Charge Orbital and Spin Ordering in RBa Fe

Charge, Orbital and Spin Ordering in RBa. Fe 2 O 5+x Perovskites Patrick M. Woodward Department of Chemistry Ohio State University Pavel Karen Department of Chemistry University of Oslo

Charge, Orbital and Spin Ordering Charge Ordering (TCO) – T > TCO Delocalized Electrons Single oxidation state – T < TCO Localized Electrons Distinct oxidation states Orbital Ordering (TOO) – Preferential (anisotropic) occupation of given d-orbitals – Cooperative Jahn-Teller distortion Spin Ordering (TN or TC) – Long range magnetic ordering Mixed Valency (Robin & Day Classification) – Type III Metallic e- transport Single oxidation state – Type II Activated e- transport Two oxidation states at a given instant, but CO pattern is fluctional – Type I Insulating Two oxidation states, with a regular (long range) CO pattern
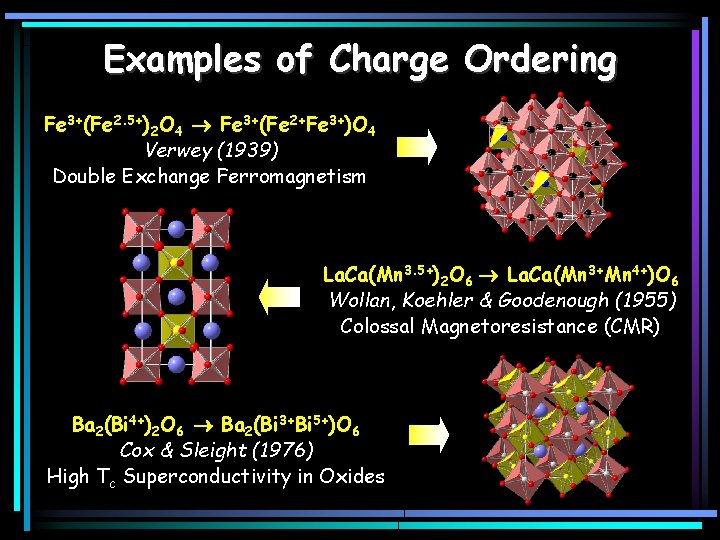
Examples of Charge Ordering Fe 3+(Fe 2. 5+)2 O 4 Fe 3+(Fe 2+Fe 3+)O 4 Verwey (1939) Double Exchange Ferromagnetism La. Ca(Mn 3. 5+)2 O 6 La. Ca(Mn 3+Mn 4+)O 6 Wollan, Koehler & Goodenough (1955) Colossal Magnetoresistance (CMR) Ba 2(Bi 4+)2 O 6 Ba 2(Bi 3+Bi 5+)O 6 Cox & Sleight (1976) High Tc Superconductivity in Oxides
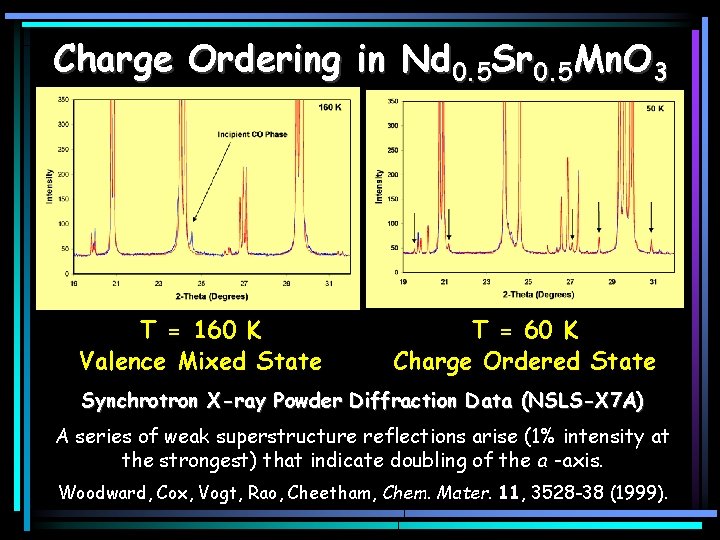
Charge Ordering in Nd 0. 5 Sr 0. 5 Mn. O 3 T = 160 K Valence Mixed State T = 60 K Charge Ordered State Synchrotron X-ray Powder Diffraction Data (NSLS-X 7 A) A series of weak superstructure reflections arise (1% intensity at the strongest) that indicate doubling of the a -axis. Woodward, Cox, Vogt, Rao, Cheetham, Chem. Mater. 11, 3528 -38 (1999).
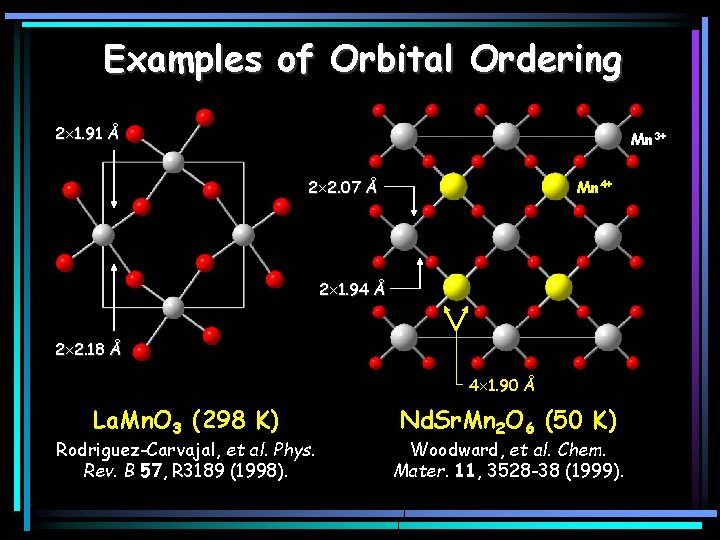
Examples of Orbital Ordering 2 1. 91 Å Mn 3+ 2 2. 07 Å Mn 4+ 2 1. 94 Å 2 2. 18 Å 4 1. 90 Å La. Mn. O 3 (298 K) Rodriguez-Carvajal, et al. Phys. Rev. B 57, R 3189 (1998). Nd. Sr. Mn 2 O 6 (50 K) Woodward, et al. Chem. Mater. 11, 3528 -38 (1999).
