CHE 2060 Topic 1 Atoms orbitals bonding Atoms

CHE 2060 Topic 1: Atoms, orbitals & bonding Atoms, Orbitals & Bonding Topics: 1. Very quick history of chemistry… 2. What is organic chemistry? 3. Atomic models: nuclear to quantum 4. All about orbitals 5. How orbitals fill: electron configuration 6. Basic bonding: valence electrons & molecular orbitals 7. Lewis dot structures of molecules 8. Electronegativity & bond polarity 9. Resonance: a critical concept 10. Orbital hybridization: key to carbon’s “flexibility” sp 3 sp 2 sp 11. Free electron pairs & radicals 12. VSEPR: classifying molecular geometry Daley & Daley, Chapter 1 & orbital hybridization Atoms, Orbitals & Bonds

CHE 2060 Topic 1: Atoms, orbitals & bonding Basic bonding: valence electrons & molecular orbitals

Lewis dots “visualize” valence e- • • Lewis dot diagrams simply represent each valence electron as a dot placed around an elements “letter” symbol. • • • • / • • ° ° • / Mg • / B • / • C • H • / Cl • ° • N • • O • • ° +1 +2 +3 ? -3 -2 -1 In order to obtain a full valence shell atoms will often gain or lose valence electrons. • If their shell is less then half full, they lose electrons to empty that shell, revealing the next intact shell. Octet • If greater than half full they gain electrons to reach an octet. Rule Gain or loss of electrons causes neutral atoms to become charged ions. The Law of Conservation of Mass requires that lost electrons are gained by other atoms, so the formation of cations & anions is a complementary process. Once formed, oppositely charged ions are attracted to one another via the Law of Electrostatic Attraction, & form molecules via ionic bonding. Molecular net charge = 0. . . . Mg. + 2: Cl. Mg : Cl: (Mg. Cl 2 = magnesium chloride). . --> : Cl: . . -1 +2 -1

Carbon forms covalent bonds… …because it’s valence shell is half-full. So it’s not likely to achieve a full valence shell by gaining or losing. Instead, it pools its resources to achieve an octet. • A covalent bond is defined as two e- that are shared when orbitals overlap. H: H . . . C: C. . . H. + H. --> H : H + heat --> H. + H. . . C: H. . . : O: H. For which are all octets satisfied? Why the heat? Heat is released by the formation of bonds, and is required to break bonds. This is called bond enthalpy; here 104 kcal/mol for the H-H bond. Bond length? 0. 74 x 10 -10 meters = 0. 74 Å Bond length & bond enthalpy are characteristic of covalent bonds linking specific pairs of atoms. D&D p. 28 -29

