Chem 1 B 830 Lecture Welcome to Chem

Chem. 1 B – 8/30 Lecture

Welcome to Chem 1 B Lecture Section 1 • Today’s Meeting – Introduction to Class – Roll Call (put name on notepad) – First Day Stuff: • • • Printed Handout (not the syllabus) Materials Needed Adding this Class Syllabus Lab – first two weeks – Chem 1 A Review (very brief) – First Topic: Chemical Equilibria
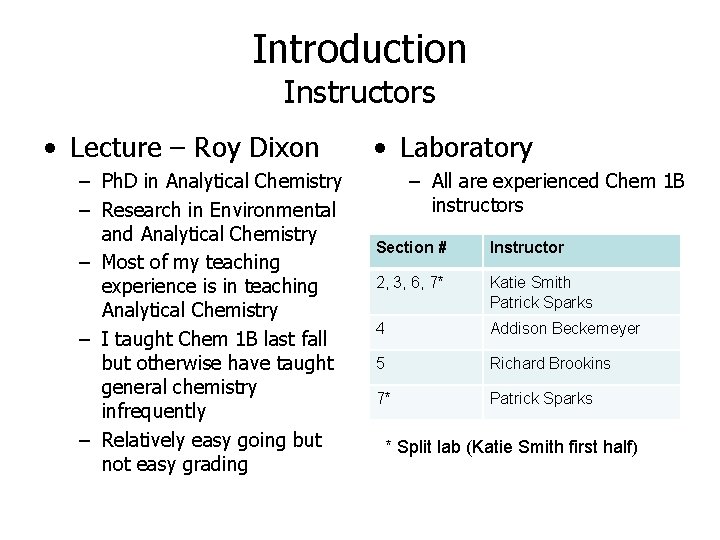
Introduction Instructors • Lecture – Roy Dixon – Ph. D in Analytical Chemistry – Research in Environmental and Analytical Chemistry – Most of my teaching experience is in teaching Analytical Chemistry – I taught Chem 1 B last fall but otherwise have taught general chemistry infrequently – Relatively easy going but not easy grading • Laboratory – All are experienced Chem 1 B instructors Section # Instructor 2, 3, 6, 7* Katie Smith Patrick Sparks 4 Addison Beckemeyer 5 Richard Brookins 7* Patrick Sparks * Split lab (Katie Smith first half)

Introduction Class – Main Teaching/Learning Goals • Lecture – – Topics: • Equilibrium Related Topics (Chapters 14 – 16) • Thermodynamics • Electrochemistry • Metal-Ligand Chemistry • Intro to Organic Chemistry – Besides learning topics, there is an emphasis on: • Higher level problem solving (can’t just use dimensional analysis) • Connecting topics to their importance • Lab – Topics: • Qualitative Analysis (what ions are in my water? ) • Practical aspects of equilibria (titrations) • Other topics: thermodynamics, electrochemistry and inorganic chemistry – Emphasis on: • Report Writing • Working Efficiently

Roll Call (today only) • Put name on passed sheet

First Day Stuff Class Materials • Class Handout – summary of materials • Websites for Class – My faculty website (see handout) – Mastering Site (online chemistry homework – should be set up) – Sac. CT (not set up yet – scores and keys) – PAL site (link is up) • Text • Scantrons (2 types - show) • Other Lab Supplies (covered in lab)

First Day Stuff Adding This Class • The Class Size is Limited to 144 (24 students per each of 6 lab sections) • Adding is done for lab sections – Only can add students to make up for dropped students (not showing up will get you dropped) – Priority based on waitlist # (for lab section); plus other priorities for “crashing” students – Was briefly full – Sections not full (My. Sac. State just before class) – Sect # (vacancies): 2 (2), 4 (1), 7 (1) – Most students will be added on second meeting with adding possible in 2 nd week to replace other dropped students • Lecture Only – If you are repeating the class and hope to skip the lab, see me

First Day Stuff Syllabus – Download from Website • It is your responsibility to read and understand the complete syllabus. I will go over certain parts, and you can ask questions • Page 1 notes: – lab instructor office hours posted on website – Sequoia 502 is the Help Office (don’t see instructor there about personal problems) – A C- in Chem 1 A is insufficient – Bring a calculator to lecture – Safety equipment is necessary to work in lab

