Check Mate 025 A randomized openlabel phase III

Check. Mate 025: A randomized, open-label, phase III study of nivolumab versus everolimus in advanced renal cell carcinoma Padmanee Sharma, Bernard Escudier, David F. Mc. Dermott, Saby George, Hans J. Hammers, Sandhya Srinivas, Scott S. Tykodi, Jeffrey A. Sosman, Giuseppe Procopio, Elizabeth R. Plimack, Daniel Castellano, Howard Gurney, Frede Donskov, Petri Bono, John Wagstaff, Thomas C. Gauler, Takeshi Ueda, Li-An Xu, Ian M. Waxman, Robert J. Motzer, on behalf of the Check. Mate 025 investigators

Introduction ▪ Each year, an estimated 338, 000 new cases of renal-cell carcinoma are diagnosed worldwide, and approximately 30% of patients present with metastatic disease at the time of diagnosis ▪ A number of targeted therapies have been approved for the treatment of advanced or metastatic RCC based on PFS ▪ Current therapies provide limited OS benefit in patients who have been previously treated, highlighting a significant unmet medical need ▪ This phase III study compared nivolumab, a PD-1 immune checkpoint inhibitor, versus everolimus in patients with m. RCC after prior systemic therapy RCC, renal cell carcinoma; PFS, progression-free survival; OS, overall survival; PD-1, programmed death-1. 2
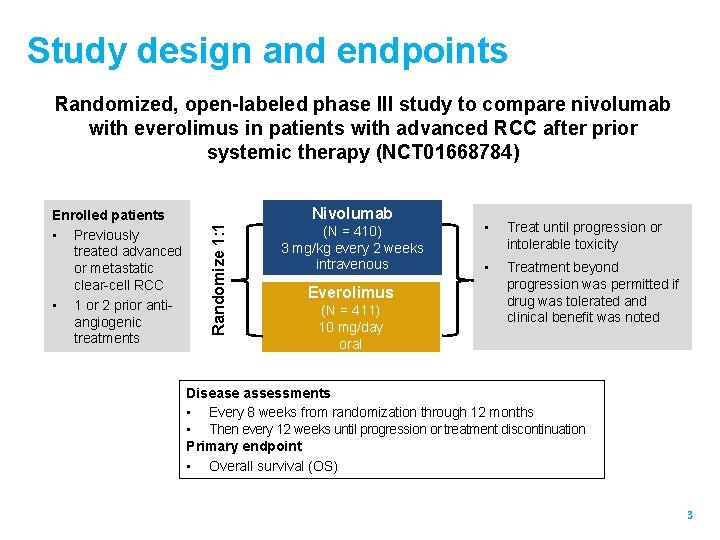
Study design and endpoints Randomized, open-labeled phase III study to compare nivolumab with everolimus in patients with advanced RCC after prior systemic therapy (NCT 01668784) Nivolumab Randomize 1: 1 Enrolled patients • Previously treated advanced or metastatic clear-cell RCC • 1 or 2 prior antiangiogenic treatments (N = 410) 3 mg/kg every 2 weeks intravenous Everolimus (N = 411) 10 mg/day oral • Treat until progression or intolerable toxicity • Treatment beyond progression was permitted if drug was tolerated and clinical benefit was noted Disease assessments • Every 8 weeks from randomization through 12 months • Then every 12 weeks until progression or treatment discontinuation Primary endpoint • Overall survival (OS) 3
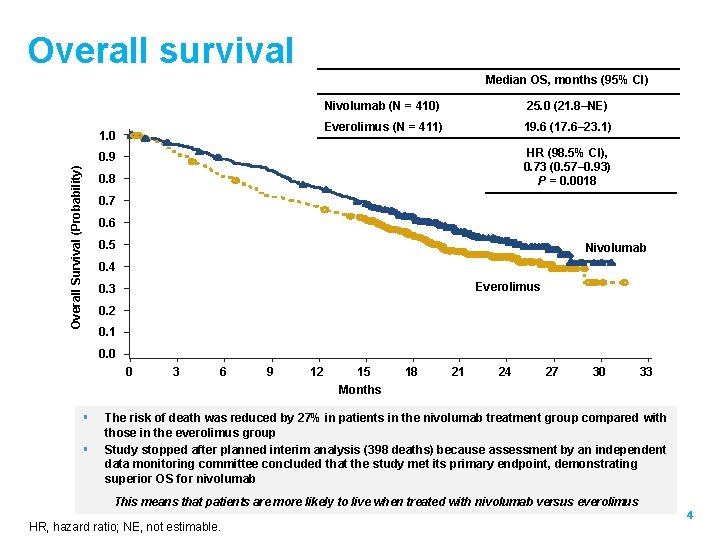
Overall survival Median OS, months (95% CI) 1. 0 Nivolumab (N = 410) 25. 0 (21. 8–NE) Everolimus (N = 411) 19. 6 (17. 6– 23. 1) HR (98. 5% CI), 0. 73 (0. 57– 0. 93) P = 0. 0018 Overall Survival (Probability) 0. 9 0. 8 0. 7 0. 6 0. 5 Nivolumab 0. 4 Everolimus 0. 3 0. 2 0. 1 0. 0 0 3 6 9 12 15 18 21 24 27 30 33 Months § § The risk of death was reduced by 27% in patients in the nivolumab treatment group compared with those in the everolimus group Study stopped after planned interim analysis (398 deaths) because assessment by an independent data monitoring committee concluded that the study met its primary endpoint, demonstrating superior OS for nivolumab This means that patients are more likely to live when treated with nivolumab versus everolimus HR, hazard ratio; NE, not estimable. 4
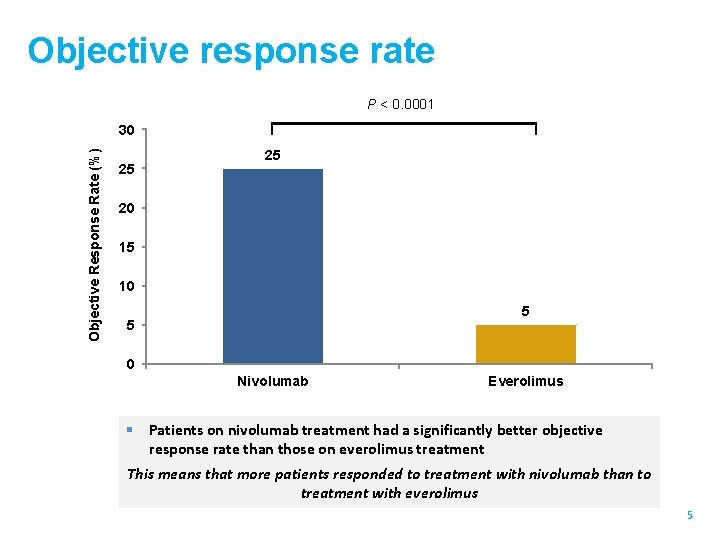
Objective response rate P < 0. 0001 Objective Response Rate (%) 30 25 25 20 15 10 5 5 0 Nivolumab Everolimus § Patients on nivolumab treatment had a significantly better objective response rate than those on everolimus treatment This means that more patients responded to treatment with nivolumab than to treatment with everolimus 5

