Chapters 4 5 Learning Targets Describe characteristics of

Ø Ø Ø Ø Ø Chapters 4 & 5: Learning Targets Describe characteristics of a wave (wavelength, frequency, energy) Explain how wave characteristics are related Explain how light and different colors of light are produced Define the electromagnetic spectrum Explain Bohr models including their advantages and disadvantages Determine the electron configuration and/or orbital diagram of any atom or ion; use these to determine groups, periods and identities of elements along with things such as number of unpaired electrons Define the periodic trends and be able to compare to elements and explain their difference with regards to these trends Describe the role of probability in orbital theory Determine if an ion for a given element will be a cation or anion and explain how each type of ion is formed

Ø Ø Ø Ø Ø Chapters 4 & 5: Key Words Ground state Excited state Wavelength Frequency Energy Photon Electromagnetic spectrum Energy level Orbital Valence electron Ø Ø Ø Core electron Alkali metal Alkaline earth metal Halogen Actinide Lanthanide Atomic radius Ionization energy Electronegativity Transition element Main-group element

Section 1: Properties of Light Pages 91 -97 RBQs #1 b, 1 c, 6, 8 Ø For a chemical reaction to occur, the atoms and/or molecules involved must physically collide with each other Ø If two atoms react, which part of the atoms will interact with each other first? Ø The structure and arrangement of _____________ therefore, is the key to understanding chemical reactivity
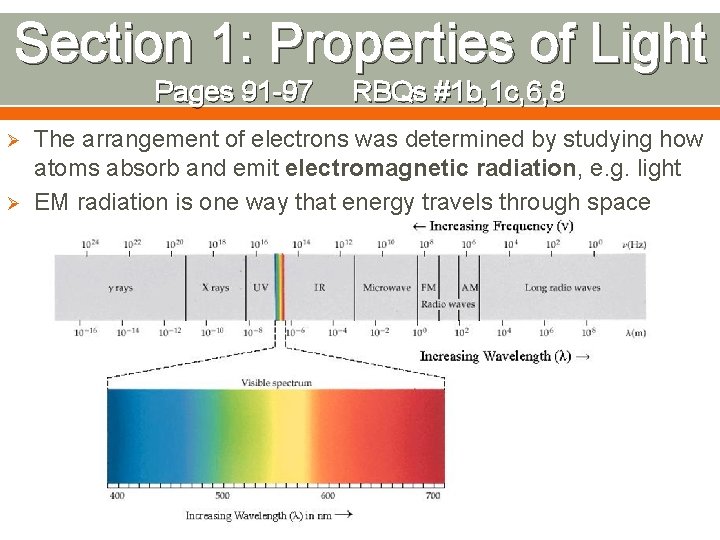
Section 1: Properties of Light Pages 91 -97 Ø Ø RBQs #1 b, 1 c, 6, 8 The arrangement of electrons was determined by studying how atoms absorb and emit electromagnetic radiation, e. g. light EM radiation is one way that energy travels through space

Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø EM radiation has 3 characteristics: speed, wavelength, and frequency Ø The speed of light (c), is a constant for all forms of EM radiation in a vacuum Ø We will always assume that EM radiation is moving through a vacuum Ø c = 3. 0 x 108 m/s
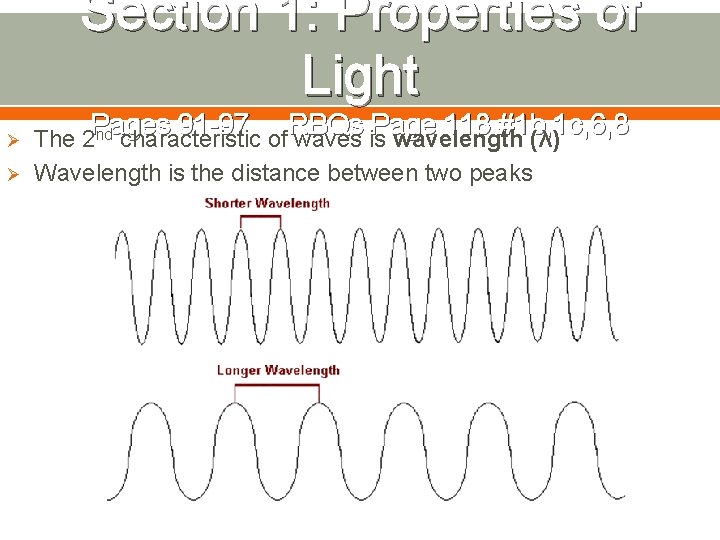
Section 1: Properties of Light Ø Ø Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 The 2 nd characteristic of waves is wavelength (λ) Wavelength is the distance between two peaks
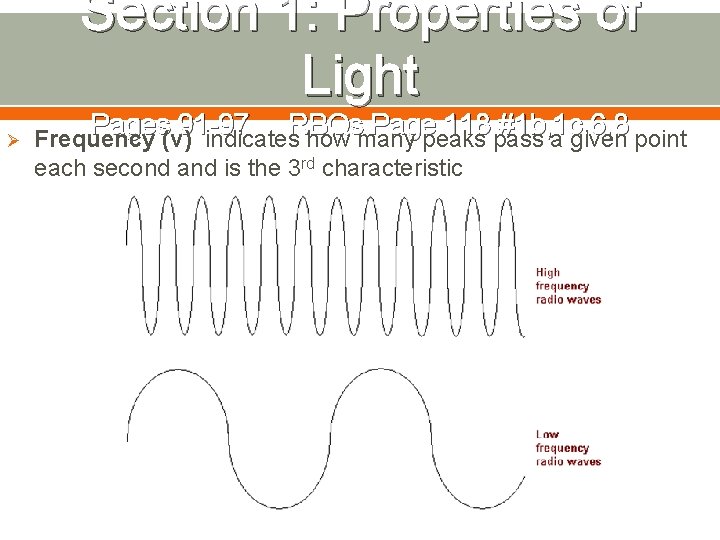
Section 1: Properties of Light Ø Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Frequency (ν) indicates how many peaks pass a given point each second and is the 3 rd characteristic
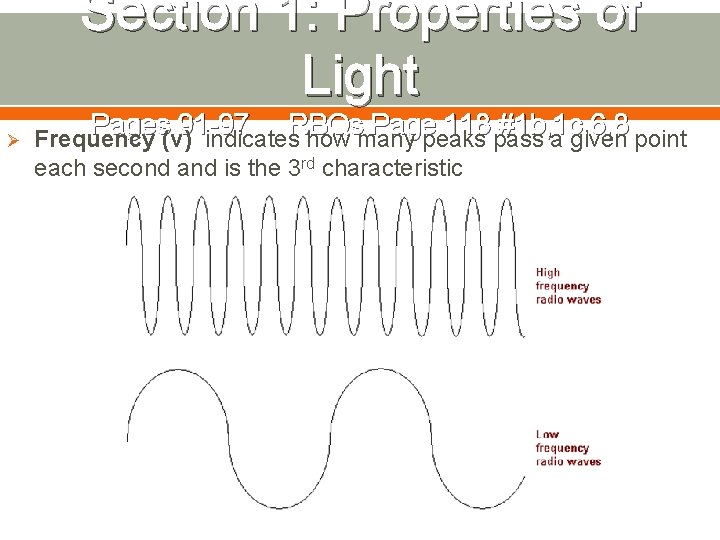
Section 1: Properties of Light Ø Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Frequency (ν) indicates how many peaks pass a given point each second and is the 3 rd characteristic

Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø The characteristics are related by the following equation: Ø Be careful with units! Make sure everybody matches up! Given that c is a constant, what kind of relationship does this show between wavelength and frequency? Ø

Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø The energy of the radiation is also related to the frequency of the radiation Ø The fun part is that light behave like a wave, but it also shows behaviors characteristic of particles, in this case called photons Ø The equation below gives the energy of EM radiation, and is equal to the energy of one single photon; QUANTIZED!

Calculate the frequency of light with a wavelength of 6. 50 x 102 nm. Also, determine the unit. 11 A. B. C. D. 1. 95 x 10 4. 61 x 105 1. 95 x 102 4. 62 x 1014

What is the increment of energy (the quantum) that is emitted at 4. 50 x 102 nm? Determine the unit, also. 4. 42 x 10 -19 B. 4. 42 x 10 -28 C. 8. 95 x 10 -32 D. 1. 01 x 1048 A.
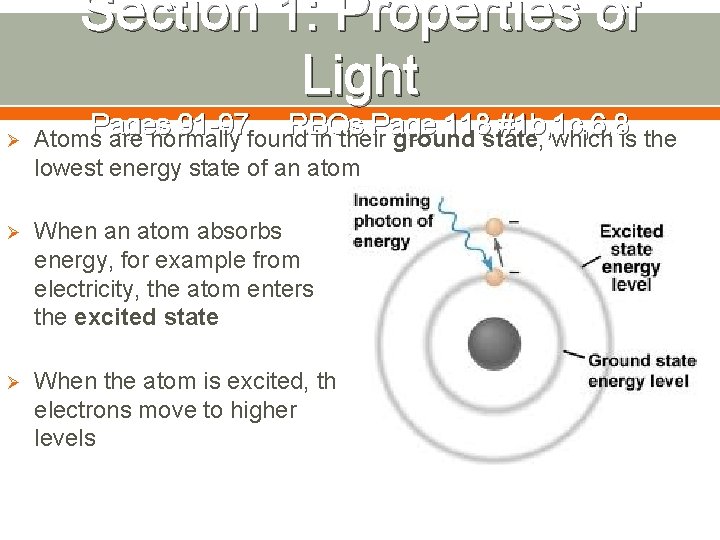
Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø Atoms are normally found in their ground state, which is the lowest energy state of an atom Ø When an atom absorbs energy, for example from electricity, the atom enters the excited state Ø When the atom is excited, the electrons move to higher levels
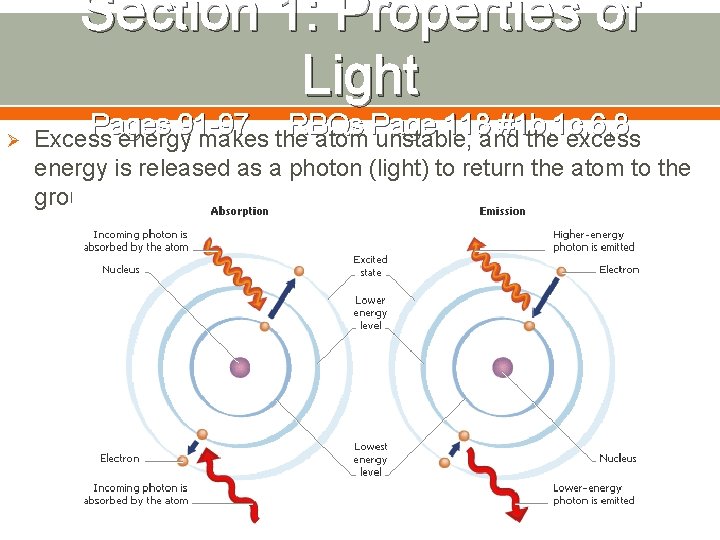
Section 1: Properties of Light Ø Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Excess energy makes the atom unstable, and the excess energy is released as a photon (light) to return the atom to the ground state
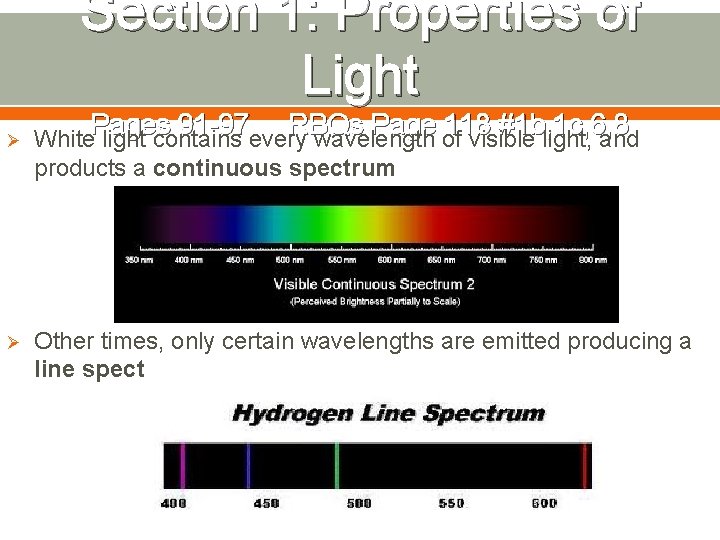
Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø White light contains every wavelength of visible light, and products a continuous spectrum Ø Other times, only certain wavelengths are emitted producing a line spectrum

Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø Substances emit different wavelengths because they absorb and emit different amounts of energy due to differing numbers and arrangements of electrons Ø Each substance has its own unique line spectrum Ø These can be used to identify unknown substances
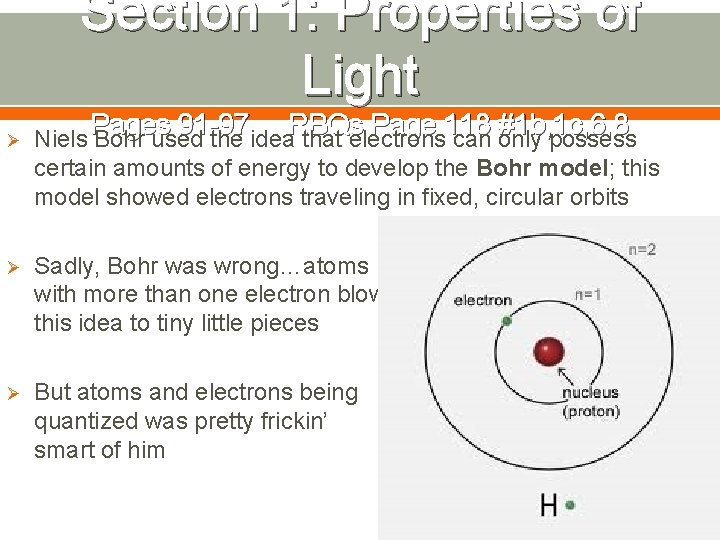
Section 1: Properties of Light Pages 91 -97 RBQs Page 118 #1 b, 1 c, 6, 8 Ø Niels Bohr used the idea that electrons can only possess certain amounts of energy to develop the Bohr model; this model showed electrons traveling in fixed, circular orbits Ø Sadly, Bohr was wrong…atoms with more than one electron blow this idea to tiny little pieces Ø But atoms and electrons being quantized was pretty frickin’ smart of him

Section 2: Orbitals Page 100 Ø A key development in understanding the structure of the atom was Schrodinger’s wave equation, which lead to quantum mechanics. . . Heisenberg helped a little, too. . . Ø Solving this equation leads to a series of wave functions, which is essentially a set of x, y, and z coordinates Each point graphed as a result of these coordinates indicates the possible location of an electron Ø

Section 2: Orbitals Page 100 Ø Graphing a lot of these points gives a map of the probable location of an electron; the exact location of an electron is pretty much impossible to determine Ø Each point gives a possible electron location; more dots means a higher probability of an electron being there Ø The area of space in which an electron is most likely to be found is called an orbital
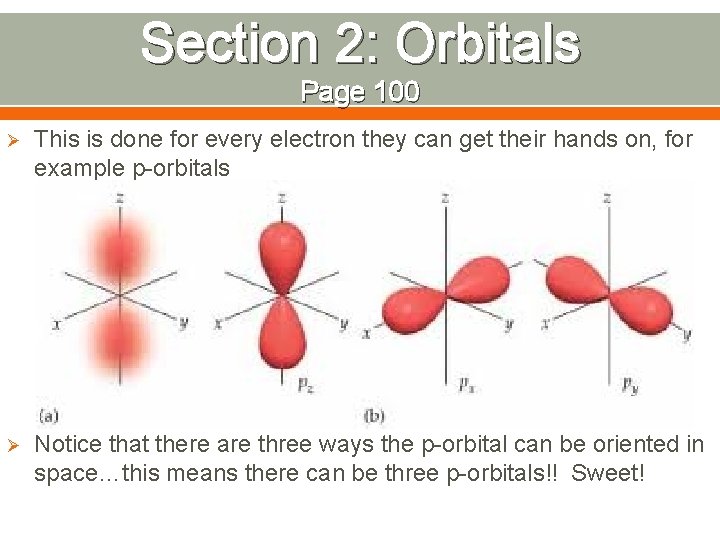
Section 2: Orbitals Page 100 Ø This is done for every electron they can get their hands on, for example p-orbitals Ø Notice that there are three ways the p-orbital can be oriented in space…this means there can be three p-orbitals!! Sweet!
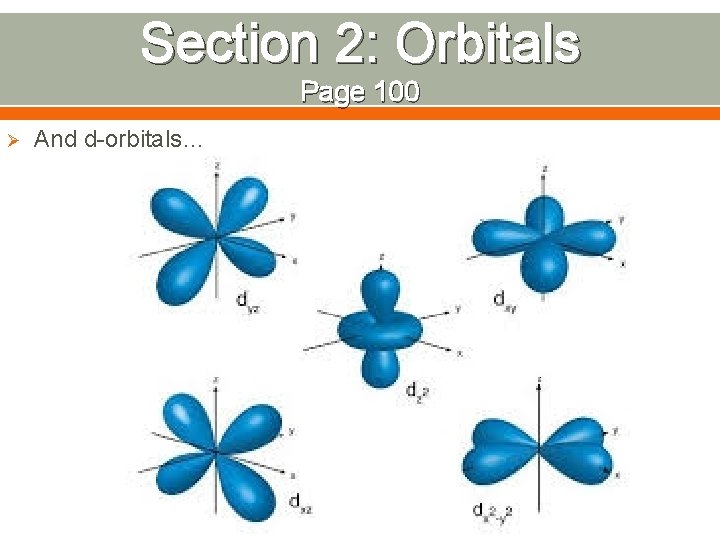
Section 2: Orbitals Page 100 Ø And d-orbitals…
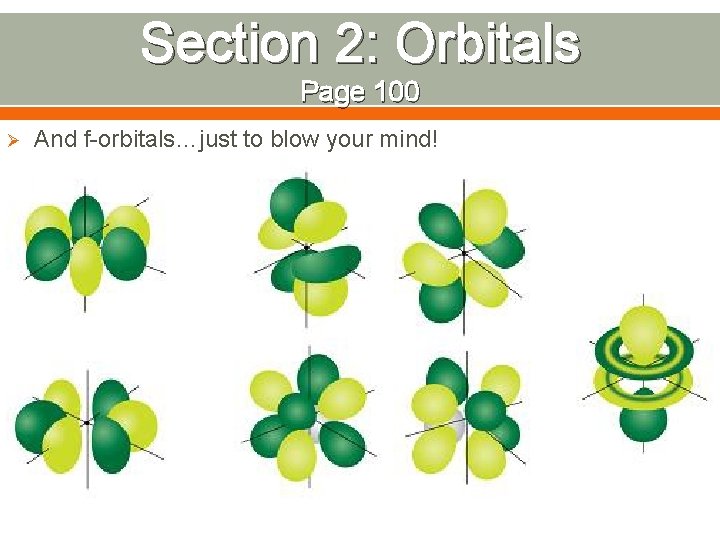
Section 2: Orbitals Page 100 Ø And f-orbitals…just to blow your mind!

