1. 5 x 10 -2 2 (<2) 5. 8 x 10 -2 2 (<2) 5 (<5) 7 (<7)

0. 015 0. 058 1. 5 x 1. 8 10 -2 5. 8 x 10 -2 log [H 3 O+] log (0. 015 -1. 823908741 -log [H 3 O+] p. H = 1. 8 log [H 3 O+] log (1. 5 E-2 -1. 823908741 -log [H 3 O+] p. H = 1. 8

EQUILIBRIUM CO 2 & Oceanic Bicarbonate Buffering CO 2(g) + H 2 O (l) HCO 3 -1(aq) + H+1(aq) CO 3 -2(aq) + H+1(aq) Oceans: p. H ~ 8. 1 and falling http: //www. tos. org/oceanography/issues/issue_archive/22_4. html Increasing CO 2 is decreasing ocean p. H; long term effects? http: //sos. noaa. gov/datasets/Ocean/ocean_acidification. html
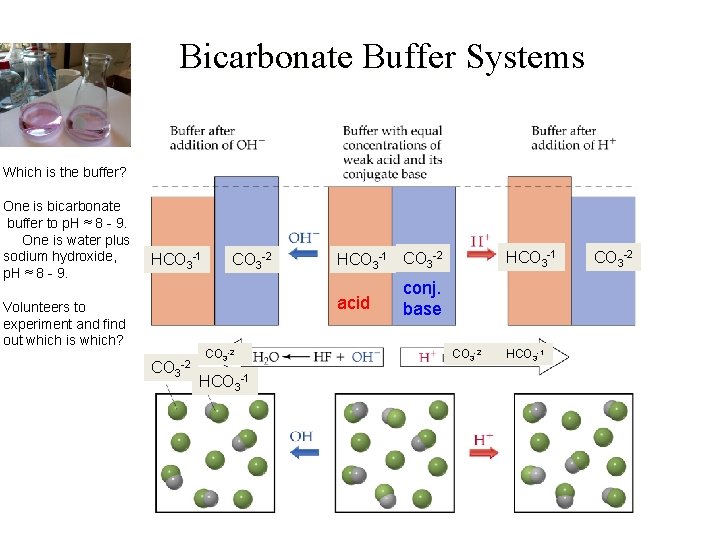
Bicarbonate Buffer Systems Which is the buffer? One is bicarbonate buffer to p. H ≈ 8 - 9. One is water plus sodium hydroxide, p. H ≈ 8 - 9. HCO 3 -1 CO 3 -2 acid Volunteers to experiment and find out which is which? CO 3 -2 HCO 3 -1 CO 3 -2 conj. base CO 3 -2 HCO 3 -1 CO 3 -2

Chem 108: Lab Part 1 Done with a partner. Completed last week.

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water H 2 O(l) 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. MKOH = ?

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt water 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. ? MKOH = = [MHNO 3 x VHNO 3 / VKOH ] [? mol. KOH / ? mol. HNO 3] x 0. 02500 LHNO 3 x 1 mol. KOH = 0. 2231 MKOH LHNO 3 x 0. 02420 LKOH x 1 mol. HNO 3 0. 2160 mol. HNO 3

QUESTION A 35. 00 m. L sample of 0. 2250 M HBr was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2719 M 0. 1860 M 0. 3720 M
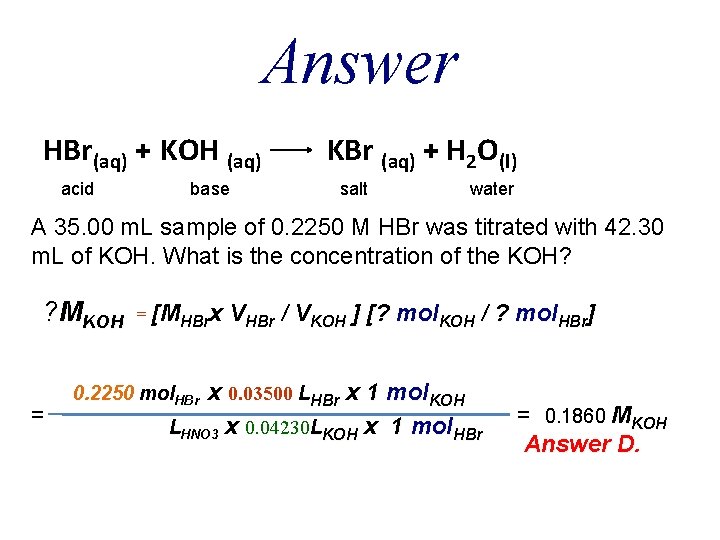
Answer HBr(aq) + KOH (aq) acid base KBr (aq) + H 2 O(l) salt water A 35. 00 m. L sample of 0. 2250 M HBr was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? ? MKOH = = [MHBrx VHBr / VKOH ] [? mol. KOH / ? mol. HBr] x 0. 03500 LHBr x 1 mol. KOH LHNO 3 x 0. 04230 LKOH x 1 mol. HBr 0. 2250 mol. HBr = 0. 1860 MKOH Answer D.