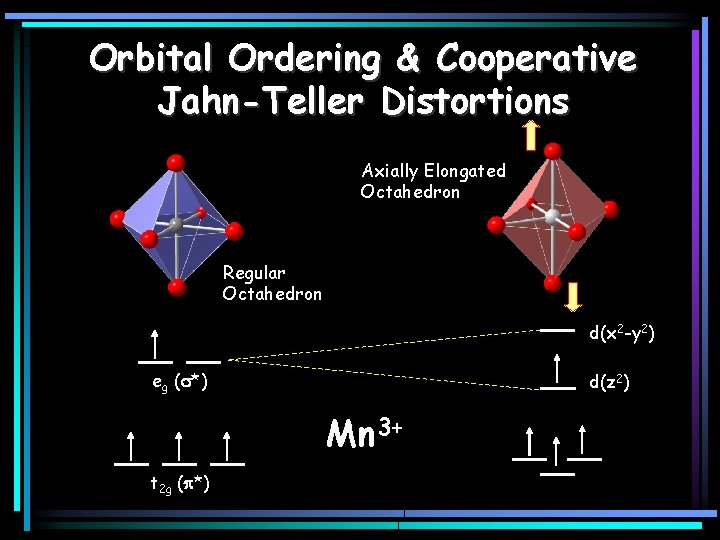
Orbital Ordering & Cooperative Jahn-Teller Distortions Axially Elongated Octahedron Regular Octahedron d(x 2 -y 2) eg (s*) d(z 2) Mn 3+ t 2 g (p*)
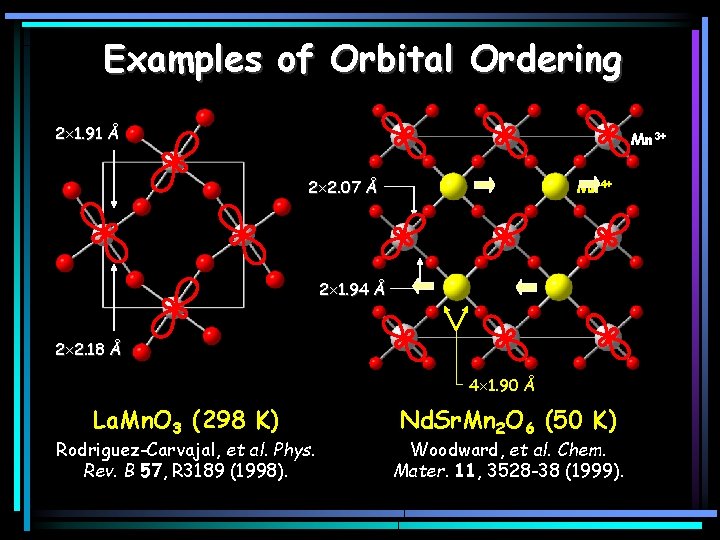
Examples of Orbital Ordering 2 1. 91 Å Mn 3+ 2 2. 07 Å Mn 4+ 2 1. 94 Å 2 2. 18 Å 4 1. 90 Å La. Mn. O 3 (298 K) Rodriguez-Carvajal, et al. Phys. Rev. B 57, R 3189 (1998). Nd. Sr. Mn 2 O 6 (50 K) Woodward, et al. Chem. Mater. 11, 3528 -38 (1999).

Orbital Ordering in Nd 0. 5 Sr 0. 5 Mn. O 3 Upon cooling below 150 K, the a & c-axes expand the b-axis contracts. This is the signature of orbital ordering Woodward, Cox, Vogt, Rao, Cheetham, Chem. Mater. 11, 3528 -38 (1999).
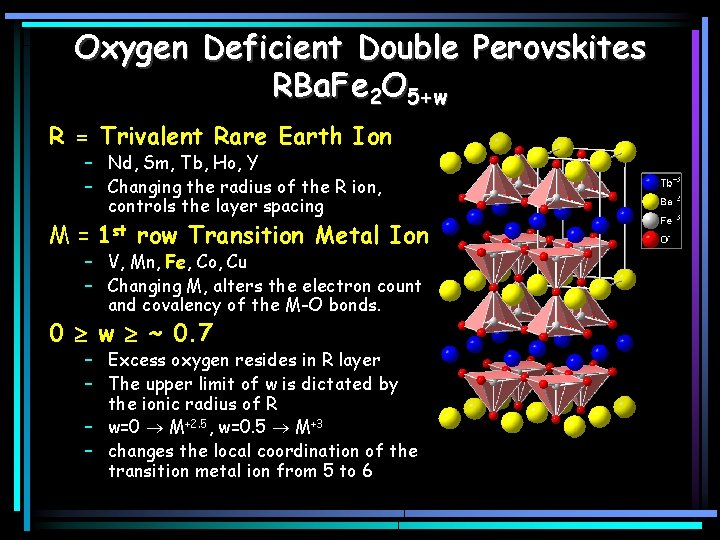
Oxygen Deficient Double Perovskites RBa. Fe 2 O 5+w R = Trivalent Rare Earth Ion – Nd, Sm, Tb, Ho, Y – Changing the radius of the R ion, controls the layer spacing M = 1 st row Transition Metal Ion – V, Mn, Fe, Co, Cu – Changing M, alters the electron count and covalency of the M-O bonds. 0 w ~ 0. 7 – Excess oxygen resides in R layer – The upper limit of w is dictated by the ionic radius of R – w=0 M+2. 5, w=0. 5 M+3 – changes the local coordination of the transition metal ion from 5 to 6

Synthesis REBa. Fe 2 O 5 RE 2 O 3 + Citric Acid Fe 2 O 3 + Nitric Acid Heat Ba. CO 3 & Stir 1. Amorphous Organic. Inorganic Precursor The sample is dehydrated at ~180 °C. 2. The sample is then heated at ~400 °C to drive off the organic content and produce an amorphous precursor. 3. The precursor is calcined (800 -900 °C) and then sintered at high temperature (1000 -1150 °C) in a carefully controlled p. O 2 atmosphere. 4. The sintered pellets are heated at a lower temperature (600860 °C) in a controlled atmosphere to attain the desired oxygen content.

