Chem 108 Lab Week 12 Sign in Pick

Chem 108: Lab Week 12 Sign in / Pick up Papers Pick up Global Warming Handout Questions plus Scantron (Due next week)

Chem 108: Class/ Lab Week 12 1) Fluid Exchange (Handout) Due Today 2) Acid-Base Equilibrium Experiment (Handout) Due Today 3) Any missing assignments Due Today Pick up Global Warming Handout Questions plus Scantron (Due next week)
Did you have a Happy Halloween, “All Hallows Eve”? Do you believe in Vampires & their thirst for blood. . . . ?

TODAY: The day after All Hallows Eve Día de los Muertos / Day of the Dead

Blood Light & The Chemistry of Vision Color(s) of Blood & Vampires
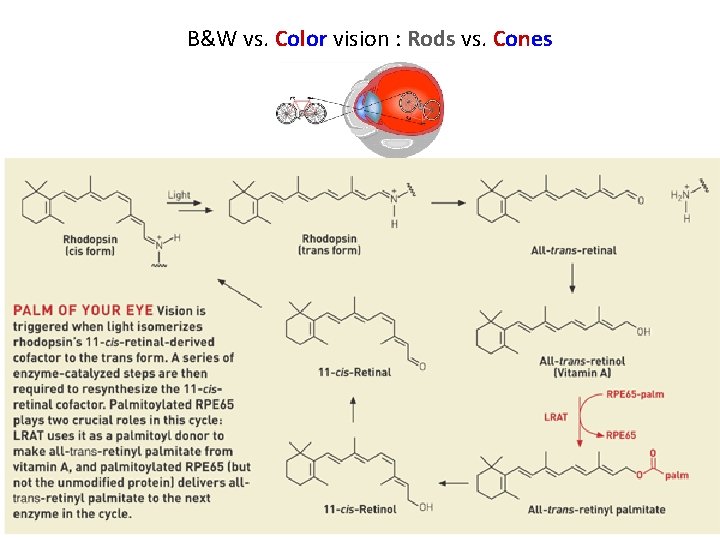
B&W vs. Color vision : Rods vs. Cones
B&W vs. Color vision : Rods vs. Cones Color Vision Experiment/Exercise
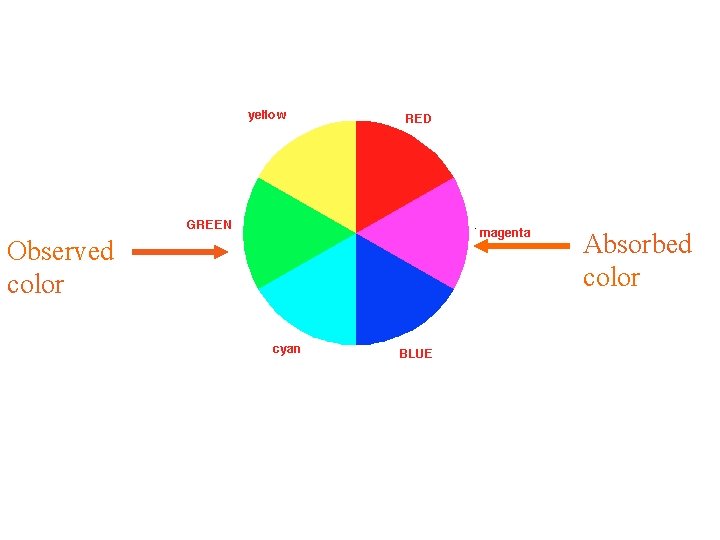
Observed color Absorbed color
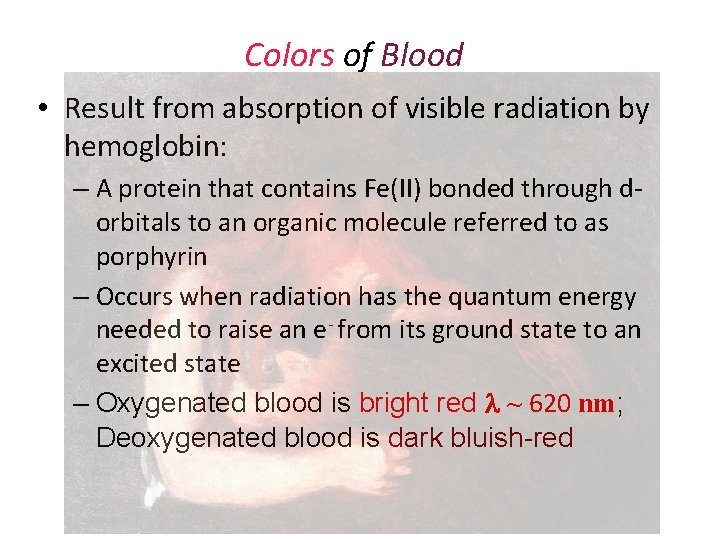
