Chem 108 Lab Sign in Roster front of

Chem 108: Lab Sign in: Roster @ front of lab Pick up handouts & a combination lock Go to your Lab Drawer
Lab Drawer Equipment Check Out Take out your Lab Manual

Lab Drawer Equipment Check Out Write down the combination of the lock for your records. Everyone must separately provide all of the information in each of the following 3 forms, sign and turn them in before today’s lab experiment can begin. Keep the lock’s combination where you can find it; bring to lab next week Read carefully & sign Check that all of the equipment on the list is in the drawer and unbroken.

Check Out On the pink form note what is missing or broken. If everything is OK, sign form, note combination for your use, turn in with completed/ signed acknowledgment form and card to Dr. R. If anything is missing or broken, take the pink form to the Chemistry Stockroom. Directions on next slide.

Missing or Broken Equipment Go to stockroom window to replace missing or broken equipment. Stockroom window is in the middle of this building. Turn right out of lab, walk past 2 lab rooms and look for the double glass doors. Window is in the back. When everything is OK, sign form, note combination for your use, turn in with completed/ signed acknowledgment form and card to Dr. R. Turn in: completed safety handout Double doors with window & counter set back X Stockroom Window You are here X

http: //chemconnections. org/general/chem 108/calendar-108 -s 20. html Monday CHEM 108 Wednesday

http: //chemconnections. org/general/chem 108/calendar-108 -s 20. html Monday CHEM 108 Wednesday

Reading (Handout) http: //chemconnections. org/general/chem 108/Math%20% 26%20 Measurement-2018. pdf

Select a lab partner. Discussion Question The title of today’s experiment is: A. Measurement of the Properties of Gases B. Metal Measurement C. Measuring the Energy of Combustion D. Metric Measurement E. Mathematics & Measurement

Answer D. Experiment 1 – Metric Measurement
![Doing: Lab Experiments Metric Measurement [Experiment #1] Background & Preparation [Graded Guiding Questions] http: Doing: Lab Experiments Metric Measurement [Experiment #1] Background & Preparation [Graded Guiding Questions] http:](http://slidetodoc.com/presentation_image_h2/e93bfdb6734d472d0dfb527f4a4b0880/image-11.jpg)
Doing: Lab Experiments Metric Measurement [Experiment #1] Background & Preparation [Graded Guiding Questions] http: //chemconnections. org/general/chem 108/Measurements. Units-Guide. html Must be submitted before noon Friday to receive any credit.
![Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp. Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp.](http://slidetodoc.com/presentation_image_h2/e93bfdb6734d472d0dfb527f4a4b0880/image-12.jpg)
Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp. 12 -15 [Report Form]) http: //chemconnections. org/general/chem 108/calendar-108 -f 19. html Collaboration is encouraged, but individual record keeping and submissions are required. MUST use Lab Manual pages for record keeping. Black or blue ink preferred without erasures, but pencil OK for Chem 108.
![Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp. Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp.](http://slidetodoc.com/presentation_image_h2/e93bfdb6734d472d0dfb527f4a4b0880/image-13.jpg)
Doing: Lab Experiments Metric Measurement [Experiment #1] (Course/ Lab Manual pp. 9 -11; pp. 12 -15 [Report Form])

Exp. 1 – Metric Measurement Ø Goal: Using instruments having different levels of precision, make accurate measurements of length, area, volume, and mass Ø Convert measurements to different units using Dimensional Analysis

Exp. 1 – Metric Measurement Work with a partner Ø Keep separate records. On each partner’s Lab REPORT FORM, write your name first & then your partner’s in (____) S. Curry (D. Green) and on your partner’s form D. Green (S. Curry)

Exp. 1 – Metric Measurement Ø Do each measurement separately and independently. Then, compare your value with your partner’s. The values should be very close within the precision limits of the device used. If not, repeat the measurement together and correct the Report Form entries.

