CHEM 1011 Laboratory Techniques Using the Laboratory Gas

CHEM 1011 Laboratory Techniques: Using the Laboratory Gas Burner; Making Laboratory Measurements

Safety Put on Goggles And Apron

Objectives • • Learn to use Bunsen burner Learn to use laboratory equipment Learn to take Measurements Record Measurements using the proper number of significant figures

Bunsen Burner • Allows Controlled Mixture of Gas and Air • Hottest Flame – Nonluminous – Violet outer cone – Complete combustion occurs CH 4 (g) +2 O 2 (g) → CO 2 (g) + 2 H 2 O (g)

Bunsen Burner • Cooler Flame – Less Air – Luminous Yellow Flame – No Inner Cone – Smokey due to incomplete combustion

Glassware • Glassware in lab is a borosilicate glass – Consists of silica and borax – Does not soften much under 800˚C – Pyrex or Kimax – Low thermal Expansion coefficient • Unlike soft Glass (Si. O 2) – softens from 300 -400˚C – High thermal Expansion coefficient

Laboratory Measurements • Metric system uses base 10 for measurements and conversions • Conversions are done by multiplying or dividing by 10

Laboratory Measurements • Evaluate physical and chemical environment – length, volume, mass, energy, and temperature • Good laboratory techniques are invaluable

Accuracy • Closest to the true value – True value = 34. 878 • Device measures – 34. 881 – 34. 876 – 34. 877

Precision • Reproducibility of the measurement • Little variability – True value = 34. 878 • Device measures – 35. 001 – 35. 002 – 35. 000
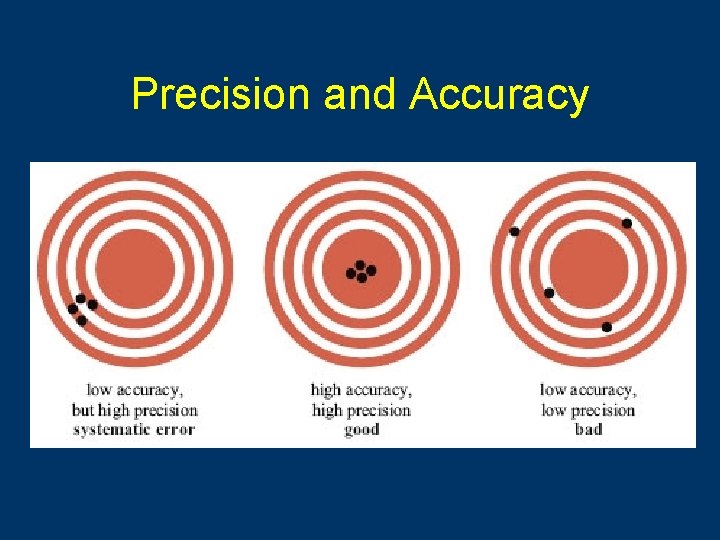
Precision and Accuracy

Significant Figures • When measuring: – (1) uncertainty in last digit recorded • 25. 45 – (2) the # of significant figures contains all known #’s + one estimated • 25. 45 – known = 25. 4; estimated = 0. 05

Examples

Division • The Answer’s # of sig. fig. are same as number with the fewest digits being divided • 12. 3 ÷ 6. 235 = 1. 972734563 = 1. 97

Multiplication • The Answer’s # of sig. fig. are same as number with the fewest digits being multiplied • 0. 72 × 1. 145 = 0. 8244 = 0. 82

Addition • The Answer’s # of sig. fig. are same as number with the fewest digits behind decimal being added 4. 23 + 3. 292 + 7. 849 = 15. 371 = 15. 37

Subtraction • The Answer’s # of sig. fig. are same as number with the fewest digits behind decimal being subtracted 6. 8384 – 3. 63 = 3. 2084 = 3. 21

Calculations • Length: Record measurement to the nearest 16 th of an inch
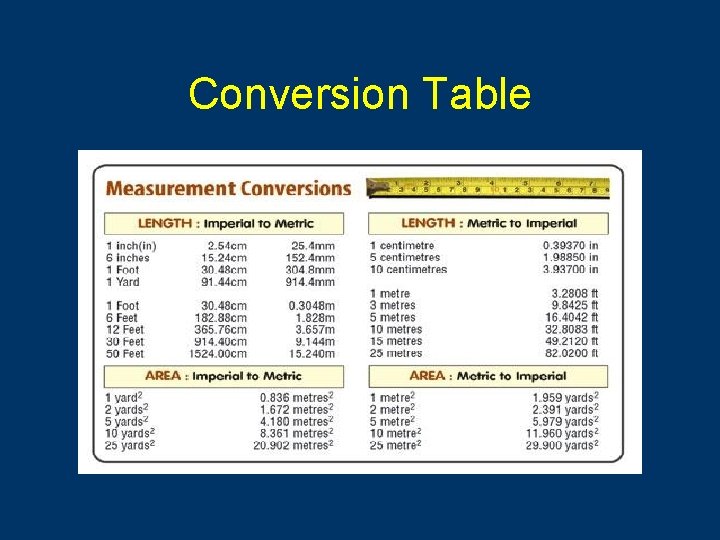
Conversion Table

Calculations (Measurement) • Record measurements • Conversions can be made by dividing or multiplying by powers of 10 • Ex. 34 cm × 10 mm/1 cm = 340 mm • Area L × W (340 mm × 23 mm) = 8160 mm 2

Calculations (Volume) • Record Volume • Convert using the conversion chart e. g. , 48 m. L × 1 L/1000 m. L = 0. 048 L • Calculate percent error = (theoretical value – actual value)/ theoretical value × 100

Calculations (Mass) • Record values • Convert from g to mg e. g. , 15 g × 1000 mg/1 g = 15000 mg

Calculations (Temperature) • Record the Value in degrees Celsius • Convert to Degrees Fahrenheit and Kelvin • Degrees Fahrenheit use (9/5 (C) + 32 • Degrees Kelvin use (C + 273)
- Slides: 23