Section 2: Orbitals Page 100 Ø Each orbital (or area of space) can hold a maximum of 2 electrons. Ø One or more orbitals combine to make sublevels or subshells Ø The sublevels consist of different shapes of orbitals, and are designated using letters Ø The four different shapes of orbitals are s, p, d, and f Ø So an f-sublevel consists of f-orbitals, a d-sublevel consists of d -orbitals and so on…

Section 2: Orbitals Page 100 Ø Something to help clarify this is to think of an atom as a hotel Ø If the atom is a hotel, each energy level is a floor of the hotel Ø The sublevels are different types of rooms on that floor Ø The orbitals are the individual rooms on each floor Ø Each room (orbital) has a maximum occupancy of 2 people (electrons)

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Electron configurations and orbital diagrams are used to show the location (probable) of electrons in an atom Electron configurations use the notation seen below: Ø Orbital notation looks like this fun stuff down here: Ø

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Electron configurations and orbital notations are determined by applying the relationship between the periodic table and the structure of atoms (which you figured out in your lab) Ø Let’s find the e- configuration and orbital notation for oxygen. . . and while we’re at it, say hello to Pauli and Hund!

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Now how about iron? Ø And what about bromine?
Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø And lets try neodymium (atomic #60) ? Ø And for fun, lead!

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø What is incorrect about orbital notation below?
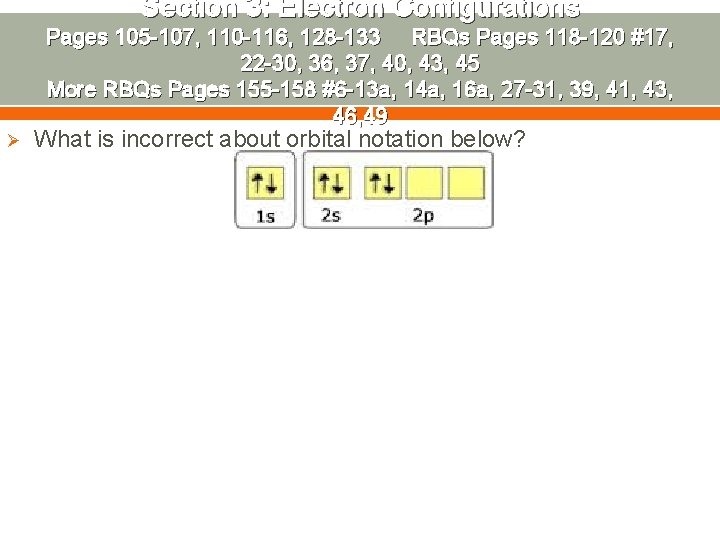
Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø What is incorrect about orbital notation below?

Which element is represented by the electron configuration seen below? A. B. C. D. Vanadium Argon Iron Cobalt
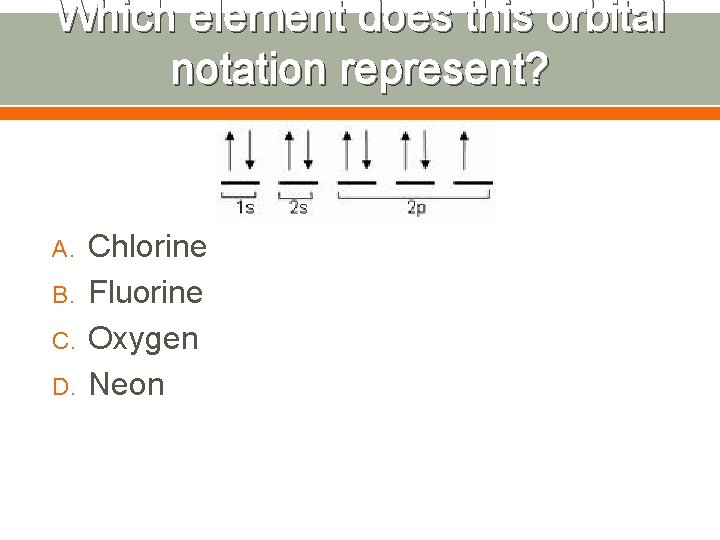
Which element does this orbital notation represent? A. B. C. D. Chlorine Fluorine Oxygen Neon