Exp. 1 – Metric Measurement Mass Measurement with an Electronic Balance: https: //www. youtube. com/watch? v=Qtn. Pi. KSKKt. I
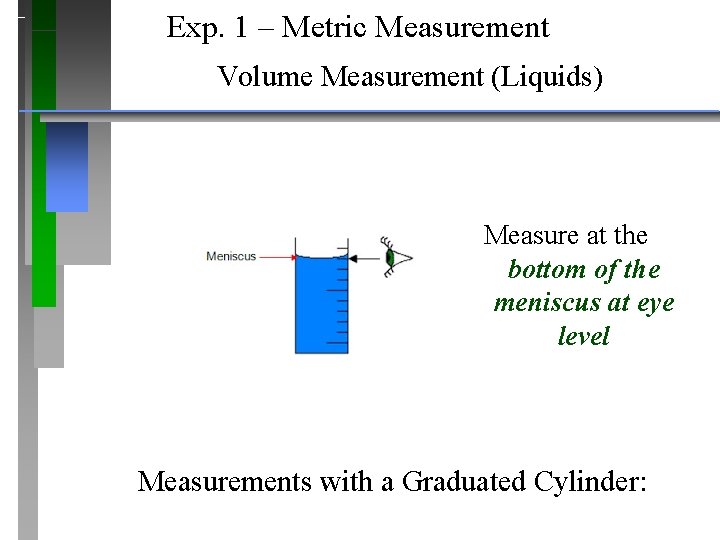
Exp. 1 – Metric Measurement Volume Measurement (Liquids) Measure at the bottom of the meniscus at eye level Measurements with a Graduated Cylinder:
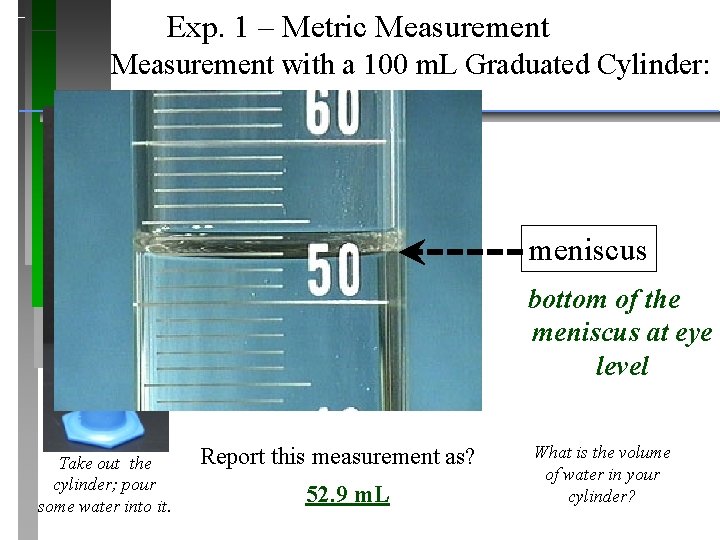
Exp. 1 – Metric Measurement with a 100 m. L Graduated Cylinder: meniscus bottom of the meniscus at eye level Take out the cylinder; pour some water into it. Report this measurement as? 52. 9 m. L What is the volume of water in your cylinder?

Exp. 1 – Metric Measurement with a 10 m. L Graduated Cylinder: Measure at the bottom of the meniscus at eye level meniscus Report this measurement as? Take out the cylinder; pour some water into it. 6. 31 m. L What is the volume of water in your cylinder?