Powder Diffraction Data Collection • Synchrotron X-ray Powder Diffraction – NSLS - X 7 A (Dave Cox, Tom Vogt) – NSLS – X 3 B (Peter Stephens, Sylvina Pagola) – ESRF – BM 1 B (Swiss-Norwegian Beamline) • Neutron Powder Diffraction – – NIST – BT 1 (Brian Toby) - YBa. Fe 2 O 5 ILL – D 2 B (Emmanuel Suard) - Ho. Ba. Fe 2 O 5 & Nd. Ba. Fe 2 O 5 PSI - HRPT (Peter Fischer) - Tb. Ba. Fe 2 O 5 ANSTO – MRPD (Andrew Studer) - Tb. Ba. Fe 2 O 5 • Neutron Thermodiffractometry – ILL – D 20 (Emmanuel Suard) - Ho. Ba. Fe 2 O 5 & Nd. Ba. Fe 2 O 5 • Mossbauer Spectroscopy – Abo Akademi (Johan Linden)
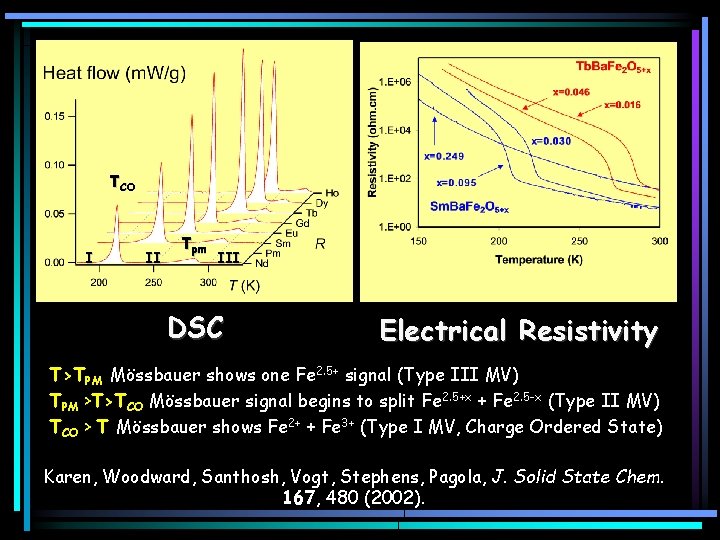
TCO I II Tpm III DSC Electrical Resistivity T>TPM Mössbauer shows one Fe 2. 5+ signal (Type III MV) TPM >T>TCO Mössbauer signal begins to split Fe 2. 5+x + Fe 2. 5 -x (Type II MV) TCO > T Mössbauer shows Fe 2+ + Fe 3+ (Type I MV, Charge Ordered State) Karen, Woodward, Santhosh, Vogt, Stephens, Pagola, J. Solid State Chem. 167, 480 (2002).
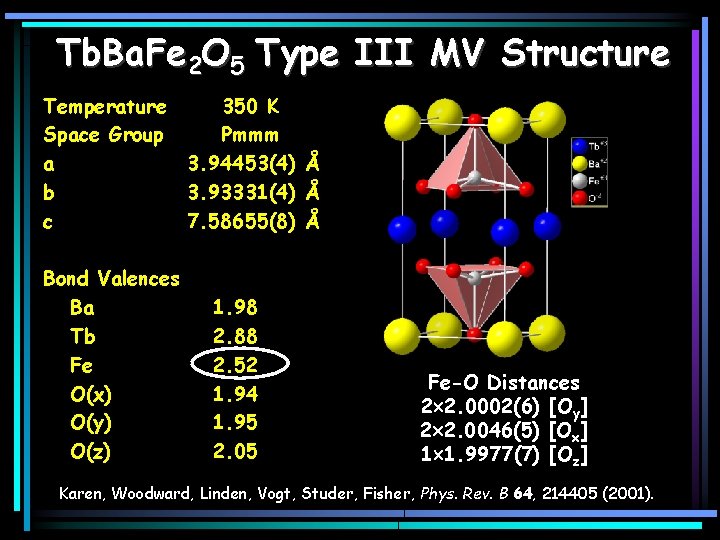
Tb. Ba. Fe 2 O 5 Type III MV Structure Temperature Space Group a b c Bond Valences Ba Tb Fe O(x) O(y) O(z) 350 K Pmmm 3. 94453(4) Å 3. 93331(4) Å 7. 58655(8) Å 1. 98 2. 88 2. 52 1. 94 1. 95 2. 05 Fe-O Distances 2 2. 0002(6) [Oy] 2 2. 0046(5) [Ox] 1 1. 9977(7) [Oz] Karen, Woodward, Linden, Vogt, Studer, Fisher, Phys. Rev. B 64, 214405 (2001).