What element does the orbital notation represent? A. B. C. D. Vanadium Phosphorous Oxygen Sulfur
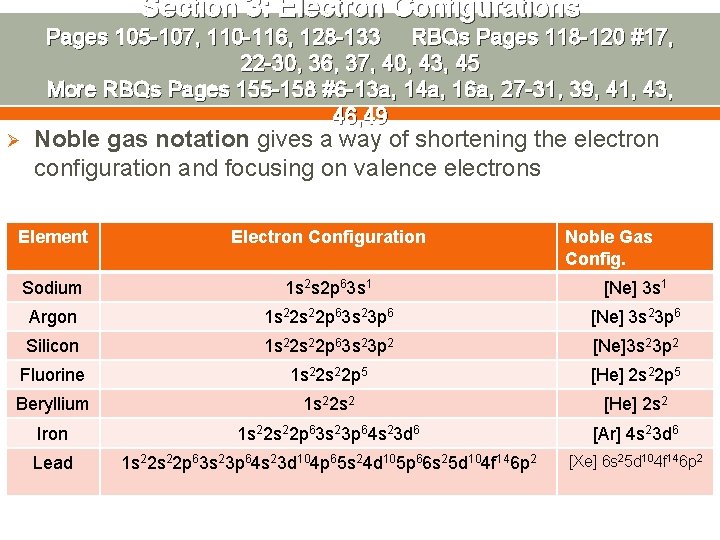
Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Noble gas notation gives a way of shortening the electron configuration and focusing on valence electrons Element Electron Configuration Noble Gas Config. Sodium 1 s 2 s 2 p 63 s 1 [Ne] 3 s 1 Argon 1 s 22 p 63 s 23 p 6 [Ne] 3 s 23 p 6 Silicon 1 s 22 p 63 s 23 p 2 [Ne]3 s 23 p 2 Fluorine 1 s 22 p 5 [He] 2 s 22 p 5 Beryllium 1 s 22 s 2 [He] 2 s 2 Iron 1 s 22 p 63 s 23 p 64 s 23 d 6 [Ar] 4 s 23 d 6 Lead 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 66 s 25 d 104 f 146 p 2 [Xe] 6 s 25 d 104 f 146 p 2

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Based on the noble gas configurations on the previous slide, write steps to use in determining the noble gas configuration of an element.
![The noble gas configuration below is that of which element? [Kr] 5 s 24 The noble gas configuration below is that of which element? [Kr] 5 s 24](http://slidetodoc.com/presentation_image_h/904a4019bc660119b320f0235402ca59/image-36.jpg)
The noble gas configuration below is that of which element? [Kr] 5 s 24 d 8 A. B. C. D. Ni Pd Pt Ag
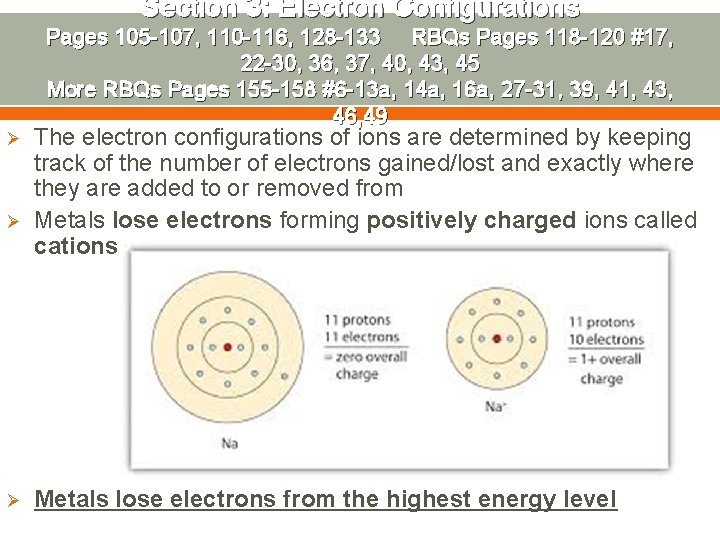
Section 3: Electron Configurations Ø Ø Ø Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 The electron configurations of ions are determined by keeping track of the number of electrons gained/lost and exactly where they are added to or removed from Metals lose electrons forming positively charged ions called cations Metals lose electrons from the highest energy level
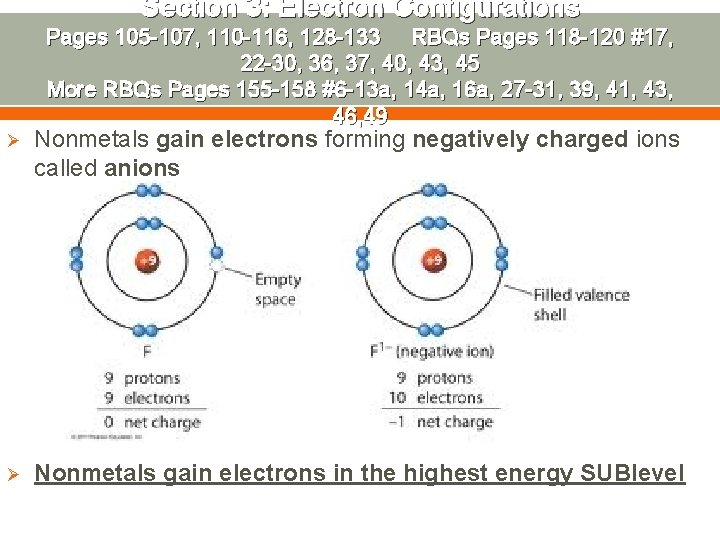
Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Nonmetals gain electrons forming negatively charged ions called anions Ø Nonmetals gain electrons in the highest energy SUBlevel

Section 3: Electron Configurations Pages 105 -107, 110 -116, 128 -133 RBQs Pages 118 -120 #17, 22 -30, 36, 37, 40, 43, 45 More RBQs Pages 155 -158 #6 -13 a, 14 a, 16 a, 27 -31, 39, 41, 43, 46, 49 Ø Write the electron configuration and noble gas configuration for the nitride ion, N 3 -. Ø Write the electron configuration and noble gas configuration for the Cadmium ion, Cd+2.

