Characterization of weakly bound HCF 3 OCS H

Characterization of weakly bound HCF 3 OCS, H 2 CF 2 -OCS, H 3 CF-OCS, and HCF 3 CO 2 van der Waals complexes by ab initio calculations and microwave spectroscopy Michal M. Serafin, Sean A. Peebles Department of Chemistry, 600 Lincoln Avenue, Eastern Illinois University, Charleston, IL 61920 USA

Introduction • Systematic study of fluorinated methane molecules complexed with OCS and CO 2 • Good prototype systems that display weak interactions • Four projects included thus far: Ø HCF 3 -OCS (M. M. Serafin, S. A. Peebles, J. Phys. Chem. A. , 2006, 110, 11938) Ø H 2 CF 2 -OCS (Normal and 2 isotopomers assigned, tentative structure assigned) Ø H 3 CF-OCS (Manuscript in preparation) Ø HCF 3 -CO 2 (A and E states assigned for normal isotopomer, barrier to rotation determined) • Similar structures for all low energy complexes obtained from ab initio calculations
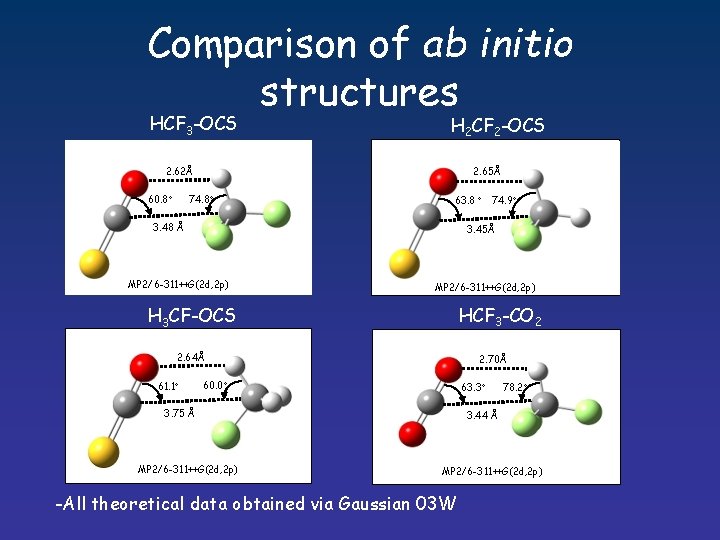
Comparison of ab initio structures HCF 3 -OCS H 2 CF 2 -OCS 2. 62Å 2. 65Å 74. 8 60. 8 63. 8 74. 9 3. 48 Å 3. 45Å MP 2/6 -311++G(2 d, 2 p) HCF 3 -CO 2 H 3 CF-OCS 2. 64Å 61. 1 2. 70Å 60. 0 63. 3 3. 75 Å MP 2/6 -311++G(2 d, 2 p) 78. 2 3. 44 Å MP 2/6 -311++G(2 d, 2 p) -All theoretical data obtained via Gaussian 03 W

Experimental Details • Balle-Flygare type Fourier-transform microwave (FTMW) spectrometer, General Valve (Series 9), operating on the 5 -16 GHz region • Sample bulb contains approx. 1. 5% each gas under investigation, filled to a backing pressure of ~1. 5 -2. 5 bar of He/Ne • Stark effect experiments performed to acquire dipole data