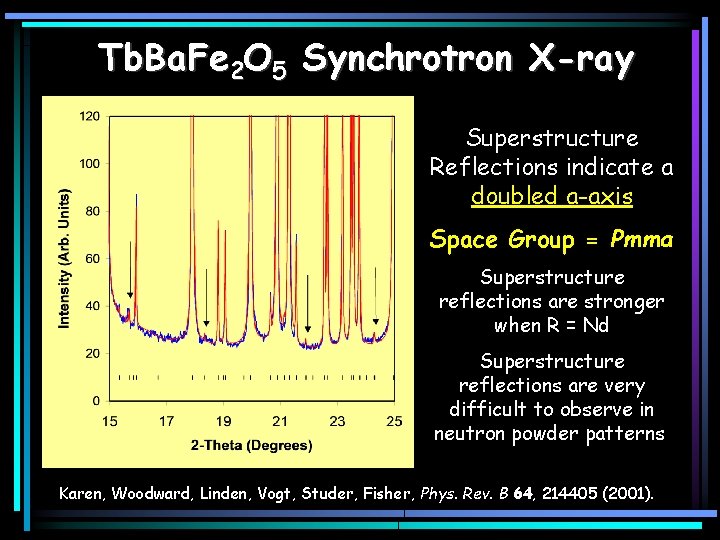
Tb. Ba. Fe 2 O 5 Synchrotron X-ray Superstructure Reflections indicate a doubled a-axis Space Group = Pmma Superstructure reflections are stronger when R = Nd Superstructure reflections are very difficult to observe in neutron powder patterns. Karen, Woodward, Linden, Vogt, Studer, Fisher, Phys. Rev. B 64, 214405 (2001).
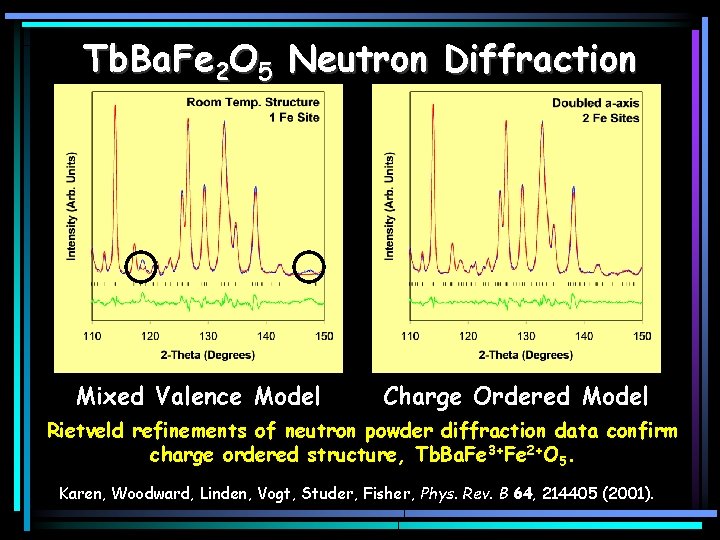
Tb. Ba. Fe 2 O 5 Neutron Diffraction Mixed Valence Model Charge Ordered Model Rietveld refinements of neutron powder diffraction data confirm charge ordered structure, Tb. Ba. Fe 3+Fe 2+O 5. Karen, Woodward, Linden, Vogt, Studer, Fisher, Phys. Rev. B 64, 214405 (2001).
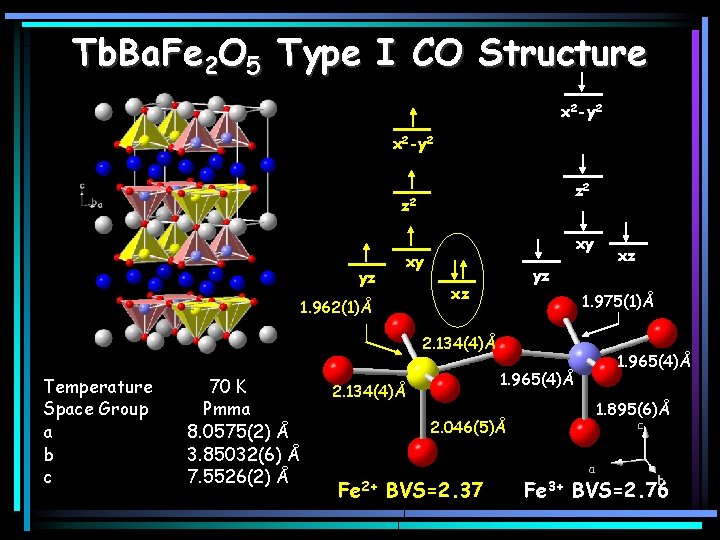
Tb. Ba. Fe 2 O 5 Type I CO Structure x 2 -y 2 z 2 yz 1. 962(1)Å xy xy yz xz 1. 975(1)Å 2. 134(4)Å Temperature Space Group a b c 70 K Pmma 8. 0575(2) Å 3. 85032(6) Å 7. 5526(2) Å 1. 965(4)Å 2. 134(4)Å 2. 046(5)Å Fe 2+ BVS=2. 37 xz 1. 965(4)Å 1. 895(6)Å Fe 3+ BVS=2. 76
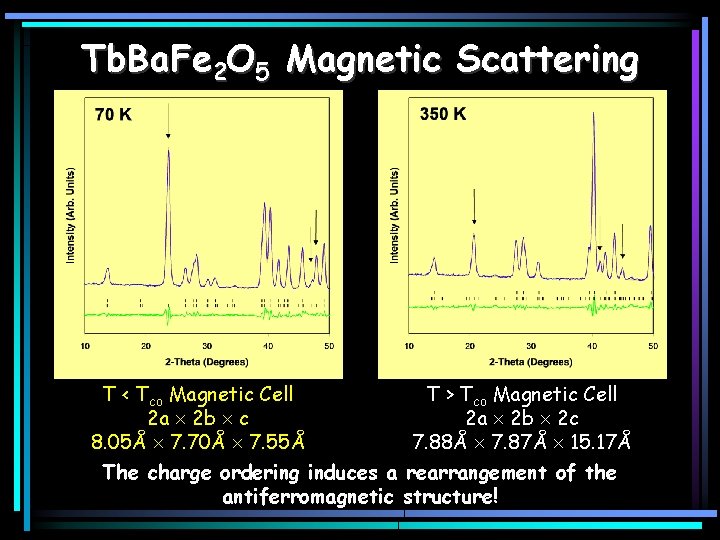
Tb. Ba. Fe 2 O 5 Magnetic Scattering T < Tco Magnetic Cell 2 a 2 b c 8. 05Å 7. 70Å 7. 55Å T > Tco Magnetic Cell 2 a 2 b 2 c 7. 88Å 7. 87Å 15. 17Å The charge ordering induces a rearrangement of the antiferromagnetic structure!
