Chem 108 Class Lab Week 14 2019 f

Chem 108: Class/ Lab Week 14: 2019 f Sign in / Pick up Papers Due Today: 1) Fluid Exchange (Handout) 2) Acid-Base: p. H (Handout) 1 completed form per group with names of all who contributed Do Today: Laboratory Manual Acid & Base Titration Procedure (pp. 91 -93) Each of you are to take a clean 250 m. L erlenmeyer flask and place it in the gray plastic tub on the lab’s side bench.

https: //www. youtube. com/watch? v=9 Dk. B 82 x. Lv. NE Neutralization Reactions Titration http: //chemconnections. org/general/movies/acidbasetitration. mov H+(aq) + OH -(aq) H 3 O+(aq) + OH -(aq) H 2 O(l)

Neutralizations / Titrations Chem 108 titration: phenolphthalein indicator Chem 120/121 Titration Curves
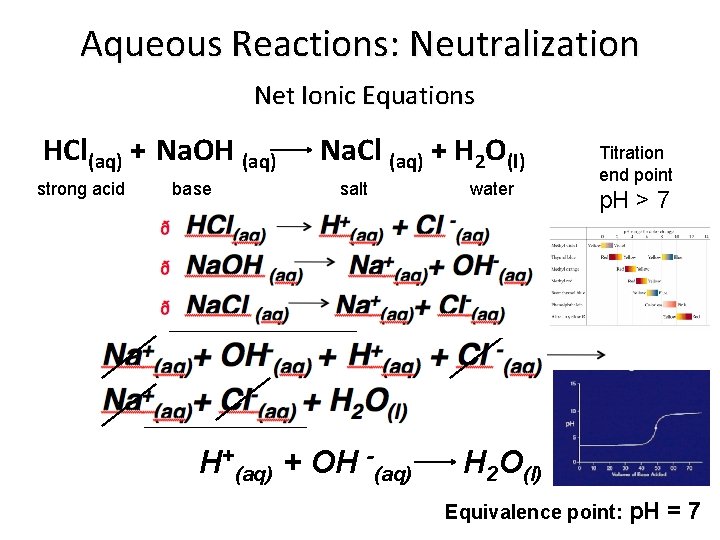
Aqueous Reactions: Neutralization Net Ionic Equations HCl(aq) + Na. OH (aq) strong acid base Na. Cl (aq) + H 2 O(l) salt H+(aq) + OH -(aq) water Titration end point p. H > 7 H 2 O(l) Equivalence point: p. H =7
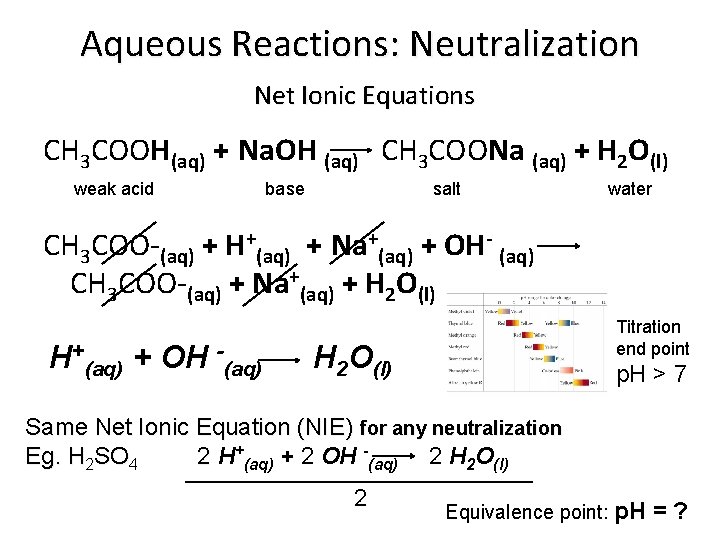
Aqueous Reactions: Neutralization Net Ionic Equations CH 3 COOH(aq) + Na. OH (aq) CH 3 COONa (aq) + H 2 O(l) weak acid base salt water CH 3 COO-(aq) + H+(aq) + Na+(aq) + OH- (aq) CH 3 COO-(aq) + Na+(aq) + H 2 O(l) H+(aq) + OH -(aq) Titration end point H 2 O(l) p. H > 7 Same Net Ionic Equation (NIE) for any neutralization Eg. H 2 SO 4 2 H+(aq) + 2 OH -(aq) 2 H 2 O(l) _____________ 2 Equivalence point: p. H =?
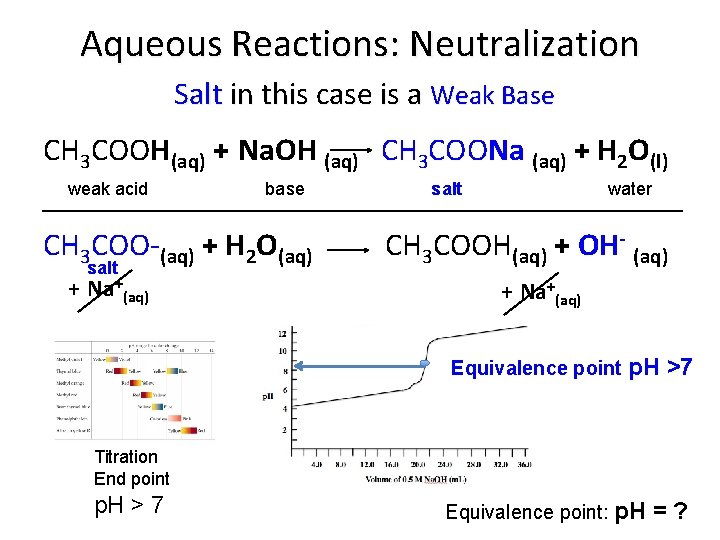
Aqueous Reactions: Neutralization Salt in this case is a Weak Base CH 3 COOH(aq) + Na. OH (aq) CH 3 COONa (aq) + H 2 O(l) weak acid base salt water ________________________ CH 3 COO-(aq) + H 2 O(aq) salt + Na+(aq) CH 3 COOH(aq) + OH- (aq) + Na+(aq) Equivalence point p. H >7 Titration End point p. H > 7 Equivalence point: p. H =?