Colors of Blood • Result from absorption of visible radiation by hemoglobin: – A protein that contains Fe(II) bonded through dorbitals to an organic molecule referred to as porphyrin – Occurs when radiation has the quantum energy needed to raise an e- from its ground state to an excited state – Oxygenated blood is bright red ~ 620 nm; Deoxygenated blood is dark bluish-red
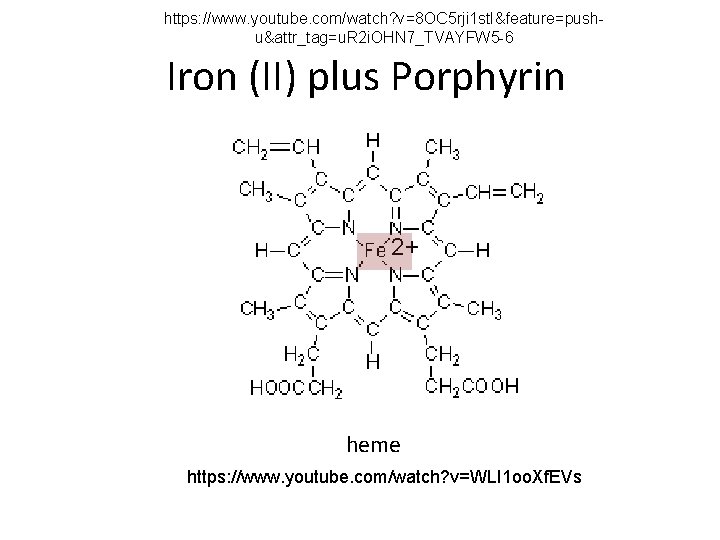
https: //www. youtube. com/watch? v=8 OC 5 rji 1 st. I&feature=pushu&attr_tag=u. R 2 i. OHN 7_TVAYFW 5 -6 Iron (II) plus Porphyrin 2+ heme https: //www. youtube. com/watch? v=WLI 1 oo. Xf. EVs
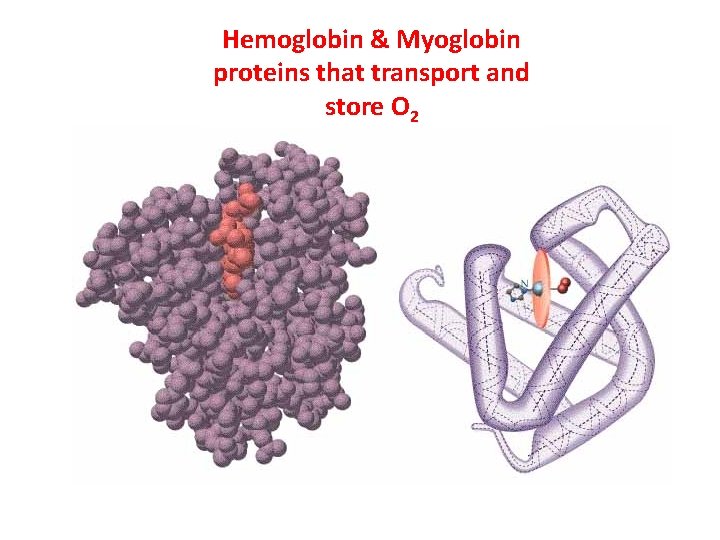
Hemoglobin & Myoglobin proteins that transport and store O 2
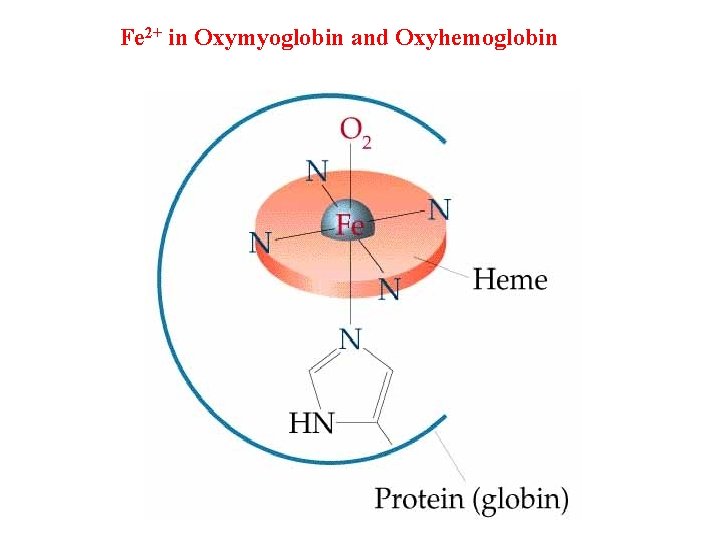
Fe 2+ in Oxymyoglobin and Oxyhemoglobin
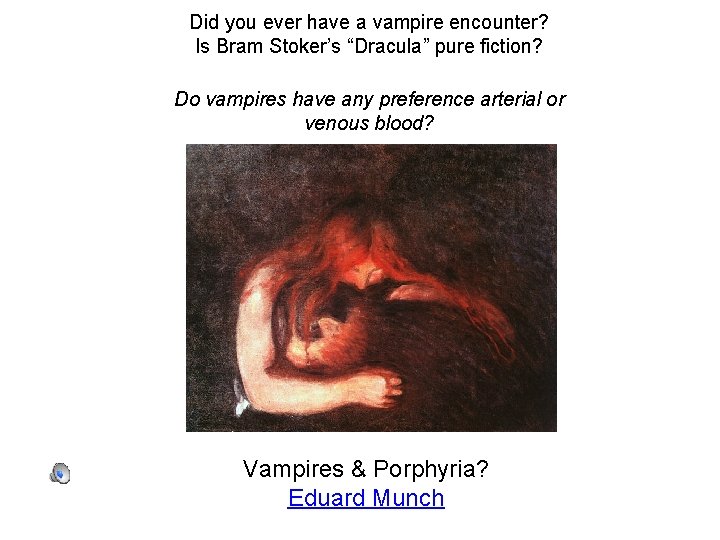
Did you ever have a vampire encounter? Is Bram Stoker’s “Dracula” pure fiction? Do vampires have any preference arterial or venous blood? Vampires & Porphyria? Eduard Munch