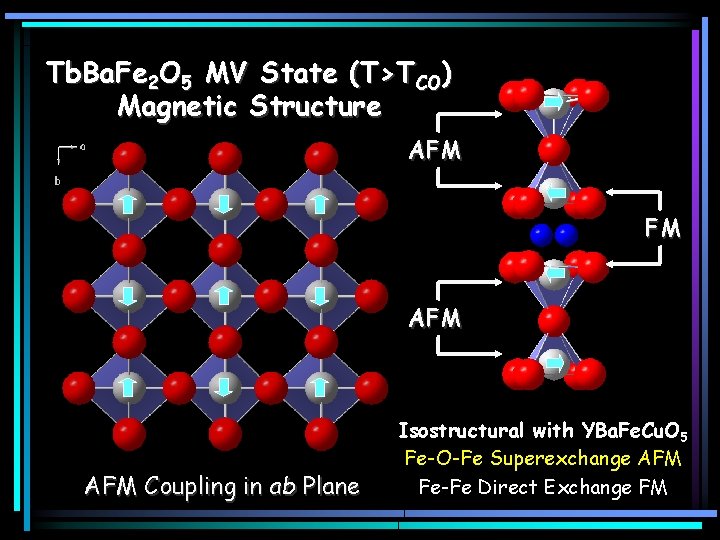
Tb. Ba. Fe 2 O 5 MV State (T>TCO) Magnetic Structure AFM FM AFM Coupling in ab Plane Isostructural with YBa. Fe. Cu. O 5 Fe-O-Fe Superexchange AFM Fe-Fe Direct Exchange FM
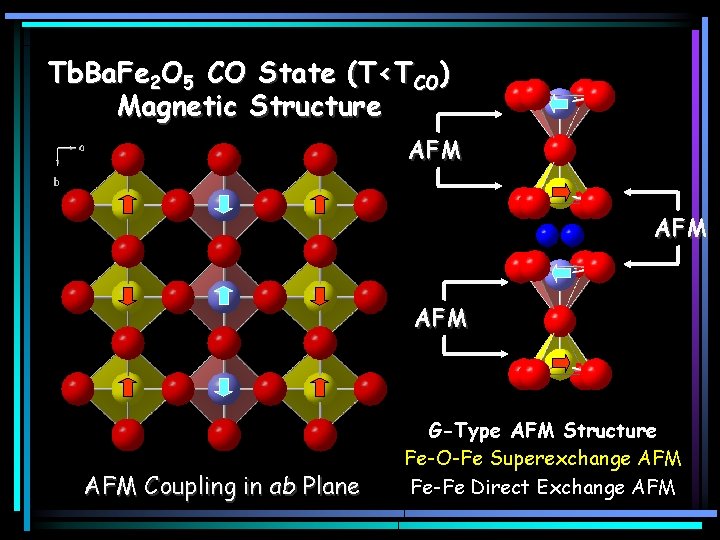
Tb. Ba. Fe 2 O 5 CO State (T<TCO) Magnetic Structure AFM AFM Coupling in ab Plane G-Type AFM Structure Fe-O-Fe Superexchange AFM Fe-Fe Direct Exchange AFM
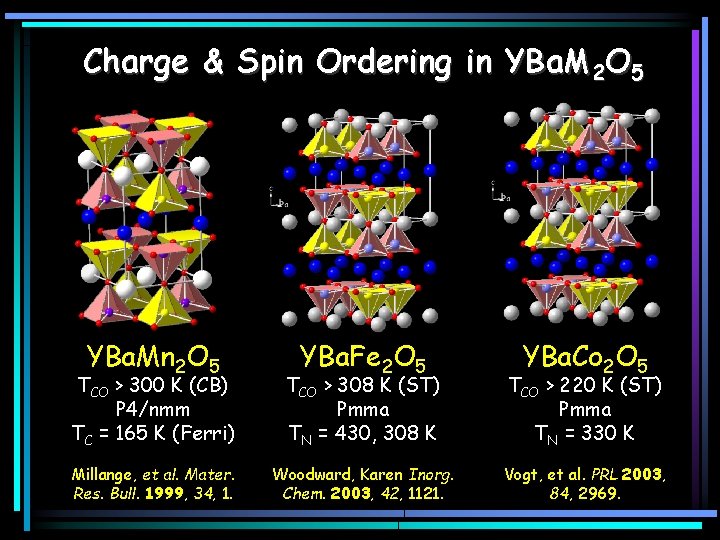
Charge & Spin Ordering in YBa. M 2 O 5 YBa. Mn 2 O 5 YBa. Fe 2 O 5 YBa. Co 2 O 5 TCO > 300 K (CB) P 4/nmm TC = 165 K (Ferri) TCO > 308 K (ST) Pmma TN = 430, 308 K TCO > 220 K (ST) Pmma TN = 330 K Millange, et al. Mater. Res. Bull. 1999, 34, 1. Woodward, Karen Inorg. Chem. 2003, 42, 1121. Vogt, et al. PRL 2003, 84, 2969.
