CHE 2060 Topic 1 Atoms orbitals bonding sp

CHE 2060 Topic 1: Atoms, orbitals & bonding sp 2 hybridization of carbon
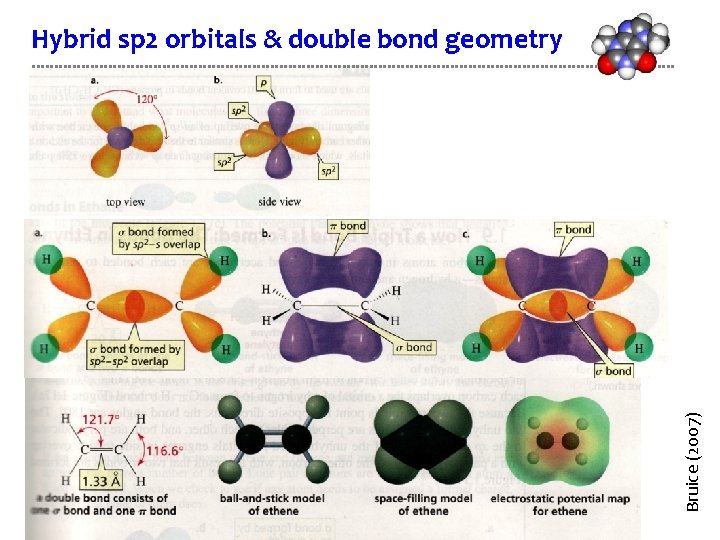
Bruice (2007) Hybrid sp 2 orbitals & double bond geometry
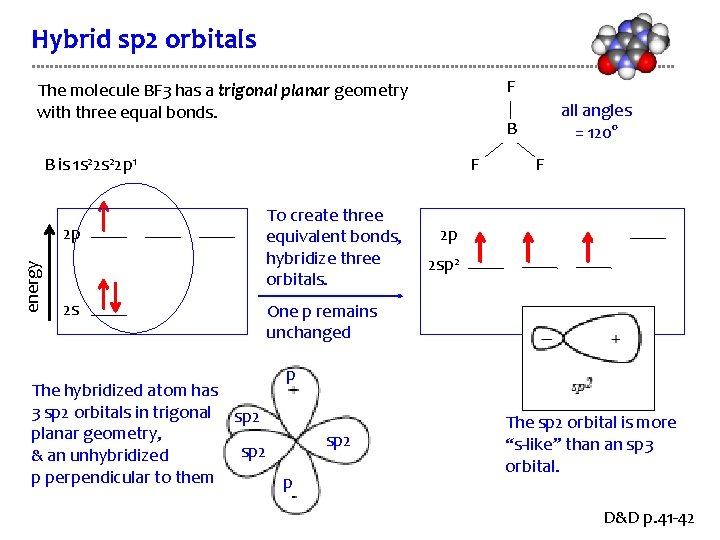
Hybrid sp 2 orbitals F The molecule BF 3 has a trigonal planar geometry with three equal bonds. B B is 1 s 22 p 1 energy 2 p 2 s F To create three equivalent bonds, hybridize three orbitals. all angles = 120° F 2 p 2 sp 2 One p remains unchanged p The hybridized atom has 3 sp 2 orbitals in trigonal sp 2 planar geometry, sp 2 & an unhybridized p perpendicular to them p sp 2 The sp 2 orbital is more “s-like” than an sp 3 orbital. D&D p. 41 -42
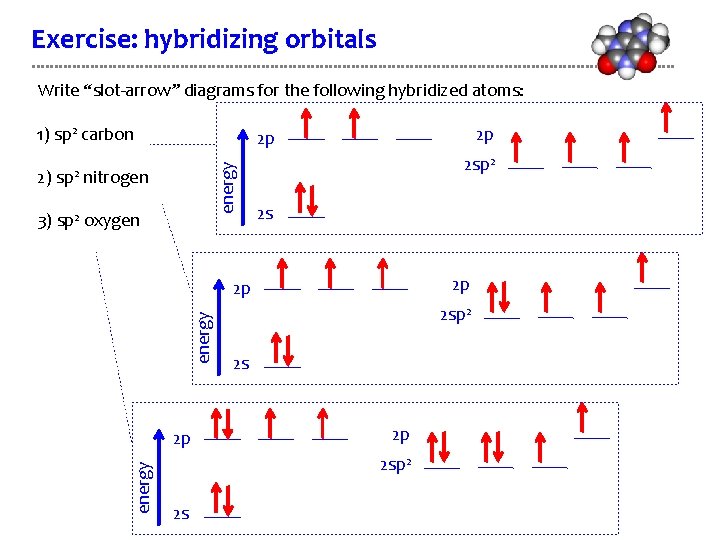