Porphyria Erythropoietic porphyria symptoms: severe photosensitivity, swelling, pain, and burning; requires avoidance of bright sunlight; produces brown canine teeth that fluoresce in ultraviolet light; produces hemolytic anemia. Drinking large quantities of blood can alleviate the symptoms. Garlic activates an enzyme that destroys them faster and makes the symptoms of porphyria worse. http: //en. wikipedia. org/wiki/Porphyria Q. "Why is a baby like a vampire? A "Because it sleeps all day and feeds all night. "

Edvard Munch (1863– 1944) SF MOMA July 1 -October 5, 2017 Eduard Munch

Chem 108: Exam 2: Divide raw score by 117 (normalized to 100%) Raw point average = 76. 6 +/- 23. 6 Exam 1 Raw point average = 76. 9 +/- 15. 9 Complete Grades will be posted before Monday’s class with an October 30 link on the calendar

Chem 108: Lab Week 12 To Do & DUE Today Have signed before leaving lab.

Neutralization Reactions

Neutralizations / Titrations Chem 108 titration: phenolphthalein indicator Chem 120/121 Titration Curves
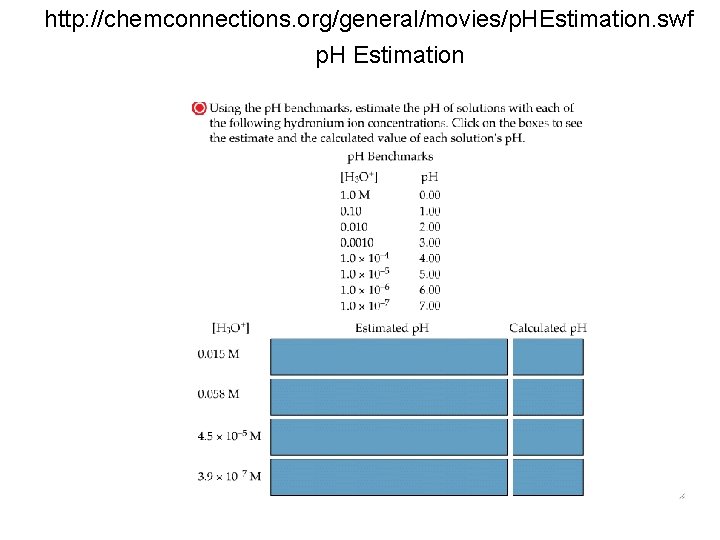
http: //chemconnections. org/general/movies/p. HEstimation. swf p. H Estimation
Due Today http: //chemconnections. org/general/chem 108/Acids. Bases%20 Guide. html