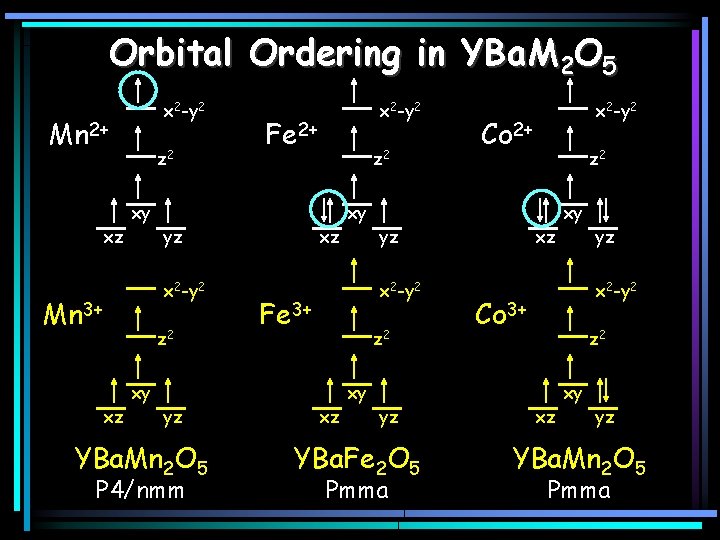
Orbital Ordering in YBa. M 2 O 5 x 2 -y 2 Mn 2+ xz z 2 xy Mn 3+ z 2 xz xy Fe 2+ yz x 2 -y 2 yz YBa. Mn 2 O 5 P 4/nmm x 2 -y 2 z 2 xz xy Fe 3+ z 2 xz xy Co 2+ yz x 2 -y 2 yz YBa. Fe 2 O 5 Pmma x 2 -y 2 z 2 xz xy yz x 2 -y 2 Co 3+ z 2 xz xy yz YBa. Mn 2 O 5 Pmma

Phase Transitions – RBa. Fe 2 O 5 • Paramagnetic to Antiferromagnetic (430 -450 K) – YBa. Fe. Cu. O 5 Type AFM Order – Small Magnetostrictive coupling leads to a subtle Tetragonal to Orthorhombic Distortion • Premonitory Charge Ordering (290 -330 K) – Subtle charge localization can be seen in DSC & Mossbauer, but not in diffraction measurements – Mixed valency changes from Type I to Type II • Long Range Charge Ordering (240 -290 K) – Induces a large orbital ordering transition – Orbital ordering stabilizes a stripe CO pattern – Stabilizes G-type Antiferromagnetic order (changes the sign of the Fe-Fe direct exchange)
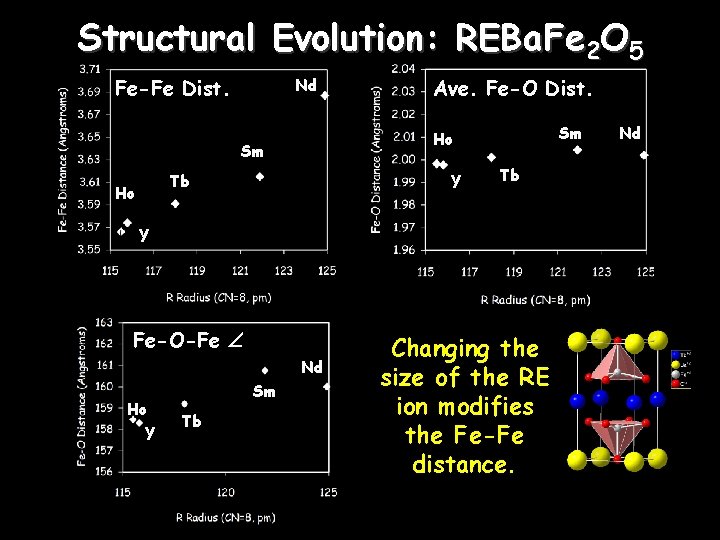
Structural Evolution: REBa. Fe 2 O 5 Fe-Fe Dist. Nd Sm Ho Sm Tb Ho Ave. Fe-O Dist. Y Tb Y Fe-O-Fe Nd Ho Y Sm Tb Changing the size of the RE ion modifies the Fe-Fe distance. Nd

Phase Transitions vs. R size Type III Mixed Valent TCO Type I Charge Ordered Type II As the radius of the R ion increases –TCO (MV II MV I) decreases significantly. –TPM (MV III MV II) decreases more gradually. –TN 1 changes very little.
