CHE 118 Introduction CHE 118 1 Topics Syllabus

CHE 118 Introduction CHE 118 1

Topics • Syllabus, experiment schedule, student drawers, CHE 118 packet • Safety items and contract • Calibration scales • Calibration Curve and Spectrophotometer CHE 118 2

Safety Concerns • Additional safety concerns that were not mentioned on the safety contract will be discussed using another Power. Point file. CHE 118 3

Calibration Scales • Know how to read the calibration marks on a piece of calibrated glassware and determine: 1. the correct number of significant figures 2. the correct volume reading BCC You. Tube video CHE 118 4

Graduated Cylinder • 10 m. L and 100 m. L sizes are most common • 0. 0 m. L when empty, 10. 00 m. L when full (or 100. 0 m. L when full) In General: 1. Find difference between numbered tick marks 2. Use the un-numbered tick marks between the two numbered marks, count the number of jumps from one numbered mark to the other 3. Determine what each un-numbered mark is worth 4. Estimate one digit beyond the un-numbered tick marks (this is the last significant digit for the volume) 5. Read the volume, using the correct number of significant figures CHE 118 5

Graduated Cylinder (in student drawer) CHE 118 6

SF and Volume Reading • Difference between numbered calibration tick marks: 8 m. L – 7 m. L = 1 m. L • There are 9 un-numbered tick marks between the 7 and 8 marks (10 jumps from 7 to 8) • Each un-numbered tick mark is worth: 1 m. L / 10 jumps = 0. 1 m. L per jump • Estimate one digit beyond unnumbered mark 0. 1? m. L (use 2 digits after decimal point for SF, . _ _ m. L) CHE 118 7

Read a Volume with Correct SF • 10 m. L graduated cylinder • Read volume at the bottom of meniscus, at eye level • More than 7, less than 8 • 7. ? ? m. L • 7. 53 m. L CHE 118 8

Calibration Curve and Spectrophotometer Work in groups of four. Everyone must make their own calibration curve. CHE 118 9
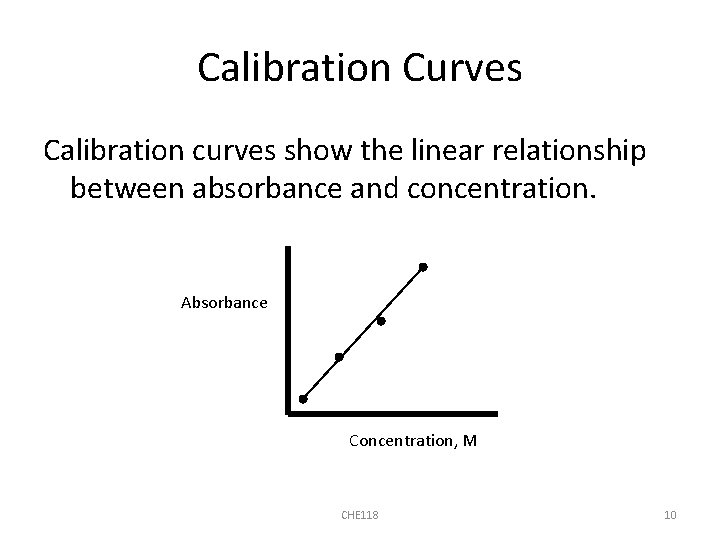
Calibration Curves Calibration curves show the linear relationship between absorbance and concentration. Absorbance Concentration, M CHE 118 10

Spectrophotometer An instrument that will indicate the amount of light (of one wavelength) absorbed by a sample slit LED readout Light source monochromator sample detector CHE 118 11

Beer – Lambert Law A = abc A is absorbance of light by the sample a is the molar extinction coefficient b is the solution path length c is the concentration of solute This is a linear relationship; y = mx + b A = mc + b CHE 118 12

Making the Calibration Curve • You will acquire an absorbance reading for each standard solution provided • Plot the corresponding concentration and absorbance values as data points on a graph to construct the calibration curve CHE 118 13
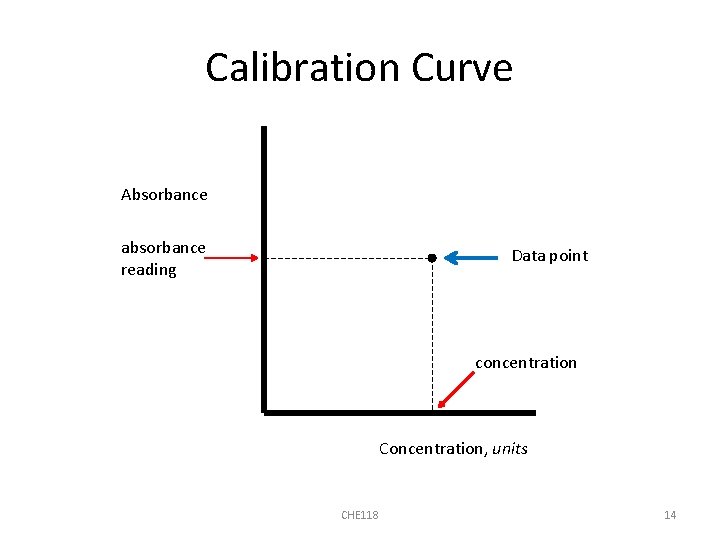
Calibration Curve Absorbance absorbance reading Data point concentration Concentration, units CHE 118 14

Find the Absorbance of the Unknown Solution 1. Use a disposable transfer pipet (plastic) to almost fill a small test tube with the unknown solution 2. Wipe the outside of the test tube (remove droplets and fingerprints) 3. Place this test tube in the sample compartment of the spectrophotometer (use the same spectrophotometer each time) 4. Record the absorbance readout from the spectrophotometer CHE 118 15

Find the Concentration of the Unknown Solution 1. You will have an absorbance reading for your solution (from the spectrophotometer). 2. Find that absorbance reading on the y axis of the calibration curve. 3. At that point on the y axis, move horizontally across to the calibration curve line (use a ruler) 4. When you reach the line, drop straight down to the x axis 5. The value that you are at on the x axis is the concentration of your solution CHE 118 16
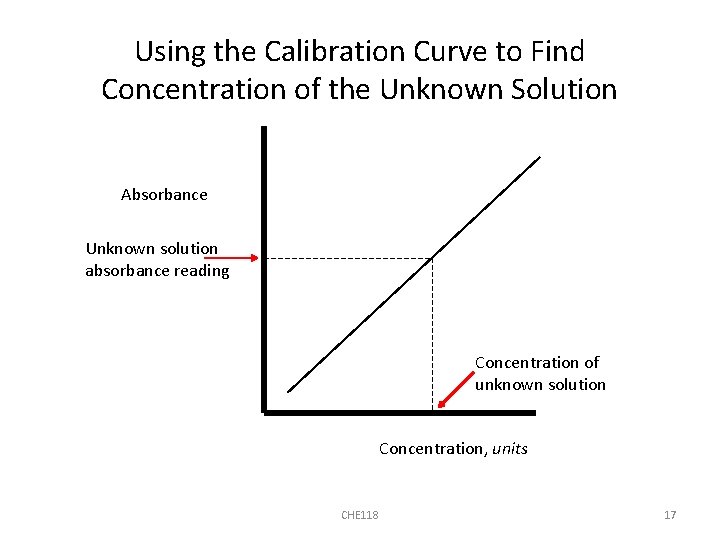
Using the Calibration Curve to Find Concentration of the Unknown Solution Absorbance Unknown solution absorbance reading Concentration of unknown solution Concentration, units CHE 118 17
- Slides: 17