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, Ø The structure of the atom, 32, as 33, reflected in the 22 -26, 35 -38, 45 periodic table, can be used to determine and predict various properties of atoms Ionization energy: the energy required to remove an electron from a gaseous atom or ion Atomic Radius: Half the distance between the nuclei of two atoms Electronegativity: the ability of an atom in a molecule to attract shared electrons Ø With each property, it is all about how strongly the nucleus of the atom attracts valence electrons; two factors impact this
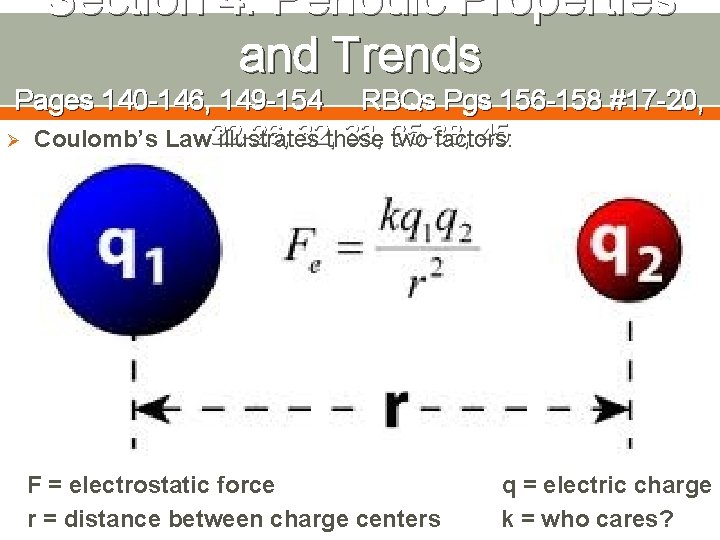
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 32, these 33, 35 -38, 45 Ø Coulomb’s Law 22 -26, illustrates two factors: F = electrostatic force r = distance between charge centers q = electric charge k = who cares?
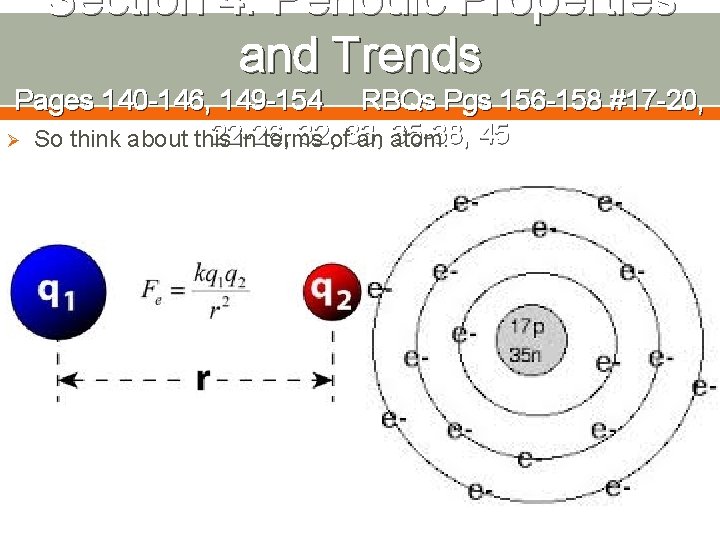
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, of 33, 35 -38, 45 Ø So think about this in terms an atom:

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, 33, than 35 -38, Ø The charge factor is about more just 45 the number of protons Ø in the nucleus The focus is on the nucleus attracting the valence electrons, so the core/inner shell/shielding electrons must be considered
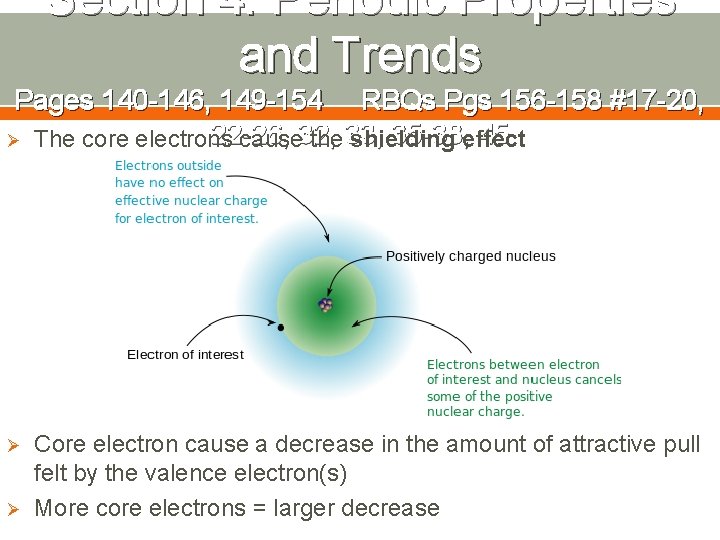
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 35 -38, effect 45 Ø The core electrons cause 32, the 33, shielding Ø Ø Core electron cause a decrease in the amount of attractive pull felt by the valence electron(s) More core electrons = larger decrease

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 33, 35 -38, 45 charge(Zeff) Ø This is summed up by the 32, effective nuclear

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, elements; 33, 35 -38, 45 describe how the Ø Find the Zeff for the following then Zeff changes moving from top to bottom within a group