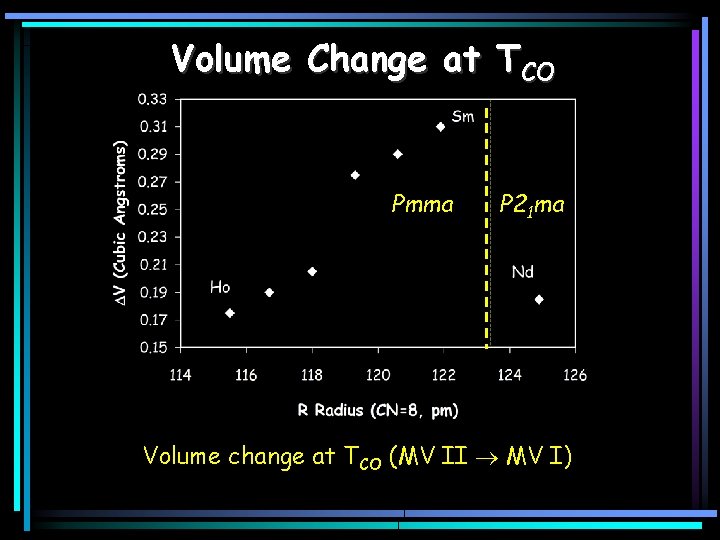
Volume Change at TCO Pmma P 21 ma Volume change at TCO (MV II MV I)
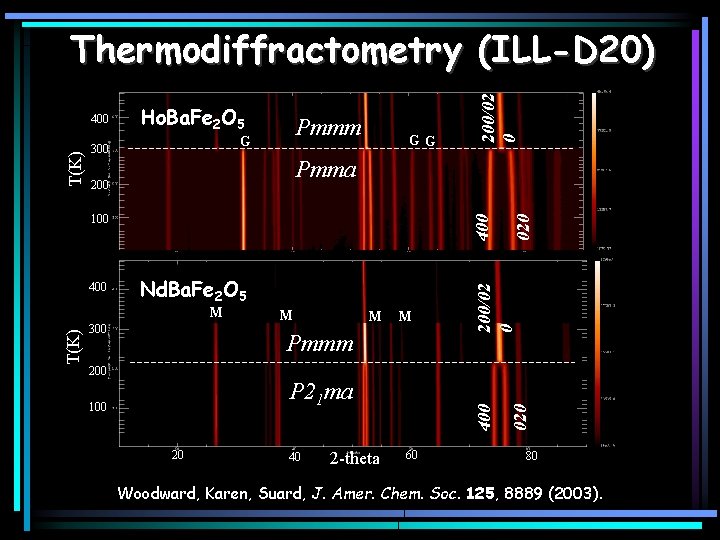
Ho. Ba. Fe 2 O 5 Pmmm G 300 GG T(K) M 300 M M M Pmmm 200 P 21 ma 100 20 40 2 -theta 60 020 Nd. Ba. Fe 2 O 5 400 400 100 020 Pmma 200/02 0 T(K) 400 200/02 0 Thermodiffractometry (ILL-D 20) 80 Woodward, Karen, Suard, J. Amer. Chem. Soc. 125, 8889 (2003).
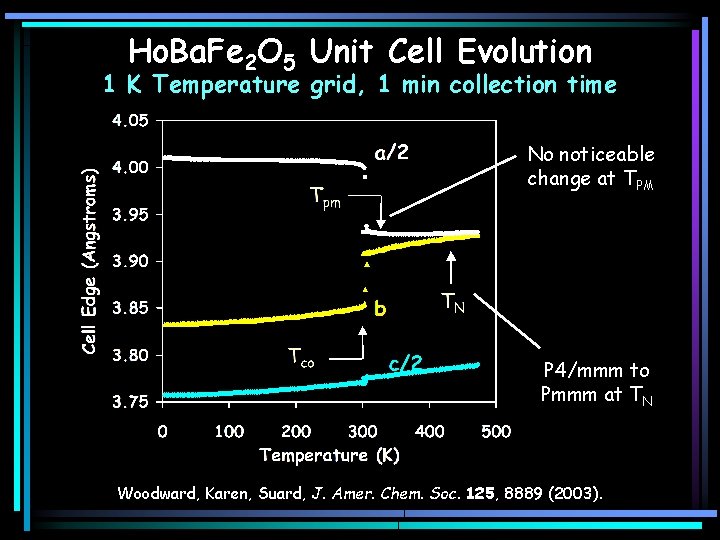
Ho. Ba. Fe 2 O 5 Unit Cell Evolution 1 K Temperature grid, 1 min collection time No noticeable change at TPM P 4/mmm to Pmmm at TN Woodward, Karen, Suard, J. Amer. Chem. Soc. 125, 8889 (2003).
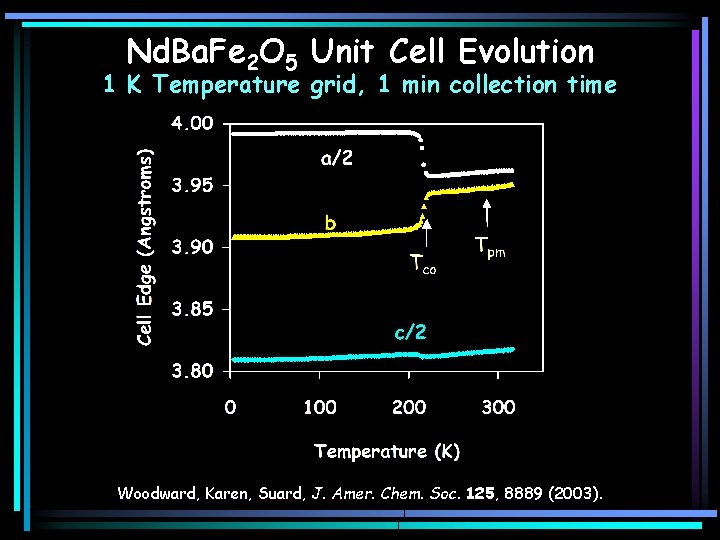
Nd. Ba. Fe 2 O 5 Unit Cell Evolution 1 K Temperature grid, 1 min collection time Woodward, Karen, Suard, J. Amer. Chem. Soc. 125, 8889 (2003).