Covalent bonds are really “molecular orbitals” Molecules are formed when atoms collide & their valence orbitals overlap, leading to sharing of e-s in pairs - one from each atom. • Shared atomic orbitals become molecular orbitals. The interaction of orbitals is like the interaction of waves: • Waves traveling in the same direction add; the summed wave is larger. • Waves traveling in opposite directions cancel one another out. Orbitals interact via in-phase or out-of-phase overlap The bonding molecular orbital In-phase overlap of 1 s Out-of-phase overlap of 1 s forms a node. Antibonding molecular orbitals are usuallynot occupied; ie empty. H 2 The antibonding molecular orbital D&D p. 30 -31
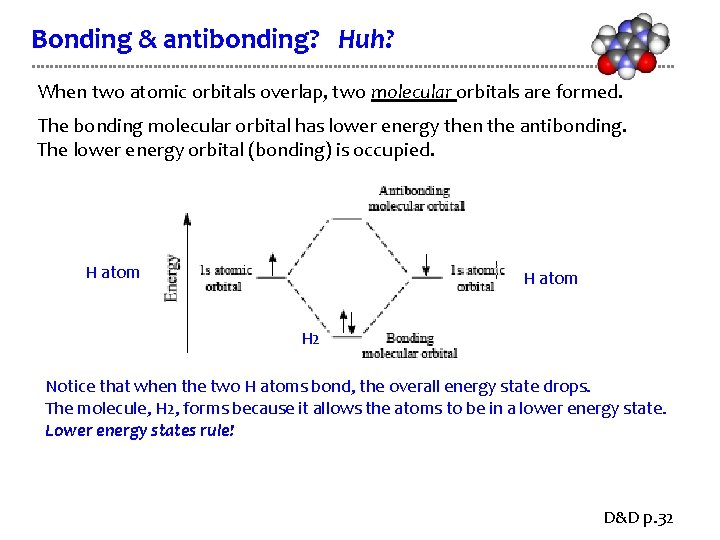
Bonding & antibonding? Huh? When two atomic orbitals overlap, two molecular orbitals are formed. The bonding molecular orbital has lower energy then the antibonding. The lower energy orbital (bonding) is occupied. H atom H 2 Notice that when the two H atoms bond, the overall energy state drops. The molecule, H 2, forms because it allows the atoms to be in a lower energy state. Lower energy states rule! D&D p. 32
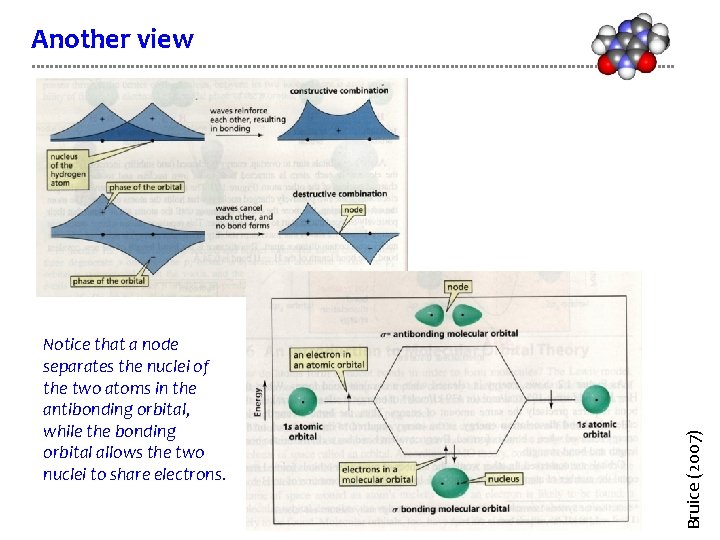
Notice that a node separates the nuclei of the two atoms in the antibonding orbital, while the bonding orbital allows the two nuclei to share electrons. Bruice (2007) Another view
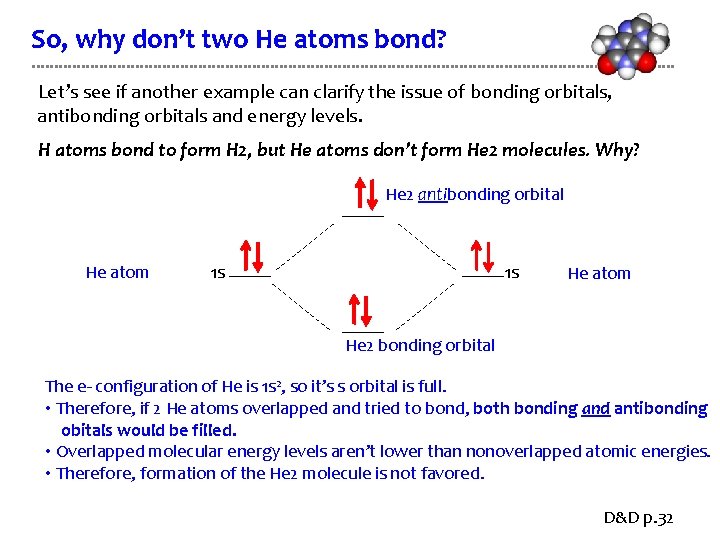
So, why don’t two He atoms bond? Let’s see if another example can clarify the issue of bonding orbitals, antibonding orbitals and energy levels. H atoms bond to form H 2, but He atoms don’t form He 2 molecules. Why? He 2 antibonding orbital He atom 1 s 1 s He atom He 2 bonding orbital The e- configuration of He is 1 s 2, so it’s s orbital is full. • Therefore, if 2 He atoms overlapped and tried to bond, both bonding and antibonding obitals would be filled. • Overlapped molecular energy levels aren’t lower than nonoverlapped atomic energies. • Therefore, formation of the He 2 molecule is not favored. D&D p. 32
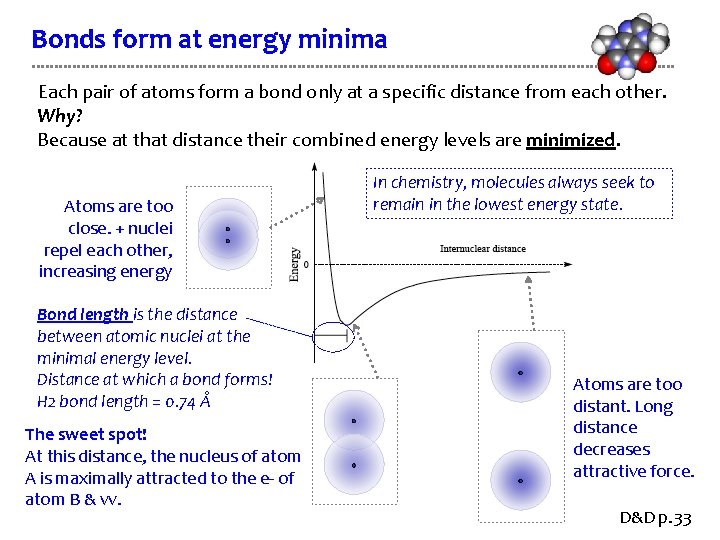