First Day Stuff Syllabus – cont. • Page 2 notes: – Lecture will be strongly focused on problem solving and conceptual understanding (meant to compliment book learning) • Page 3 notes: – Lab attendance is required – read if you need to make up a lab – Grading – breakdowns may be curved downward – but most likely by only a few % at most and in conjunction with the other section – Quizzes will be held in the lab but will be split between lecture questions and questions related to pre-lab activities

First Day Stuff Syllabus – cont. • Page 4 notes: – Midterm Exams (3 see schedule for dates) – Last year, these were 23 multiple choice questions (1 bonus) for 88 points plus one work out problem for 12 points – I expect the same format this year – The version of the scantron used (SC 982 -E) allows you to do work out problem on back – Examples tests from last year are posted

First Day Stuff Syllabus – cont. • Page 5 notes: – Pay attention to pre-lab assignments and lab report deadlines – Don’t expect your lab instructor to give you additional time (past the end time of the lab period) – Disruptions • You are not required to be in lecture, but • Others are here to learn and can’t pay attention if disruptions are common – Help: • help office, other office hours, PALs program, private tutors • have good study habits early

First Day Stuff Syllabus – cont. • Page 6 notes: – If you drop, you also need to check out of lab – Bonus points for student participation (up to 5 points for correct answers) • Page 7 notes: – Exam dates are not likely to change, but I may get one lectures behind (so could change exam content) – Homework should match where we are in lecture – See bottom for what you may need to restudy from Chem 1 A

First Day Stuff Lab – First Two Weeks • Day 1: yesterday and today – mainly dealing with adding and overview of lab – diagnostic quiz is for you to know which parts of Chem 1 A we expect you to know – read lab manual and understand procedures (many differences from Chem 1 A) • Day 2: Wed. and Thur. – check in (+ adding students) – review of Chem 1 A material (dissolution in water and solubility)

First Day Stuff Lab – First Two Weeks • Day 3: Next Wed. and Thur. (no lab Mon. /Tu. ) – Pretty extensive pre-lab and then Exp. 1 (over two lab periods) – Only 2 nd period quiz

Chem 1 A Review Important Aspects for Chem 1 B • See Bottom of p. 7 of Syllabus • Chapter 3 - Nomenclature: = the language of chemistry - First, identify substance (molecule, ionic compound, acid) - ions have charge - molecules vs. ionic compounds: ionic compounds have cation and anion and almost all cations are metals (NH 4+ is exception)/molecules are all non-metals - molecule name: prefix first element prefix second element suffix - ionic compound name: metal [oxidation state] anion - anion names: memorize, but only as much as needed

Chem 1 A Review Important Aspects for Chem 1 B • Chapter 3 – Nomenclature – How to learn: - memorize (use what ever technique works for you – e. g. flash cards) + can “categorize” – examples – Zn. Cl 2 name = _____; type = ____ – OF 2 = ______; type = ____ – Cu. Cl = ______; type = ____ – Na 2 SO 3 = _____; type = ____ – aluminum chlorite = __; type = ______ – nitrous acid = ______; type = ____ = shuĭ = water

Chem 1 A Review Important Aspects for Chem 1 B • Chapter 4 - Aqueous Reactions: - Needed for First Lab - Needed for Chapters 15 and 16 (aqueous equilibria) - Concepts to Know: - Solution concentration (mainly molarity – how to calculate) - Dilutions - How substances dissolve into water (differences between molecules and ionic compounds + acids and bases) - Various reactions (e. g. precipitation, gas evolution, etc. ) + net ionic equations - Solubility rules

Chem 1 A Review Important Aspects for Chem 1 B • Other skill: Problem solving – First determine what the question is asking for – Determine data useful in getting the answer – Map out how to get to the answer • Chapter 4 – an example problem (more in lab) – A solution is made from 0. 202 g of Ca. Cl 2 (FW = 110. 98 g/mol) using a 250 m. L volumetric flask and deionized water (totally dissolves Ca. Cl 2). – What is the concentration in M? – How does the dissolved compound exist? – What is [Cl-]? – Will any further reactions in water occur? – If we add Ag. NO 3 to the above solution, what will occur?