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, elements 33, 35 -38, 45 then describe how Ø Find the Zeff for the first four below; the Zeff changes moving from left to right within a period
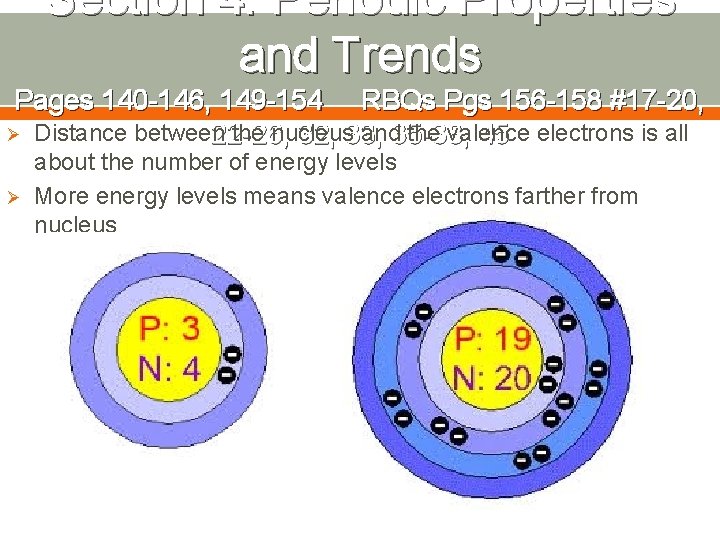
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, Ø Distance between the nucleus and 35 -38, the valence 22 -26, 32, 33, 45 electrons is all Ø about the number of energy levels More energy levels means valence electrons farther from nucleus
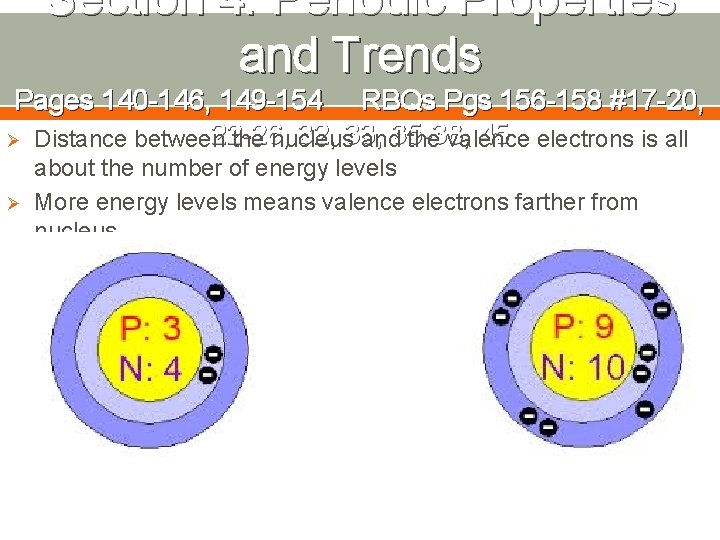
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, 33, 45 electrons is all Ø Distance between the nucleus and 35 -38, the valence Ø about the number of energy levels More energy levels means valence electrons farther from nucleus