Acid-Base Titration Part 1: Standardization will NOT be done. Part 2: Will be done individually.
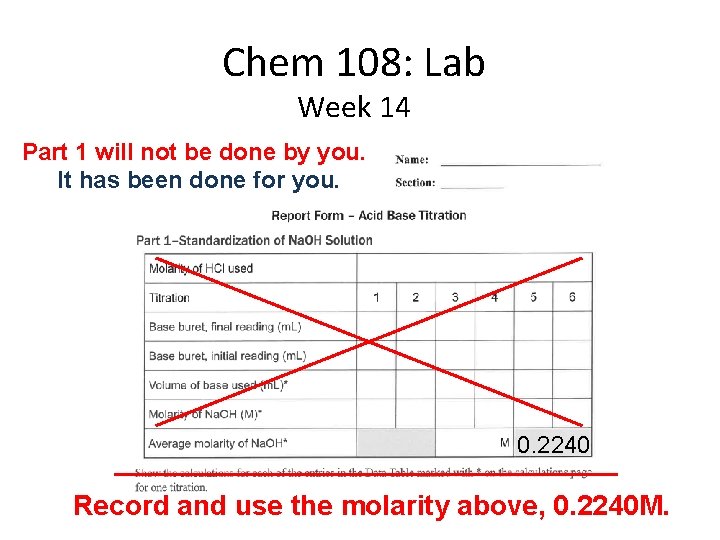
Chem 108: Lab Week 14 Part 1 will not be done by you. It has been done for you. 0. 2240 Record and use the molarity above, 0. 2240 M.
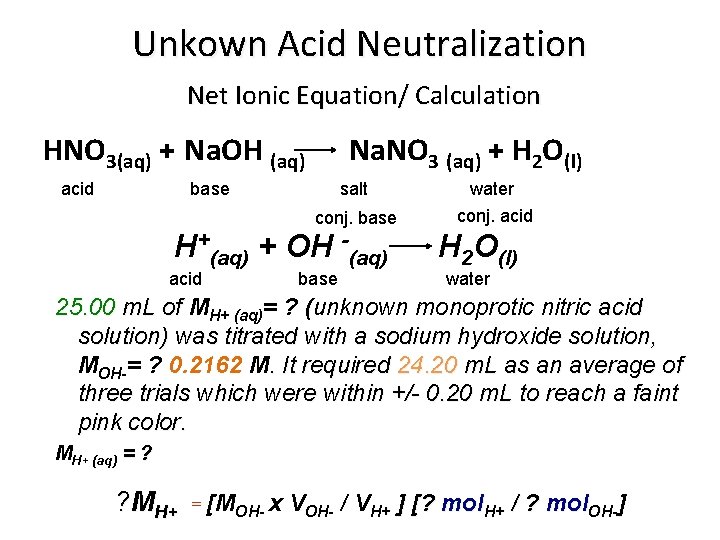
Unkown Acid Neutralization Net Ionic Equation/ Calculation HNO 3(aq) + Na. OH (aq) acid Na. NO 3 (aq) + H 2 O(l) base salt conj. base H+(aq) + OH -(aq) acid base water conj. acid H 2 O(l) water 25. 00 m. L of MH+ (aq)= ? (unknown monoprotic nitric acid solution) was titrated with a sodium hydroxide solution, MOH-= ? 0. 2162 M. It required 24. 20 m. L as an average of three trials which were within +/- 0. 20 m. L to reach a faint pink color. MH+ (aq) = ? ? MH+ = [MOH- x VOH- / VH+ ] [? mol. H+ / ? mol. OH-]