Neutralization Reactions Would there be a difference in the reaction of acetic acid versus HCl?
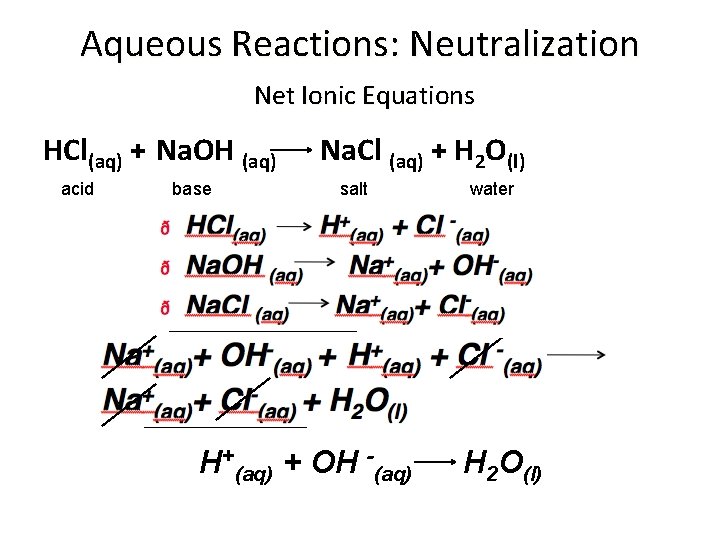
Aqueous Reactions: Neutralization Net Ionic Equations HCl(aq) + Na. OH (aq) acid base Na. Cl (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water H 2 O(l)

Aqueous Reactions: Neutralization Net Ionic Equations CH 3 COOH(aq) + Na. OH (aq) CH 3 COONa (aq) + H 2 O(l) acid base salt water CH 3 COO-(aq) + H+(aq) + Na+(aq) + OH- (aq) CH 3 COO-(aq) + Na+(aq) + H 2 O(l) H+(aq) + OH -(aq) H 2 O(l) Same Net Ionic Equation (NIE) for any neutralization Eg. H 2 SO 4 2 H+(aq) + 2 OH -(aq) 2 H 2 O(l) _____________ 2
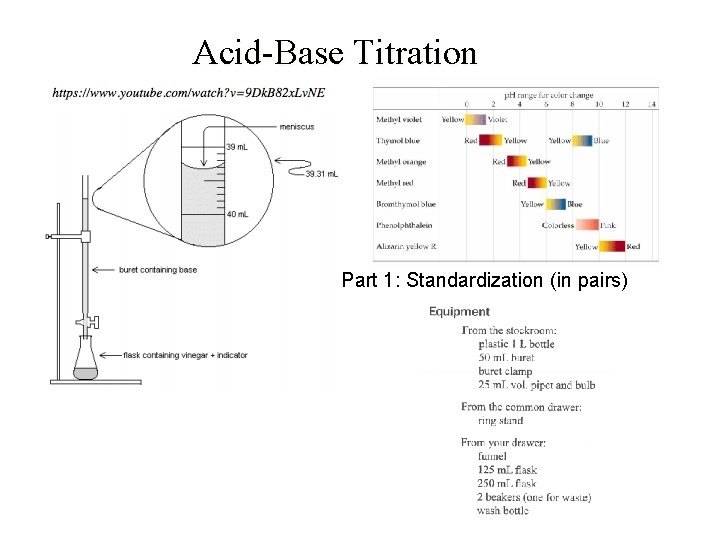
Acid-Base Titration Part 1: Standardization (in pairs)

Chem 108: Lab Week 12 To Do (with a partner) Have signed before leaving lab.

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water H 2 O(l) 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. MKOH = ?

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt water 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. ? MKOH = = [MHNO 3 x VHNO 3 / VKOH ] [? mol. KOH / ? mol. HNO 3] x 0. 02500 LHNO 3 x 1 mol. KOH = 0. 2231 MKOH LHNO 3 x 0. 02420 LKOH x 1 mol. HNO 3 0. 2160 mol. HNO 3