Exercise: hybridizing orbitals Write “slot-arrow” diagrams for the following hybridized atoms: 1) sp 2 carbon energy 2) sp 2 2 p 2 p nitrogen 3) sp 2 oxygen 2 sp 2 2 s 2 p energy 2 p 2 sp 2 2 s
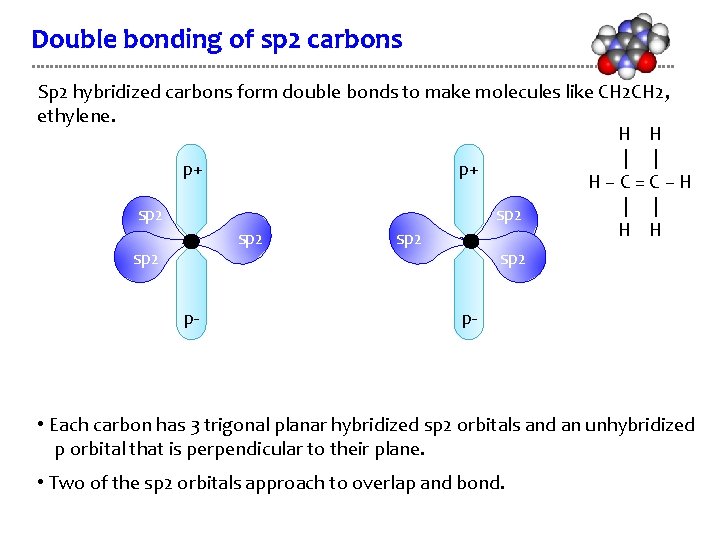
Double bonding of sp 2 carbons Sp 2 hybridized carbons form double bonds to make molecules like CH 2, ethylene. H H | | p+ p+ H–C=C–H | | sp 2 H H sp 2 p- p- • Each carbon has 3 trigonal planar hybridized sp 2 orbitals and an unhybridized p orbital that is perpendicular to their plane. • Two of the sp 2 orbitals approach to overlap and bond.
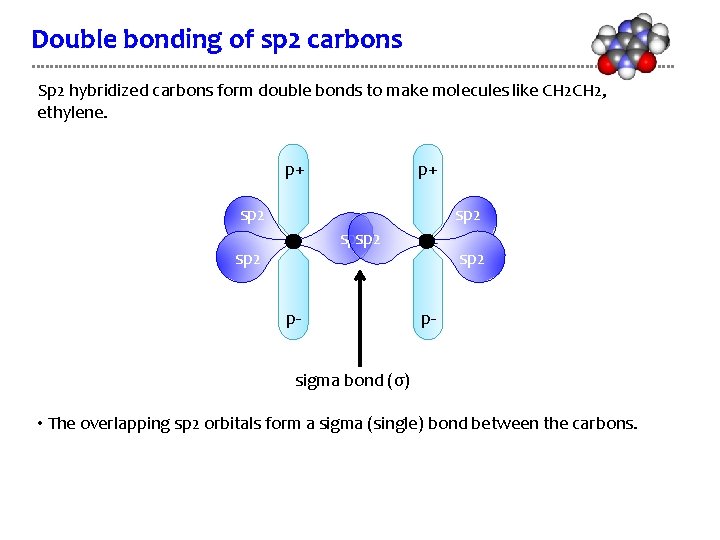
Double bonding of sp 2 carbons Sp 2 hybridized carbons form double bonds to make molecules like CH 2, ethylene. p+ p+ sp 2 sp 2 p- sigma bond (σ) • The overlapping sp 2 orbitals form a sigma (single) bond between the carbons.
