Chem 108 Lab Week 12 Chem 108 Class

Chem 108: Lab Week 12

Chem 108: Class/ Lab Week 12 1) Fluid Exchange (Handout) Due Today 2) Acid-Base Equilibrium Experiment (Handout) Due Today 3) Any missing assignments Due Today

Chem 108: Lab Week 12 To Do & DUE Today Have signed before leaving lab.

Neutralization Reactions

Neutralizations / Titrations Chem 108 titration: phenolphthalein indicator Chem 120/121 Titration Curves

Neutralization Reactions Would there be a difference in the reaction of acetic acid versus HCl?
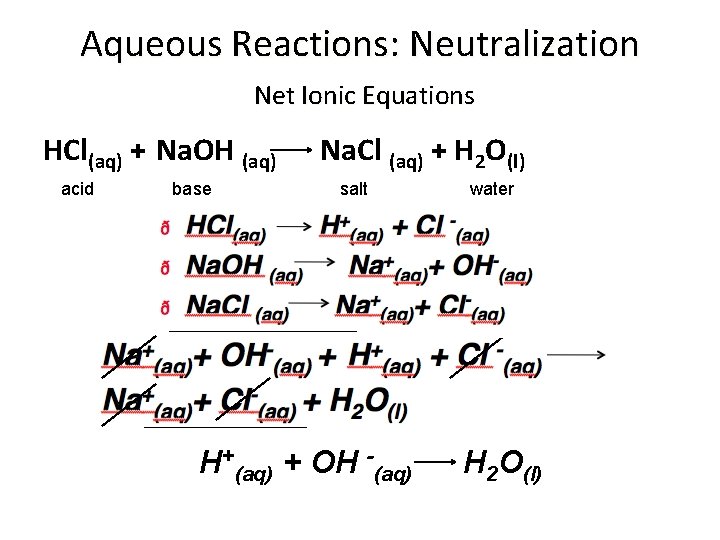
Aqueous Reactions: Neutralization Net Ionic Equations HCl(aq) + Na. OH (aq) acid base Na. Cl (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water H 2 O(l)

Aqueous Reactions: Neutralization Net Ionic Equations CH 3 COOH(aq) + Na. OH (aq) CH 3 COONa (aq) + H 2 O(l) acid base salt water CH 3 COO-(aq) + H+(aq) + Na+(aq) + OH- (aq) CH 3 COO-(aq) + Na+(aq) + H 2 O(l) H+(aq) + OH -(aq) H 2 O(l) Same Net Ionic Equation (NIE) for any neutralization Eg. H 2 SO 4 2 H+(aq) + 2 OH -(aq) 2 H 2 O(l) _____________ 2
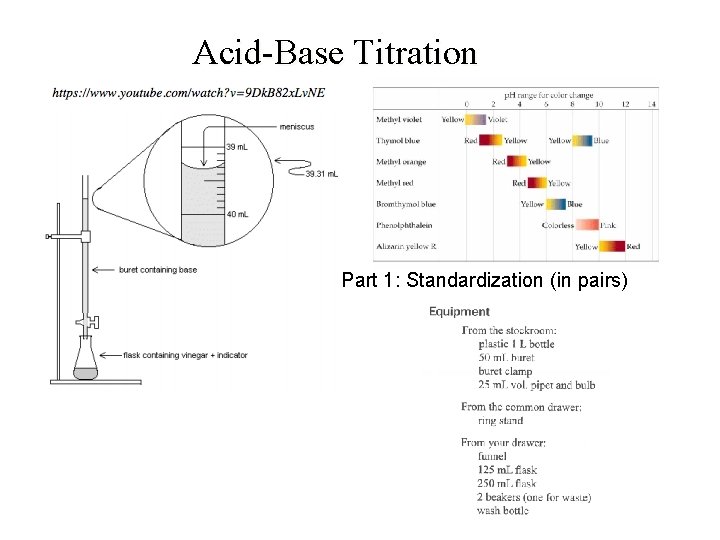
Acid-Base Titration Part 1: Standardization (in pairs)

Chem 108: Lab Week 12 To Do (with a partner) Have signed before leaving lab.

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water H 2 O(l) 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. MKOH = ?

Aqueous Reactions: Neutralization Net Ionic Equations HNO 3(aq) + KOH (aq) acid base KNO 3 (aq) + H 2 O(l) salt water 25. 00 m. L of MHCL = 0. 2160 M nitric acid solution was titrated with a potassium hydroxide solution. It required 24. 20 m. L as an average of three trials to reach a faint pink color. ? MKOH = = [MHNO 3 x VHNO 3 / VKOH ] [? mol. KOH / ? mol. HNO 3] x 0. 02500 LHNO 3 x 1 mol. KOH = 0. 2231 MKOH LHNO 3 x 0. 02420 LKOH x 1 mol. HNO 3 0. 2160 mol. HNO 3

QUESTION A 35. 00 m. L sample of 0. 2250 M HBr was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2720 M 0. 1860 M 0. 3720 M

QUESTION A 35. 00 m. L sample of 0. 2250 M H 2 SO 4 was titrated with 42. 30 m. L of KOH. What is the concentration of the KOH? A. B. C. D. E. 0. 0930 M 0. 3030 M 0. 2720 M 0. 1860 M 0. 3720 M
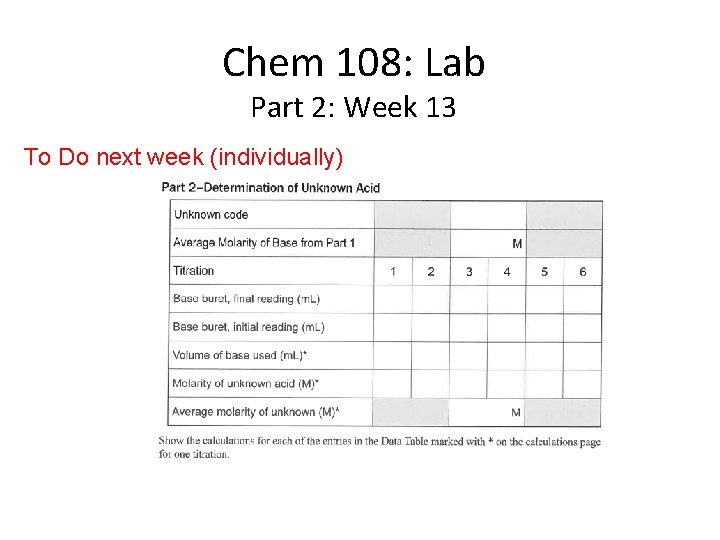
Chem 108: Lab Part 2: Week 13 To Do next week (individually)
- Slides: 15