QUESTION A 35. 00 m. L sample of 0. 2250 M HBr was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2720 M 0. 1860 M 0. 3720 M
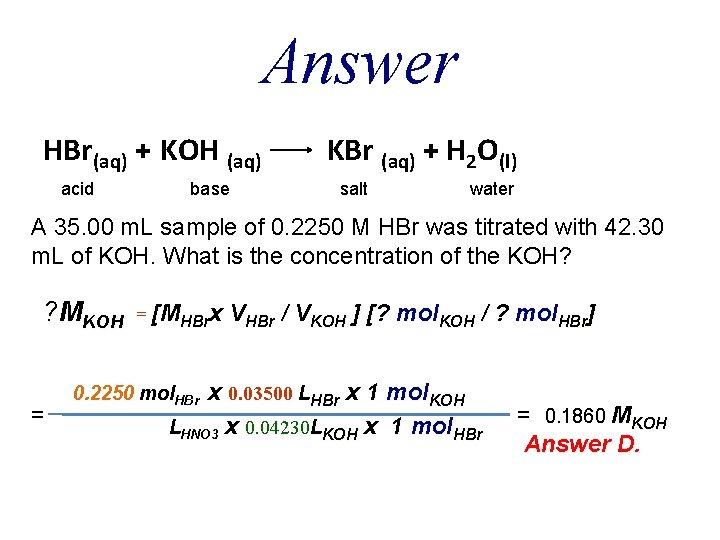
Answer HBr(aq) + KOH (aq) acid base KBr (aq) + H 2 O(l) salt water A 35. 00 m. L sample of 0. 2250 M HBr was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? ? MKOH = = [MHBrx VHBr / VKOH ] [? mol. KOH / ? mol. HBr] x 0. 03500 LHBr x 1 mol. KOH LHNO 3 x 0. 04230 LKOH x 1 mol. HBr 0. 2250 mol. HBr = 0. 1860 MKOH Answer D.

QUESTION A 35. 00 m. L sample of 0. 2250 M H 2 SO 4 was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2720 M 0. 1860 M 0. 3720 M

Answer H 2 SO 4(aq) + 2 KOH (aq) acid base K 2 SO 4 (aq) + 2 H 2 O(l) salt water A 35. 00 m. L sample of 0. 2250 M H 2 SO 4 was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? ? MKOH = = [MH 2 SO 4 x VH 2 SO 4 / VKOH ] [? mol. KOH / ? mol. H 2 SO 4] x 0. 03500 LH 2 SO 4 x 2 mol. KOH = 0. 3720 MKOH LH 2 SO 4 x 0. 04230 LKOH x 1 mol. H 2 SO 4 Answer E. 0. 2250 mol. H 2 SO 4
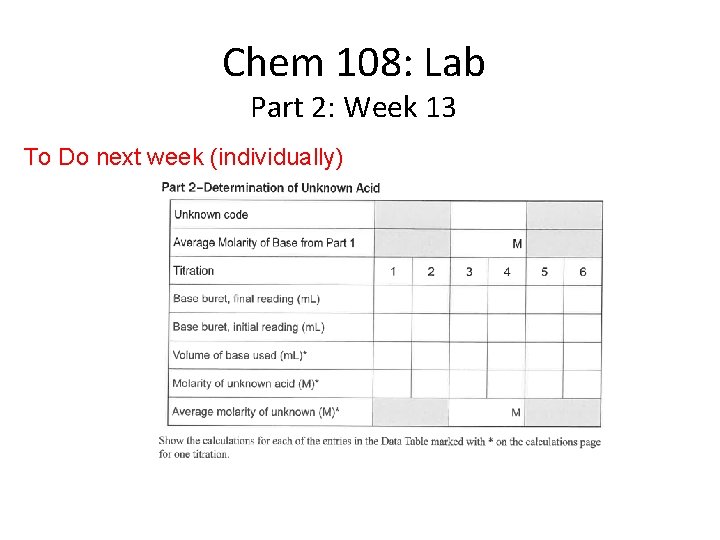
Chem 108: Lab Part 2: Week 13 To Do next week (individually)


Answer: A Chem 108: Class/ Lab A 35. 0 m. L sample of 0. 225 M HBr was titrated with 42. 3 m. L of KOH. What is the concentration of the KOH? A) 0. 157 M B) 0. 303 M C) 0. 272 M D) 0. 186 M E) none of the above Answer: D Week 12 Exactly 17. 0 m. L of a H 2 SO 4 solution was required to neutralize 45. 0 m. L of 0. 235 Na. OH. What was the concentration of the H 2 SO 4 solution? Given: H 2 SO 4 (aq) + 2 Na. OH (aq) → 2 H 2 O (l) + Na 2 SO 4 (aq) A) 5. 63 M B) 0. 622 M C) 0. 00529 M D) 0. 311 M
- Slides: 35