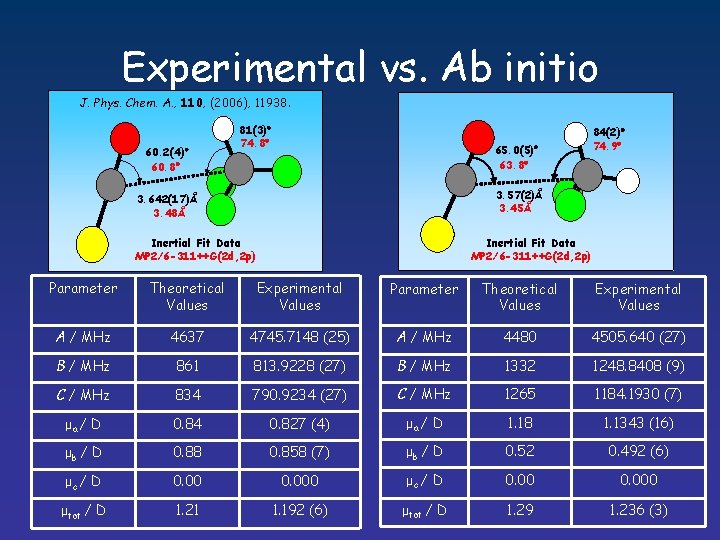
Experimental vs. Ab initio J. Phys. Chem. A. , 110, (2006), 11938. 60. 2(4) 60. 8 81(3) 74. 8 84(2) 74. 9 65. 0(5) 63. 8 3. 57(2)Å 3. 45Å 3. 642(17)Å 3. 48Å Inertial Fit Data MP 2/6 -311++G(2 d, 2 p) Parameter Theoretical Values Experimental Values A / MHz 4637 4745. 7148 (25) A / MHz 4480 4505. 640 (27) B / MHz 861 813. 9228 (27) B / MHz 1332 1248. 8408 (9) C / MHz 834 790. 9234 (27) C / MHz 1265 1184. 1930 (7) μa / D 0. 84 0. 827 (4) μa / D 1. 18 1. 1343 (16) μb / D 0. 88 0. 858 (7) μb / D 0. 52 0. 492 (6) μc / D 0. 000 μtot / D 1. 21 1. 192 (6) μtot / D 1. 29 1. 236 (3)
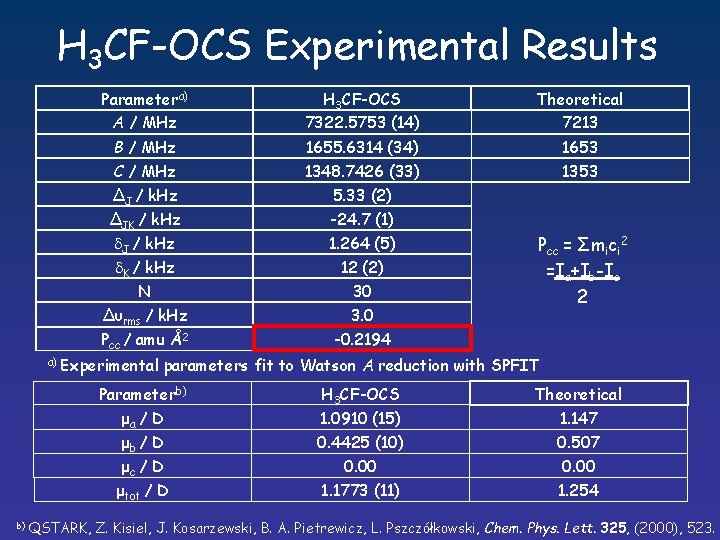
H 3 CF-OCS Experimental Results Parametera) A / MHz H 3 CF-OCS 7322. 5753 (14) Theoretical 7213 B / MHz C / MHz 1655. 6314 (34) 1348. 7426 (33) 1653 1353 ∆J / k. Hz 5. 33 (2) ∆JK / k. Hz J / k. Hz 1. 264 (5) N ∆υrms / k. Hz 30 3. 0 K / k. Hz 12 (2) Pcc / amu Å2 -0. 2194 a) Experimental Pcc = Σmici 2 =Ia+Ib-Ic 2 parameters fit to Watson A reduction with SPFIT Parameterb) μa / D μb / D μc / D μtot / D b) QSTARK, -24. 7 (1) H 3 CF-OCS 1. 0910 (15) Theoretical 1. 147 0. 4425 (10) 0. 507 0. 00 1. 1773 (11) 0. 00 1. 254 Z. Kisiel, J. Kosarzewski, B. A. Pietrewicz, L. Pszczółkowski, Chem. Phys. Lett. 325, (2000), 523.