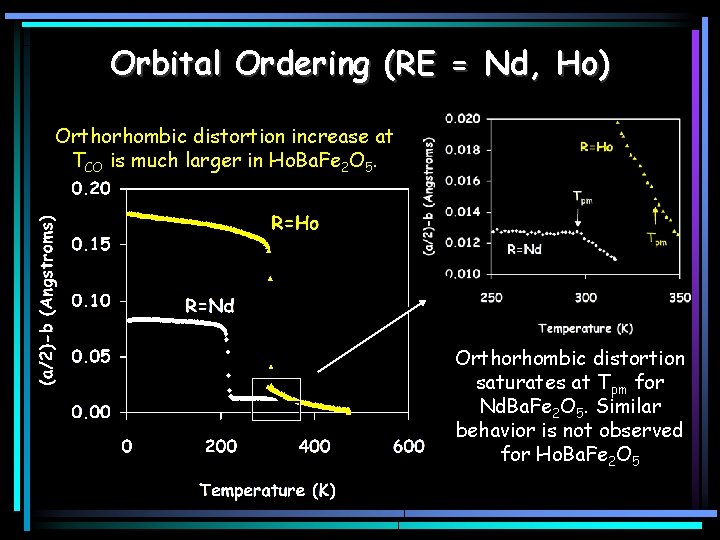
Orbital Ordering (RE = Nd, Ho) Orthorhombic distortion increase at TCO is much larger in Ho. Ba. Fe 2 O 5. Orthorhombic distortion saturates at Tpm for Nd. Ba. Fe 2 O 5. Similar behavior is not observed for Ho. Ba. Fe 2 O 5
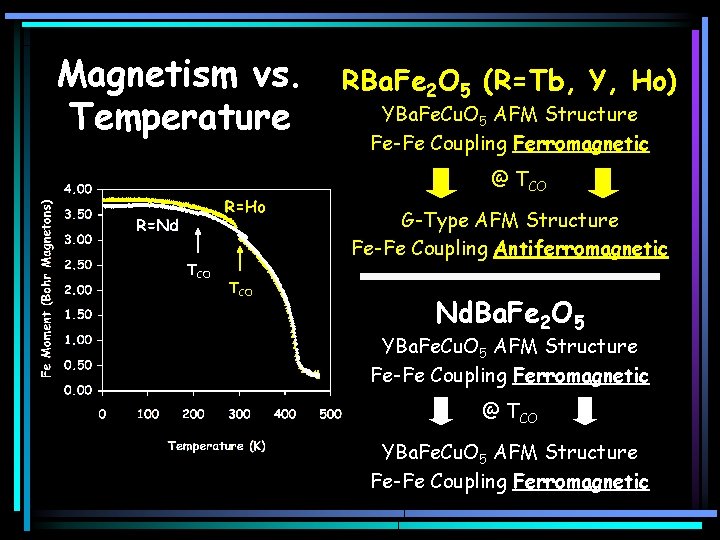
Magnetism vs. Temperature RBa. Fe 2 O 5 (R=Tb, Y, Ho) YBa. Fe. Cu. O 5 AFM Structure Fe-Fe Coupling Ferromagnetic @ TCO G-Type AFM Structure Fe-Fe Coupling Antiferromagnetic TCO Nd. Ba. Fe 2 O 5 YBa. Fe. Cu. O 5 AFM Structure Fe-Fe Coupling Ferromagnetic @ TCO YBa. Fe. Cu. O 5 AFM Structure Fe-Fe Coupling Ferromagnetic
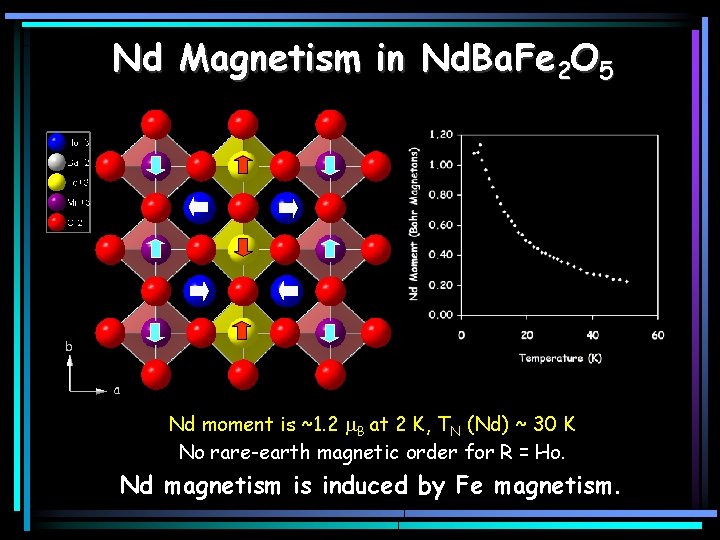
Nd Magnetism in Nd. Ba. Fe 2 O 5 Nd moment is ~1. 2 m. B at 2 K, TN (Nd) ~ 30 K No rare-earth magnetic order for R = Ho. Nd magnetism is induced by Fe magnetism.

Structural Tuning in RBa. Fe 2 O 5 As the radius of the R ion increases (R = Ho-Sm) – – The spacing across the R-layer increases TCO decreases significantly, TPM decreases more gradually TN 1 changes very little Patterns of charge, orbital and spin order remain constant Nd. Ba. Fe 2 O 5 The large size of Nd has several effects – Disrupts the ideal pattern of orbital ordering • The CO structure has P 21 ma symmetry rather than Pmma • The volume change at TCO is anomalous • The orthorhombic distortion parameter saturates at T PM – Decouples the magnetic and charge order • There is no rearrangement of the magnetic structure at T CO – Destabilizes the long range charge order • TCO is much lower than other members of the series
- Slides: 32