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, Ø So when comparing two 32, elements in a period, 22 -26, 33, 35 -38, 45 which factor causes them to have different properties? Which is ignored? Ø And when comparing two elements in the same group, which factor causes them to have different properties? Which is ignored?
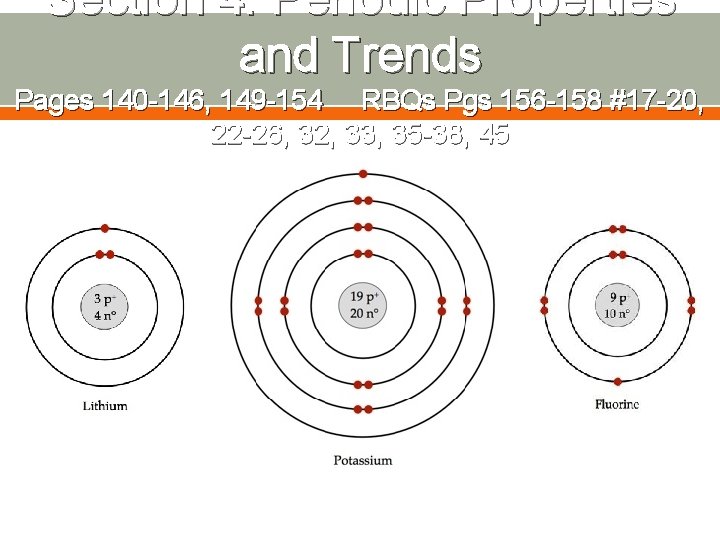
Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, 33, 35 -38, 45
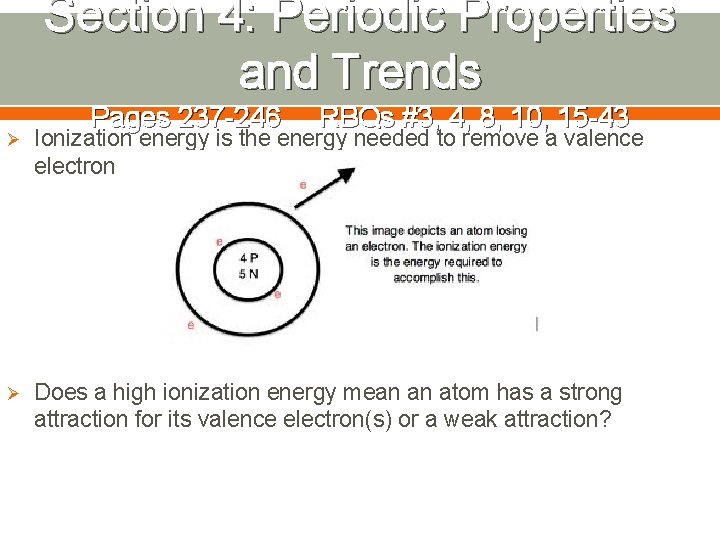
Section 4: Periodic Properties and Trends Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Ø Ionization energy is the energy needed to remove a valence electron Ø Does a high ionization energy mean an atom has a strong attraction for its valence electron(s) or a weak attraction?
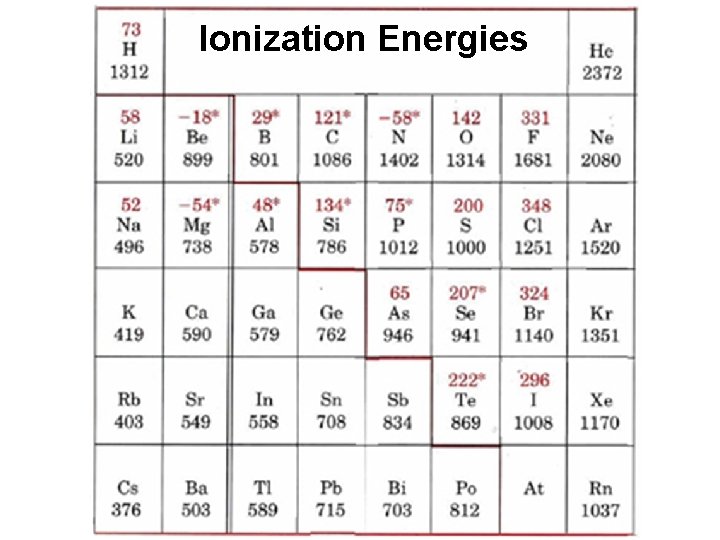
Ionization Energies
Section 4: Periodic Properties and Trends Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Ø Which has a higher ionization energy: lithium or fluorine? Explain. Ø Which has higher ionization energy: lithium or potassium? Explain.
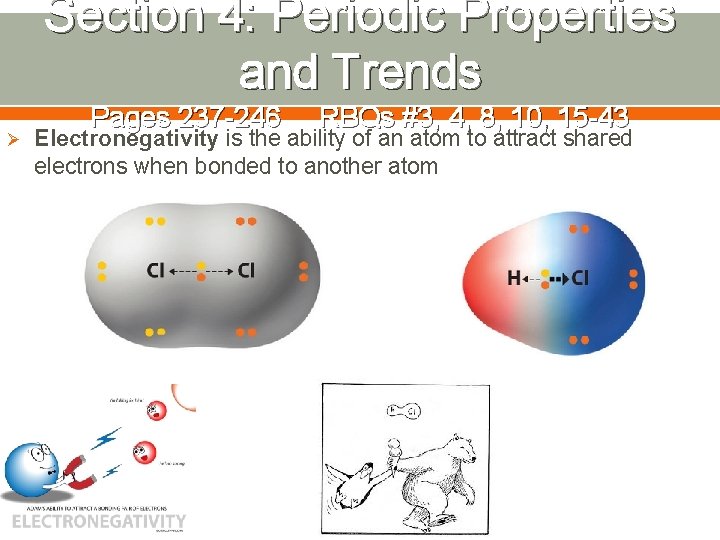
Section 4: Periodic Properties and Trends Ø Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Electronegativity is the ability of an atom to attract shared electrons when bonded to another atom
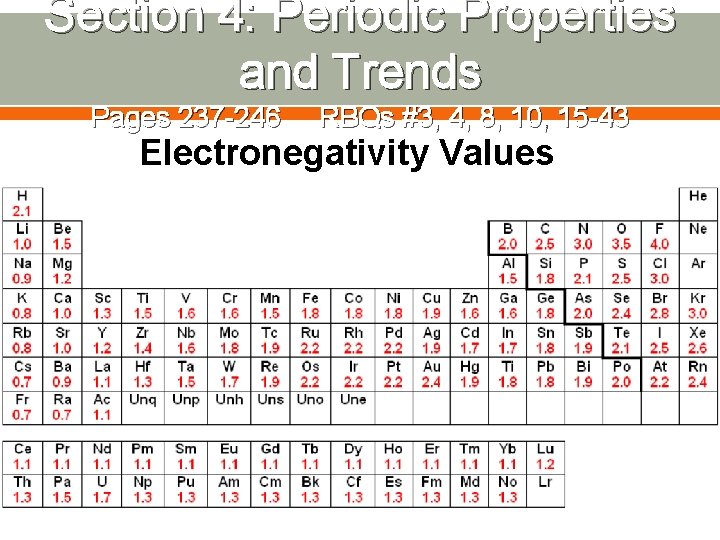
Section 4: Periodic Properties and Trends Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Electronegativity Values
Section 4: Periodic Properties and Trends Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Ø Which has a greater electronegativity: lithium or fluorine? Explain. Ø Which has a greater electronegativity: lithium or potassium? Explain.

Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, 33, 35 -38, 45 Ø Atomic radius is defined as half the distance between the nuclei in a molecule consisting of identical atoms Ø More simply, the atomic radius can be thought of as the size of an atom Ø Since the boundary of an atom can’t be defined, we can’t just measure from one side of an atom to another


Section 4: Periodic Properties and Trends Pages 140 -146, 149 -154 RBQs Pgs 156 -158 #17 -20, 22 -26, 32, 33, 35 -38, 45 Ø Which atom is larger: lithium or fluorine? Explain. Ø Which atom is larger: lithium or potassium? Explain.

Section 4: Periodic Properties and Trends Ø Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Changes that occur with ions can be determined similarly; draw a Bohr model for a neutral lithium atom and an lithium ion below. Describe how their radii differ and why this difference exists.
Section 4: Periodic Properties and Trends Ø Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 Now do the same for a Bohr model for a neutral fluorine atom and a fluoride ion below. Describe how their radii differ and why this difference exists. They are not the same size!!!
Section 4: Periodic Properties and Trends Ø Pages 237 -246 RBQs #3, 4, 8, 10, 15 -43 And now draw a fluoride ion, and oxide ion, and a sodium ion. Arrange them from smallest to largest, and explain the order selected.


Arrange the following by increasing atomic radius: Ca, Ni, Br? A. B. C. D. E. Ni, Br, Ca, Ni Ca, Br, Ni Ca, Ni, Br Br, Ni, Ca

Arrange the following in order of increasing ionization energy: F, Br, I A. B. C. D. E. I, Br, F Br, I, F F, Br, I I, F, Br Br, F, I

And now increasing electronegativity: N, Rb, Ca A. B. C. D. E. N, Rb, Ca Ca, Rb, N, Ca, Rb Rb, Ca, N
- Slides: 67