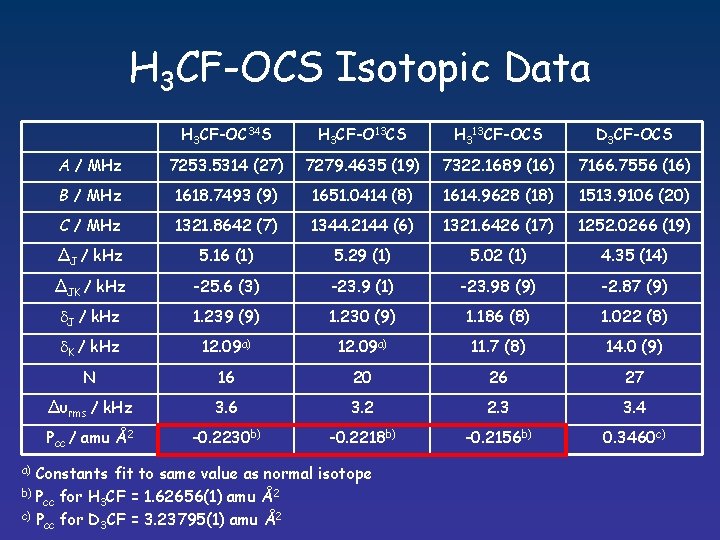
H 3 CF-OCS Isotopic Data H 3 CF-OC 34 S H 3 CF-O 13 CS H 313 CF-OCS D 3 CF-OCS A / MHz 7253. 5314 (27) 7279. 4635 (19) 7322. 1689 (16) 7166. 7556 (16) B / MHz 1618. 7493 (9) 1651. 0414 (8) 1614. 9628 (18) 1513. 9106 (20) C / MHz 1321. 8642 (7) 1344. 2144 (6) 1321. 6426 (17) 1252. 0266 (19) ∆J / k. Hz 5. 16 (1) 5. 29 (1) 5. 02 (1) 4. 35 (14) ∆JK / k. Hz -25. 6 (3) -23. 9 (1) -23. 98 (9) -2. 87 (9) J / k. Hz 1. 239 (9) 1. 230 (9) 1. 186 (8) 1. 022 (8) K / k. Hz 12. 09 a) 11. 7 (8) 14. 0 (9) N 16 20 26 27 ∆υrms / k. Hz 3. 6 3. 2 2. 3 3. 4 Pcc / amu Å2 -0. 2230 b) -0. 2218 b) -0. 2156 b) 0. 3460 c) Constants fit to same value as normal isotope b) P for H CF = 1. 62656(1) amu Å2 cc 3 c) P for D CF = 3. 23795(1) amu Å2 cc 3 a)
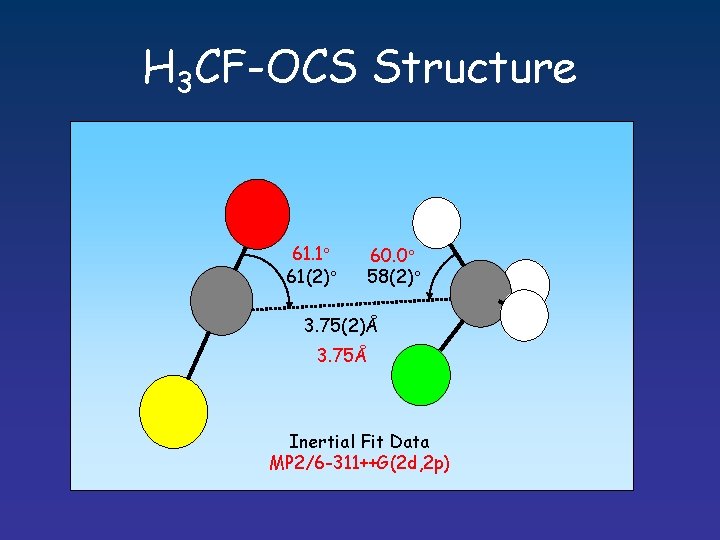
H 3 CF-OCS Structure 61. 1 61(2) 60. 0 58(2) 3. 75(2)Å 3. 75Å Inertial Fit Data MP 2/6 -311++G(2 d, 2 p)

HCF 3 -CO 2 Spectrum and Dipole Data • Near prolate asymmetric top = -0. 9967 • Spectrum split into A and E states • Forbidden b-type transitions occur despite μb = 0. 00 • V 3 = 0. 355(4) k. J mol-1 • Ia = 80. 4(1) amu Å2 • Ic(monomer)a) = 89. 23(2) amu Å2 423← 322 422← 321 A A E 8918. 5 E 8919. 5 8920. 5 Frequency / MHz Dipole Component Experimental Value Theoretical Value μa / D μb / D μc / D μtot / D 0. 431 (6) 0. 00 1. 436 (6) 1. 499 (6) 0. 399 0. 00 1. 556 1. 606 a)W. Caminati, J. C. López, J. L. Alonso, J. U. Grabow, Angew. Chem. Int. Ed. , 44, (2005), 3840.