Treatment-related AEs § Grade 3 or 4 treatment-related AEs were less frequent with nivolumab than with everolimus and treatment-related adverse events leading to discontinuation were experienced by fewer patients treated with nivolumab § The most common treatment-related AEs of any grade reported in the nivolumab arm were fatigue (33%), nausea (14%), and pruritus (14%), and in the everolimus arm, fatigue (34%), stomatitis (29%), and anemia (24%) § There were no treatment-related deaths in the nivolumab treatment arm This suggests that nivolumab has a favorable safety profile in patients with m. RCC AE, adverse event; m. RCC, metastatic renal cell carcinoma. 6

Key conclusions ▪ Check. Mate 025 met its primary endpoint, demonstrating OS superiority with nivolumab versus everolimus ▪ This is the only phase III trial to demonstrate a survival advantage in previously-treated patients with m. RCC versus standard therapy ▪ Nivolumab was associated with a greater number of objective responses than everolimus ▪ The survival improvement and favorable safety profile demonstrated in this phase III trial provides evidence for nivolumab as a potential new treatment option for previously treated patients with m. RCC ▪ Based on the positive results of this trial, nivolumab was granted a breakthrough therapy designation from the FDA for advanced RCC, reinforcing the importance of these results in a patient population with large unmet medical need 7
- Slides: 7