Unkown Acid Neutralization Net Ionic Equation/ Calculation H+(aq) + OH -(aq) acid base H 2 O(l) water 25. 00 m. L of MH+ aq= ? (unknown monoprotic acid solution) was titrated with a sodium hydroxide solution, MOH-= ? 0. 2162 M. It required 24. 20 m. L as an average of three trials which were within +/- 0. 20 m. L to reach a faint pink color. ? MH+ = = [MOH- x VOH- / VH+ ] [? mol. H+ / ? mol. OH-] x 0. 02420 LOH- x 1 mol. H+ LOH- x 0. 02500 LH+ x 1 mol. OH- 0. 2162 mol. OH- = 0. 2093 MH+

QUESTION A 35. 00 m. L sample of a monoprotic acid of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the unknown acid? A. 0. 0930 M B. 0. 3030 M C. 0. 2719 M D. 0. 1356 M E. 0. 3720 M ? MH+ H+(aq) + OH -(aq) = [MOH- x H 2 O(l) VOH- / VH+ ] [? mol. H+ / ? mol. OH-]

QUESTION A 35. 00 m. L sample of sulfuric acid (a di-protic acid) of unknown concentration was titrated with 42. 30 m. L of 0. 2250 M KOH. What is the concentration of the unknown acid? A. 0. 0930 M B. 0. 3030 M C. 0. 2719 M D. 0. 1356 M E. 0. 3720 M ? MH + 1 H 2 SO 4 = [MOH- x 2 H+(aq) + OH -(aq) 2 H 2 O(l) VOH- / VH+ ] [? mol. H 2 SO 4 / ? mol. OH-]

Chem 108: Lab Part 2: Week 13 To Do today (individually); each of you will do a separate unknown. _____ 0. 2240 3 trials must be within +/- 0. 20 m. L 1 m. L ~ 20 drops Each of you will do a separate unknown. Take an erlenmeyer flask from the gray tub and record its code & the Molarity of Base.

Chem 108: Lab Part 2: Week 13 To Do today (individually); each of you will do a separate unknown. _____ 0. 2240 3 trials must be within +/- 0. 20 m. L 1 m. L ~ 20 drops After completing enough trials that 3 of them are within +/- 0. 20 m. L show this data page to Dr. R.
- Slides: 14