Exp. 1 – Metric Measurement Ø Important to record measurements to the correct limits of the equipment used (i. e. uncertainty/significant figures). NOTE: For this experiment the uncertainty limits (+/−) of the equipment is to be included. Ø Uncertainty limits are not normally included in calculations but are inferred from the correctly reported significant figure in the experimental value. Ø Estimating to 6. 30 m. L is ok if the meniscus is viewed to be exactly on the mark. Report: 6. 30 +/- 0. 01 m. L Ø Estimating to 6. 31 m. L is also ok if the meniscus is viewed to be off the mark. Report: 6. 31 +/- 0. 01 m. L
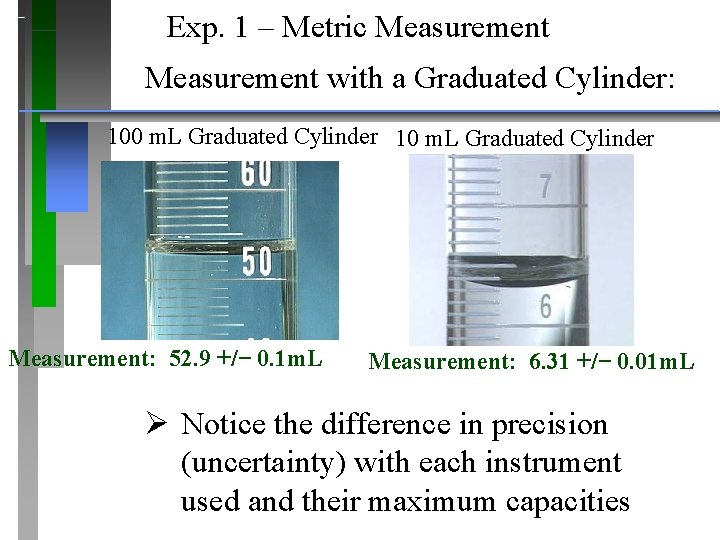
Exp. 1 – Metric Measurement with a Graduated Cylinder: 100 m. L Graduated Cylinder 10 m. L Graduated Cylinder Measurement: 52. 9 +/− 0. 1 m. L Measurement: 6. 31 +/− 0. 01 m. L Ø Notice the difference in precision (uncertainty) with each instrument used and their maximum capacities
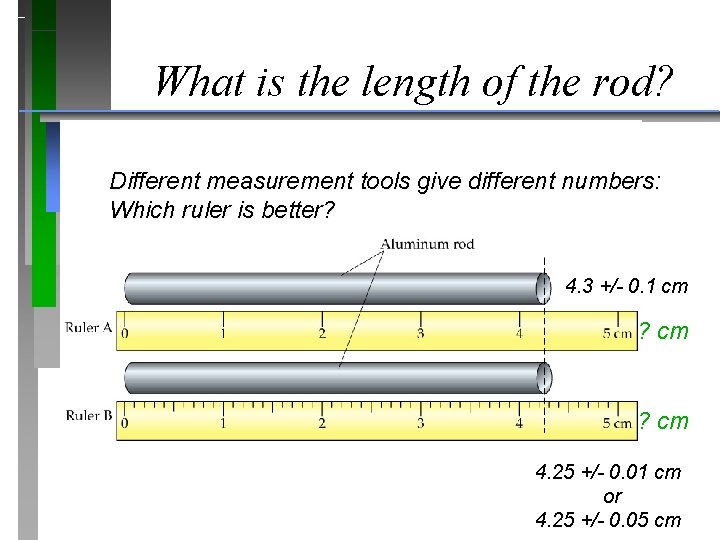
What is the length of the rod? Different measurement tools give different numbers: Which ruler is better? 4. 3 +/- 0. 1 cm ? cm 4. 25 +/- 0. 01 cm or 4. 25 +/- 0. 05 cm
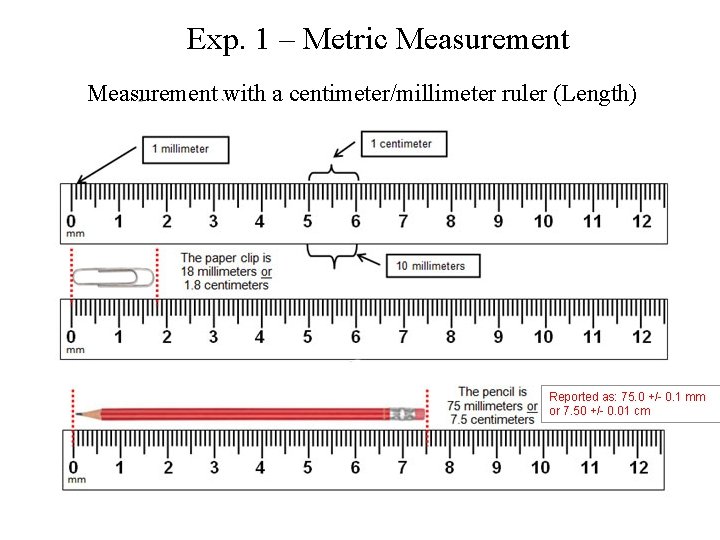
Exp. 1 – Metric Measurement with a centimeter/millimeter ruler (Length) Reported as: 75. 0 +/- 0. 1 mm or 7. 50 +/- 0. 01 cm