Chem 1 B First Topic Equilibrium • In Chapter 13 (near end of Chem 1 A), we covered kinetics (how fast reactions occur) • Past assumption was that “→“ meant reaction went to completion (all products plus limiting reagent) • Also, unfavorable reactions, such as precipitation of Ca. Cl 2 from Ca 2+ + Cl- are not supposed to occur at all • In some important cases, expected reactions will not proceed totally to completion and unexpected reactions can occur to some extent

Chem 1 B - Equilibrium Introduction • Reactions that can occur in both the forward and reverse reactions to an appreciable extent are represented with a two way arrow: A+B C or A + B ↔ C (easier to type this) • What does this mean? • From a molecular scale, reactions are occurring in both the forwards and backwards direction • When the forward rate (loss of A to form C) is equal to the backwards rate (gain of A from C), the system is said to be in equilibrium and concentrations don’t change
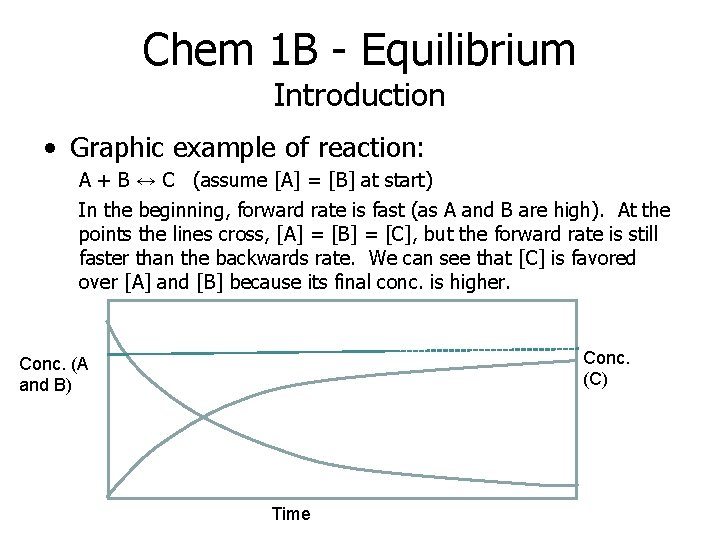
Chem 1 B - Equilibrium Introduction • Graphic example of reaction: A + B ↔ C (assume [A] = [B] at start) In the beginning, forward rate is fast (as A and B are high). At the points the lines cross, [A] = [B] = [C], but the forward rate is still faster than the backwards rate. We can see that [C] is favored over [A] and [B] because its final conc. is higher. Conc. (C) Conc. (A and B) Time

Chem 1 B - Equilibrium Introduction • Some examples: 1. Carbon dioxide (CO 2) is not very soluble (you are supposed to know it forms from acid + carbonates) – However, the acidity of rain in “pristine” conditions is supposed to be based on dissolution of CO 2: – CO 2 (g) + H 2 O (l) ↔ H 2 CO 3 (aq) ↔ H+ + HCO 3 - 2. Nitrogen gas (N 2) is difficult to react because of the strength of its triple bonds, however under certain conditions, beneficial and problematic reactions involve N 2 where both reactants and products exist. a) N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) (used in fertilizer) b) N 2 (g) + O 2 (g) ↔ 2 NO (g) (source of smog from combustion) Conditions are critical to make a) go but to limit b)

Chem 1 B - Equilibrium Constant and Equation • Whether an Equation favors reactants or products is determined by an EQUILIBRIUM CONSTANT (K) – as well as reaction stoichiometry • Generic reaction: a. A + b. B ↔ c. C + d. D • K = equilibrium constant and for above reaction, • A larger K value means products are more favored

Chem 1 B - Equilibrium Equation – Further Details • Technically, instead of concentration we have a concentration ratio to a “standard state” (e. g. 1 mol/L) – we can ignore this, but this is why K is unitless • Only species in gas phase or in solution will have concentrations. Solids and pure liquids are not included. • When gases are involved, there are two Ks: KC for concentration units (including gases) and KP in which pressure (atm) replaces concentration (assume K = KC unless specified) • Even further corrections (activity and fugacity) are needed under certain conditions but are beyond this class

Chem 1 B - Equilibrium Equation – Example • N 2 (g) + O 2 (g) ↔ 2 NO (g) or At T = 25°C KC = KP (in this case) = 4 x 10 -31 an insignificant amount of NO will form in air: PN 2 = 0. 8 atm , PO 2 = 0. 2 atm; rearranging the above equation, PNO = [KPPN 2 PO 2]0. 5 = 2 x 10 -16 atm Equilibrium at room temp. is not realistic because kinetics is too slow (insufficient collision energy to break triple bond) At car engine temp. , reactions are faster and K is much larger. This leads to significant NO formation. Fuel rich conditions (low P O 2 ) can limit NO formation.
- Slides: 25