Bonds form at energy minima Each pair of atoms form a bond only at a specific distance from each other. Why? Because at that distance their combined energy levels are minimized. Atoms are too close. + nuclei repel each other, increasing energy Bond length is the distance between atomic nuclei at the minimal energy level. Distance at which a bond forms! H 2 bond length = 0. 74 Å The sweet spot! At this distance, the nucleus of atom A is maximally attracted to the e- of atom B & vv. In chemistry, molecules always seek to remain in the lowest energy state. Atoms are too distant. Long distance decreases attractive force. D&D p. 33
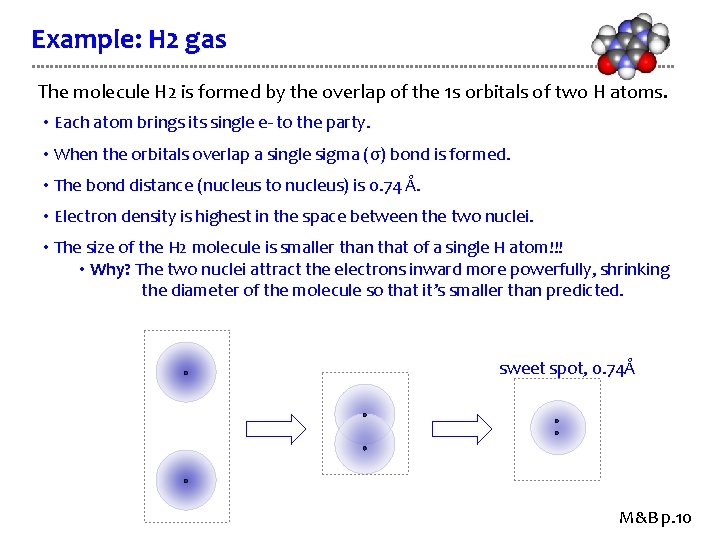
Example: H 2 gas The molecule H 2 is formed by the overlap of the 1 s orbitals of two H atoms. • Each atom brings its single e- to the party. • When the orbitals overlap a single sigma (σ) bond is formed. • The bond distance (nucleus to nucleus) is 0. 74 Å. • Electron density is highest in the space between the two nuclei. • The size of the H 2 molecule is smaller than that of a single H atom!!! • Why? The two nuclei attract the electrons inward more powerfully, shrinking the diameter of the molecule so that it’s smaller than predicted. sweet spot, 0. 74Å M&B p. 10

Shorter bonds are stronger bonds Generally, bond strength increases as bond length decreases. Bond length (nm) Bond energy (k. J/mol) C-F 0. 138 452 C - Cl 0. 177 339 C - Br 0. 194 280 C–I 0. 214 230 What does this mean for chlorofluorocarbon chemistry? Chlorofluorocarbons (CFCs) used to be used as refrigerants and aerosol propellants. The bonds between carbon and chlorine or fluorine are short and strong, so CFCs are not easily broken down; they are stable and don’t break down. When released into the atmosphere CFCs accumulate in the upper layers of the atmosphere. Exposure to UV light breaks CFC bonds and forms halogen radicals. The radicals react with ozone and break it down to oxygen gas. Loss of ozone then allows harmful levels of UV radiation to penetrate to the earth’s surface. H&P p. 11
- Slides: 11