QUESTION A 35. 00 m. L sample of 0. 2250 M H 2 SO 4 was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2719 M 0. 1860 M 0. 3720 M

Answer H 2 SO 4(aq) + 2 KOH (aq) acid base K 2 SO 4 (aq) + 2 H 2 O(l) salt water A 35. 00 m. L sample of 0. 2250 M H 2 SO 4 was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? ? MKOH = = [MH 2 SO 4 x VH 2 SO 4 / VKOH ] [? mol. KOH / ? mol. H 2 SO 4] x 0. 03500 LH 2 SO 4 x 2 mol. KOH = 0. 3720 MKOH LH 2 SO 4 x 0. 04230 LKOH x 1 mol. H 2 SO 4 Answer E. 0. 2250 mol. H 2 SO 4
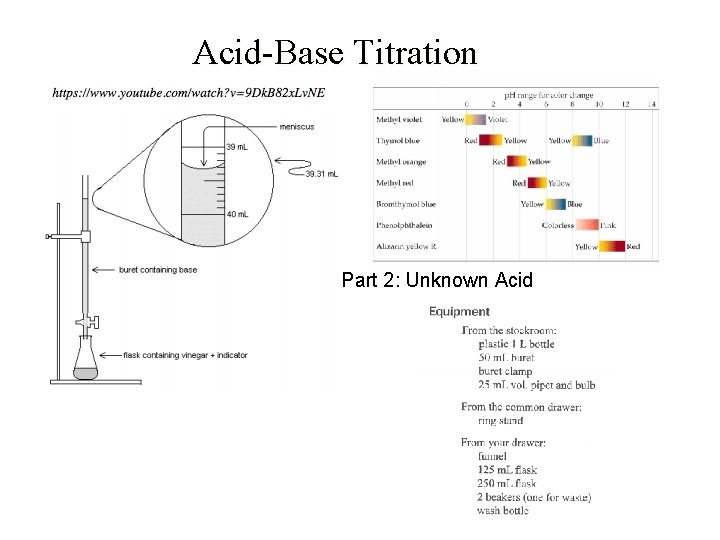
Acid-Base Titration Part 2: Unknown Acid

Chem 108: Lab Part 2: Week 13 To Do (individually) today Take a clean, dry, 125 m. L erlenmeyer flask to the stockroom window and get unknown acid solution. Record unknown number. Have data page signed before leaving lab

QUESTION A 35. 00 m. L sample of hydrochloric acid of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the HCl? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2719 M 0. 1860 M 0. 3720 M
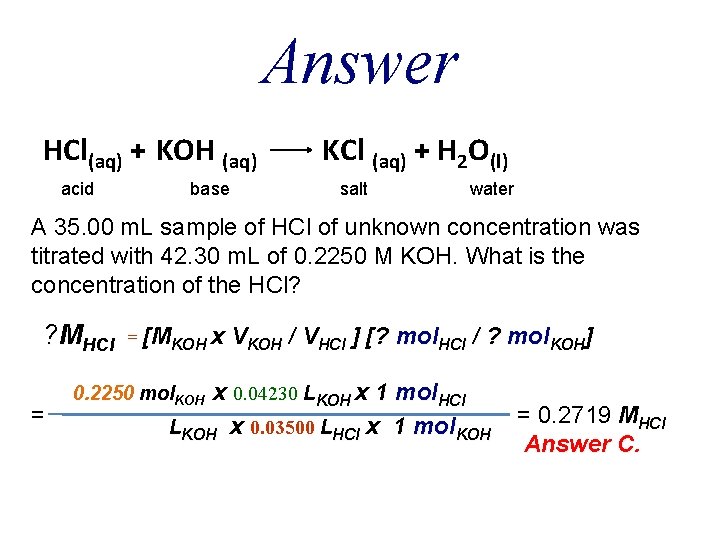
Answer HCl(aq) + KOH (aq) acid base KCl (aq) + H 2 O(l) salt water A 35. 00 m. L sample of HCl of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the HCl? ? MHCl = = [MKOH x VKOH / VHCl ] [? mol. HCl / ? mol. KOH] x 0. 04230 LKOH x 1 mol. HCl LKOH x 0. 03500 LHCl x 1 mol. KOH 0. 2250 mol. KOH = 0. 2719 MHCl Answer C.


Answer: A Chem 108: Class/ Lab A 35. 0 m. L sample of 0. 225 M HBr was titrated with 42. 3 m. L of KOH. What is the concentration of the KOH? A) 0. 157 M B) 0. 303 M C) 0. 272 M D) 0. 186 M E) none of the above Answer: D Week 12 Exactly 17. 0 m. L of a H 2 SO 4 solution was required to neutralize 45. 0 m. L of 0. 235 Na. OH. What was the concentration of the H 2 SO 4 solution? Given: H 2 SO 4 (aq) + 2 Na. OH (aq) → 2 H 2 O (l) + Na 2 SO 4 (aq) A) 5. 63 M B) 0. 622 M C) 0. 00529 M D) 0. 311 M
- Slides: 25