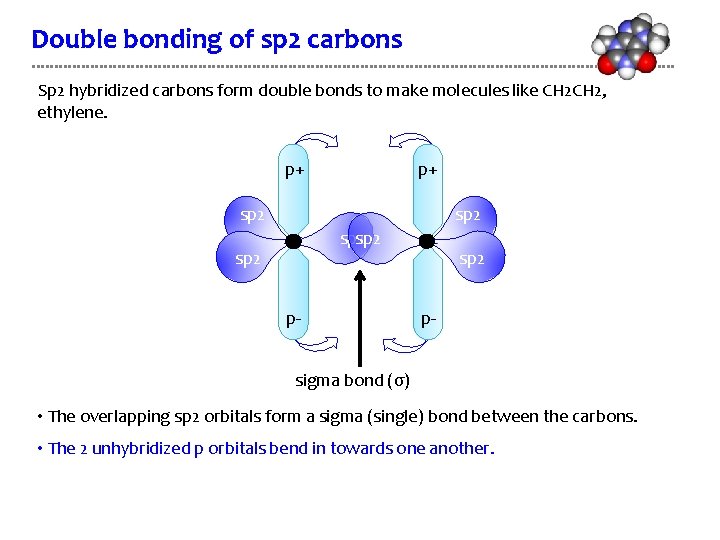
Double bonding of sp 2 carbons Sp 2 hybridized carbons form double bonds to make molecules like CH 2, ethylene. p+ p+ sp 2 sp 2 p- sigma bond (σ) • The overlapping sp 2 orbitals form a sigma (single) bond between the carbons. • The 2 unhybridized p orbitals bend in towards one another.
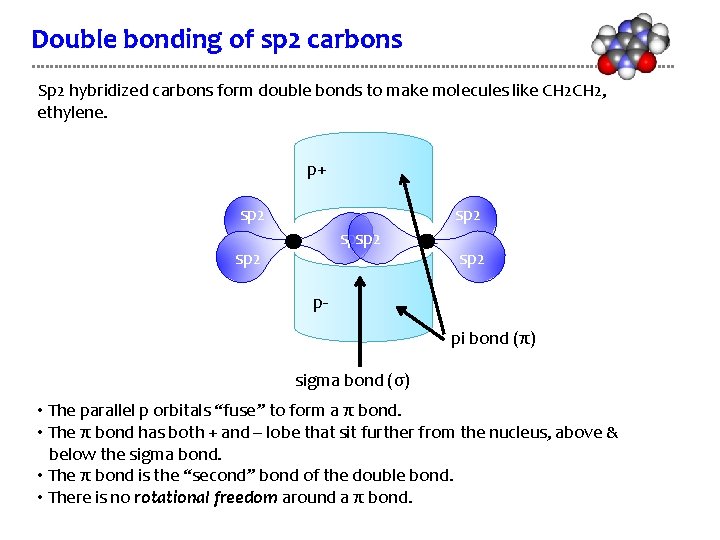
Double bonding of sp 2 carbons Sp 2 hybridized carbons form double bonds to make molecules like CH 2, ethylene. p+ sp 2 sp 2 ppi bond (π) sigma bond (σ) • The parallel p orbitals “fuse” to form a π bond. • The π bond has both + and – lobe that sit further from the nucleus, above & below the sigma bond. • The π bond is the “second” bond of the double bond. • There is no rotational freedom around a π bond.

Exercise: show hybridization & geometry Here are two molecules in which double bonds form between carbon & oxygen. For each draw a structure showing both bond angles & orbitals. O 1) Acetaldehyde || CH 3 – C – H O || 2) Methyl acetate CH 3 – C – O – CH 3 Acetaldehyde: methyl group is tetrahedral, sp 3, 109. 5 central C is sp 2 (120) & empty p forms π with O O is sp 2 w/ e- pairs in 2 sp 2 (120°), empty p forms π with C Methyl acetate: methyl groups are tetrahedral, sp 3, 109. 5° central C is sp 2 (120°) & empty p forms π with O double O is sp 2 w/ e- pairs in 2 sp 2 (120°), empty p forms π single-bonded O is tetrahedral, sp 3, 109. 5° w/ free pairs in 2 orb.

Can you? (1) Draw sp 2 hybridized orbitals? (2) Draw slot diagrams to explain sp 2 orbital hybridization? (3) Explain how VSEPR explains the spatial orientation of sp 2 orbitals? (4) Describe how overlapping sp 2 and unhybridized p orbitals form double bonds (sigma + pi bonds)? (2) Describe and explain the relative reactivities of sigma and pi bonds? Aristotle 1700’s gas pump experiment
- Slides: 10