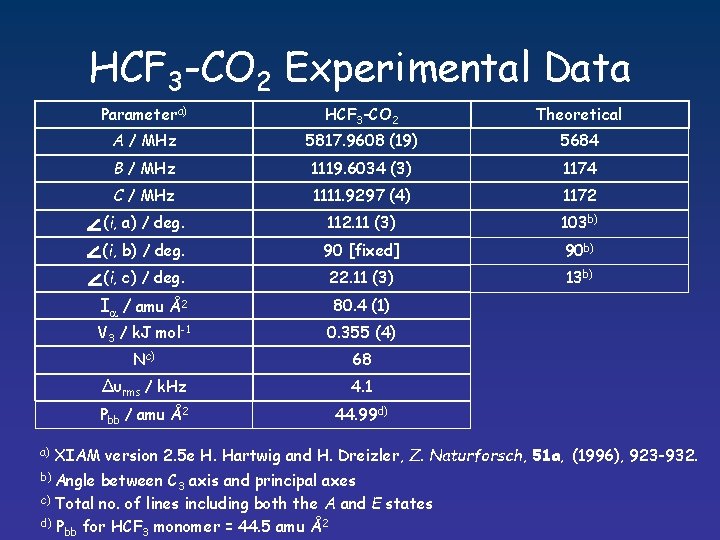
HCF 3 -CO 2 Experimental Data a) Parametera) HCF 3 -CO 2 Theoretical A / MHz 5817. 9608 (19) 5684 B / MHz 1119. 6034 (3) 1174 C / MHz 1111. 9297 (4) 1172 (i, a) / deg. 112. 11 (3) 103 b) (i, b) / deg. 90 [fixed] 90 b) (i, c) / deg. 22. 11 (3) 13 b) Ia / amu Å2 80. 4 (1) V 3 / k. J mol-1 0. 355 (4) Nc) 68 ∆υrms / k. Hz 4. 1 Pbb / amu Å2 44. 99 d) XIAM version 2. 5 e H. Hartwig and H. Dreizler, Z. Naturforsch, 51 a, (1996), 923 -932. b) Angle between C 3 axis and principal axes c) Total no. of lines including both the A and E states d) P 2 bb for HCF 3 monomer = 44. 5 amu Å

Conclusions • Lowest energy ab initio structures similar four complexes studied • Experimental structures of H 3 CF-OCS and HCF 3 -OCS obtained • HCF 3 -CO 2 barrier (V 3=0. 355(4) k. J mol-1) is relatively low compared to other complexes involving HCF 3 Ø H 3 CF-HCF 3 V 3=0. 840(5) k. J mol-1 (Angew. Chem. Int. Ed. , 44, (2005), 3840. ) Ø Thiirane-HCF 3 V 3=0. 526(3) k. J mol-1 (Chem. Phys. Lett. , 402, (2005), 4. ) Ø Oxirane-HCF 3 V 3=0. 55(1) k. J mol-1 (J. Am. Chem. Soc. , 126, (2004), 3244. ) • Isotopic substitution required for H 2 CF 2 -OCS and HCF 3 -CO 2

Acknowledgements • Dr. R. A. Peebles • American Chemical Society, Petroleum Research Fund, PRF #39752 -GB 6
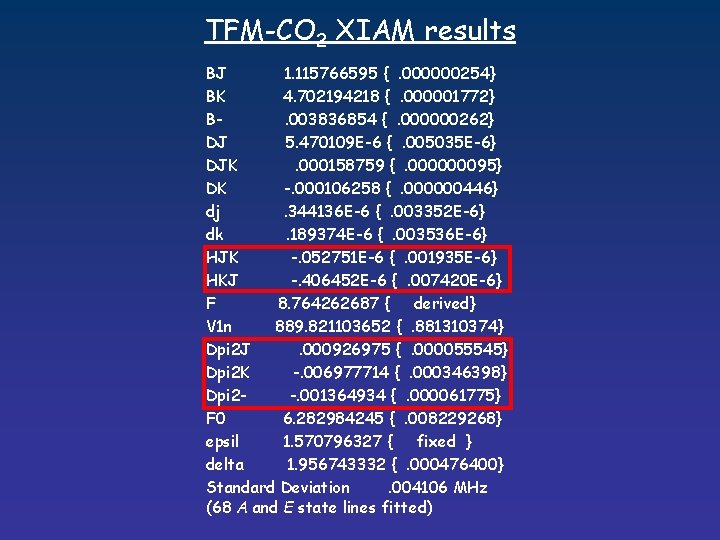
TFM-CO 2 XIAM results BJ 1. 115766595 {. 000000254} BK 4. 702194218 {. 000001772} B. 003836854 {. 000000262} DJ 5. 470109 E-6 {. 005035 E-6} DJK. 000158759 {. 000000095} DK -. 000106258 {. 000000446} dj. 344136 E-6 {. 003352 E-6} dk. 189374 E-6 {. 003536 E-6} HJK -. 052751 E-6 {. 001935 E-6} HKJ -. 406452 E-6 {. 007420 E-6} F 8. 764262687 { derived} V 1 n 889. 821103652 {. 881310374} Dpi 2 J. 000926975 {. 000055545} Dpi 2 K -. 006977714 {. 000346398} Dpi 2 -. 001364934 {. 000061775} F 0 6. 282984245 {. 008229268} epsil 1. 570796327 { fixed } delta 1. 956743332 {. 000476400} Standard Deviation. 004106 MHz (68 A and E state lines fitted)
- Slides: 13