CHARACTERISTIC INTRINSIC PROPERTIES Atomic Chemistry Physical Properties Can

















- Slides: 21

CHARACTERISTIC INTRINSIC PROPERTIES Atomic Chemistry

Physical Properties • Can be observed or measured without changing the make -up of the matter of the object • Are used to describe the object • Two categories: • non-characterisic • characteristic

Physical Properties • Examples Include: • Appearance • Texture • Color • Odor • Melting Point • Boiling Point • Density • Solubility

Non-Characteristic Properties (NCP) • A physical or chemical property that is NOT unique to one particular substance. • Can be used to describe MANY substances

NCP Examples • Temperature • Mass • Shape • Color • Volume • Acidity and Alkalinity (p. H)

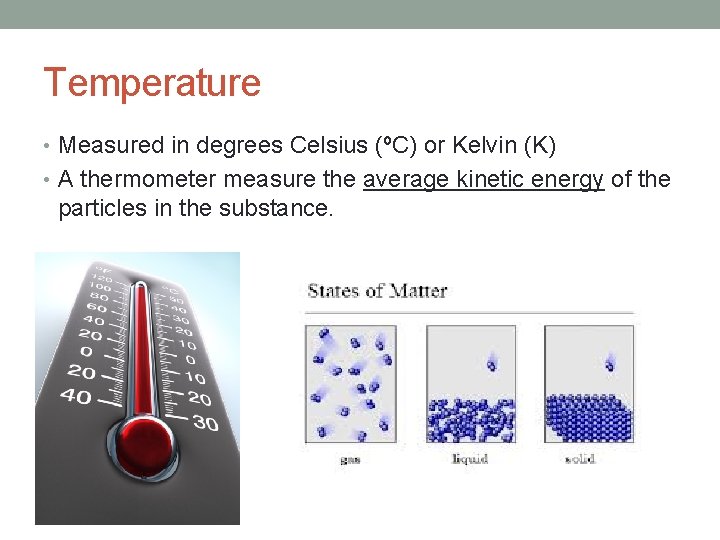
Temperature • Measured in degrees Celsius (ºC) or Kelvin (K) • A thermometer measure the average kinetic energy of the particles in the substance.