Exp. 1 – Metric Measurement Ø When measuring glassware with the ruler, use inner diameter of glassware, NOT outer diameter. Why? Ø Experimental error will occur: When pouring water out of test tube into graduated cylinder, some is always left in test tube; how does this systematic error affect accuracy of measured volume? Is the beaker a perfect cylinder? Ø Think about how equipment and handling; relate this to measurements and “systematic” errors. Ø “Human Error” is NOT acceptable error.

Exp. 1 – Metric Measurement “Human Error” is NOT acceptable in scientific measurements……. as in aeronautics. SFO July 6, 2013

Experiment 1 – Metric Measurement Complete and record all measurements today. Conversions and calculations due next lab.

Refer to last week’s handout http: //chemconnections. org/general/chem 108/WKS%20 Reading %20 Unit%20 Conversion%20 -%20 Dimensional%20 A. pdf

What are significant figures? 5300. m. L 5300 m. L 0. 01 kg 0. 0100 kg In scientific notation? 5. 300 x 103 m. L 5. 3 x 103 m. L 1 x 10 -2 kg 1. 00 x 10 -2 kg Worksheet: Working with your partner, complete questions 1&2 today. Collaboration is encouraged; you may “confer” with others beside your partner. Have Dr. R. sign individual forms before leaving lab today http: //chemconnections. org/general/chem 108/Math%20%26%20 Measure ments-WKS. f 18. pdf Completed Worksheets due 19 -Feb.
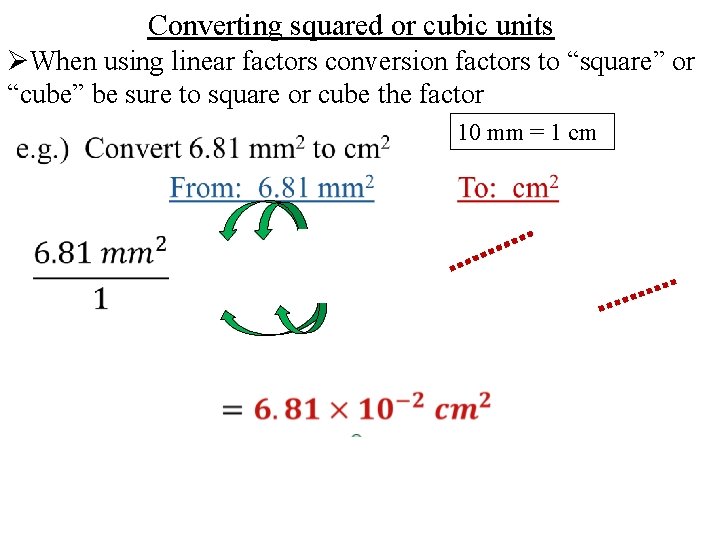
Converting squared or cubic units ØWhen using linear factors conversion factors to “square” or “cube” be sure to square or cube the factor 10 mm = 1 cm

10 mm = 1 cm

Experiment 1 – Metric Measurement Complete and record all measurements today. Have Dr. R. sign individual forms before leaving lab today. Only your measurement data is due to be signed today. Conversions and calculations due next lab.

Exp. 1 – Metric Measurement Example of an acceptable set of student data, conversions, and calculations. DO NOT COPY. Use as a guide.

Before starting today’s Lab Experiment http: //chemconnections. org/general/chem 108/Lab/Safety_focus_ques-18. pdf Put on Safety Glasses
- Slides: 34