Mass

Shape and Color

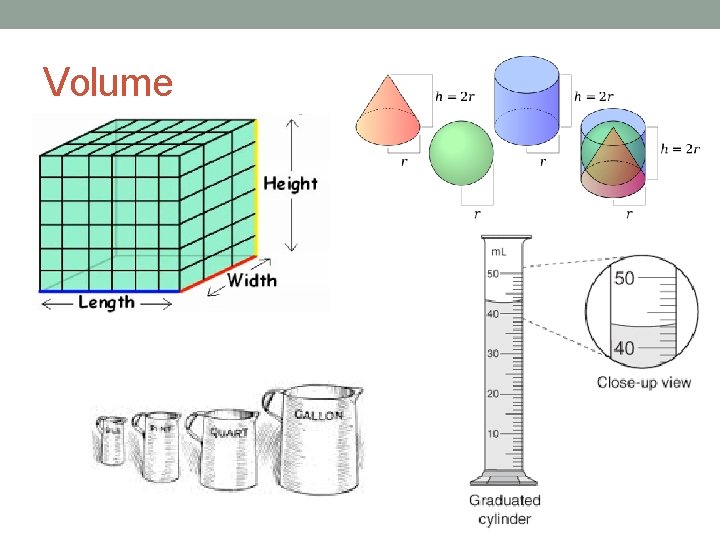
Volume

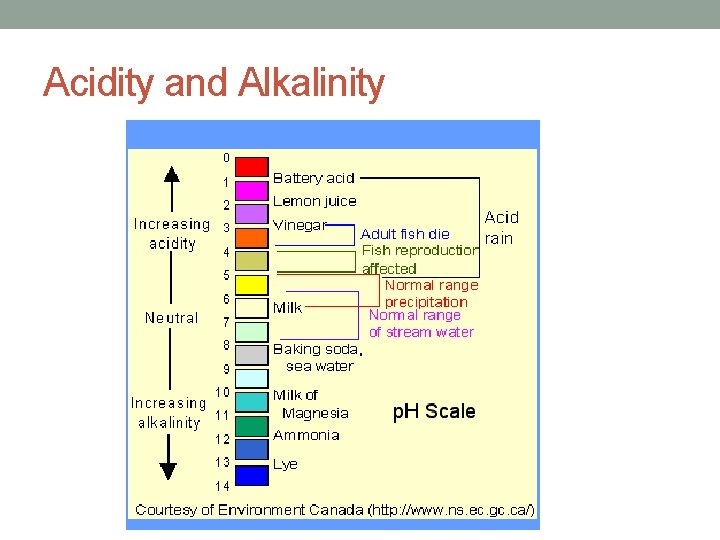
Acidity and Alkalinity

Characteristic Intrinsic Properties • A physical or chemical property that IS unique to a particular substance. • CAN be used to identify a substance

CP Examples • Density: the ratio of mass to volume (how much matter is in a given space) • Magnetism: The force of attraction between a magnet and a magnetic object • Solubility: How well a substance can dissolve in another substance. • Melting Point: Temperature at which substance changes from solid to liquid • Boiling Point: Temperature at which substance changes from liquid to gas

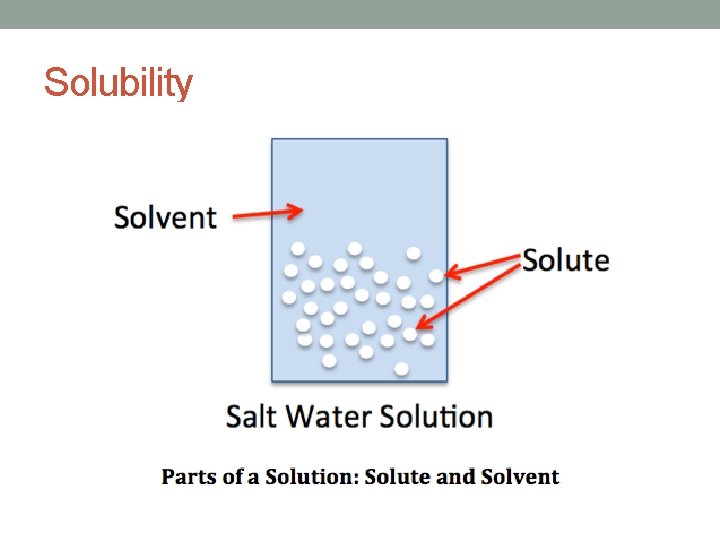
Solubility • A measure of how well a substance can dissolve in another substance • The solute: the substance that is DISSOLVED • The solvent: the substance that does the DISSOLVING • The solution: the result of mixing a solute and a solvent

Solubility

Melting & Boiling Points • Freezing point of H 2 O: 0ºC • Liquid water will freeze to solid ice at 0ºC • Melting point of ice: 0ºC • Solid ice will melt to liquid water at 0ºC • Boiling Point of H 2 O: 100ºC • Liquid water will change into gas at 100ºC

Example • The English Oak • Characteristic Property • Density = 720 kg/m 3 • NCP • Light yellow to brown in color • 25 -30 meters tall

Example #1 • Which of the following properties is an example of a characteristic property? 1. 2. 3. 4. Mass of 346 g Red in color and circular in shape Temperature of 25ºC Boiling Point of 204ºC

Example 2 • Identify ALL of the NCP below: 1. Volume of 200 m. L 2. Square shaped 3. Temperature of 45ºC 4. Freezing point of 204ºC 5. Density of 24 g/m. L

Example 3 • Julie has an unknown substance in a beaker. Its properties are described in the chart to the right. • Using the chart below, identify the mystery substance. Density Color Magnetic Iron 7. 86 g/m. L Yellow/gray Yes Water 1 g/m. L Colorless No Nitrogen 0. 00125 g/m. L Yellow No Sulphur 2. 07 g/m. L Yellow No Mass 103. 5 g Color Yellow Magnetic No Volume 50 m. L


What is the Atomic Theory? • Click here for video!