Chapter V Black box growth 1 11242020 Growth

Chapter V Black box growth 1 11/24/2020

Growth without non-catabolic product Single substrate limited growth 2 11/24/2020
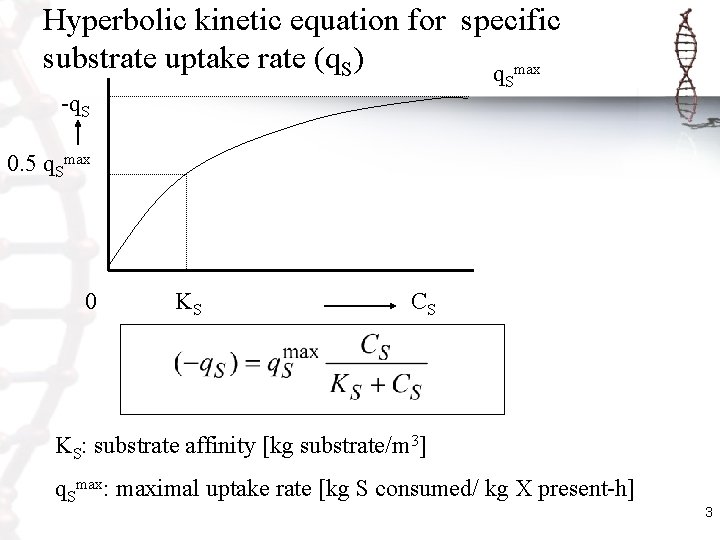
Hyperbolic kinetic equation for specific substrate uptake rate (q. S) q max -q. S S 0. 5 q. Smax 0 KS CS KS: substrate affinity [kg substrate/m 3] q. Smax: maximal uptake rate [kg S consumed/ kg X present-h] 3

Substrate uptake for maintenance Some part of substrate is used for producing energy needed for cells maintenance. This part is catabolized with a rate m. S. Hence, m. S = kg substrate catabolized for maintenance / kg biomass present–hour. 4

Herbert-Pirt relation for substrate It means the substrate taken into cells are used for growth and maintenance only. Both m. S and YSXmax are model parameters. -q. S (-m. S) Herbert-Pirt linear plot for substrate, non-catabolic product is absent 0 5

Combination of the q. S equation with Herbert. Pirt relation • CS = 0 , = m. SYSXmax = -kd • CS >>>, = [-q. Smax-(m. S)]YSXmax = max • = 0, (-q. S) = (-m. S), which occurs at CS = CSmin. The specific growth rate equation based on single substrate is then: 6
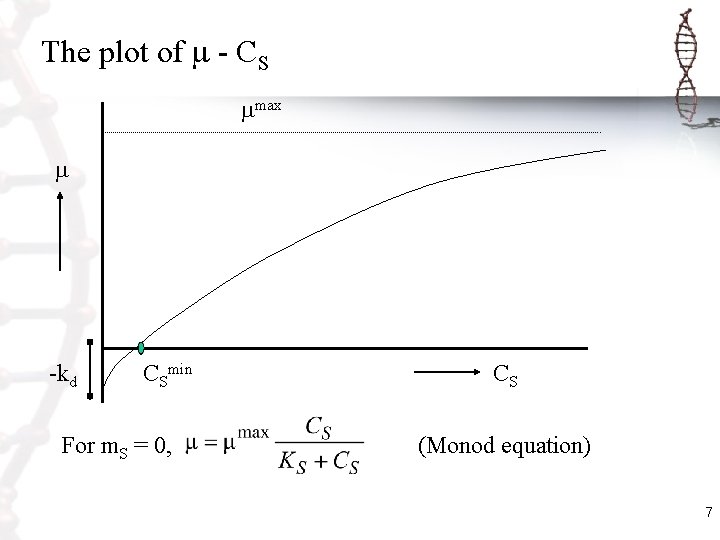
The plot of - CS max -kd CSmin CS For m. S = 0, (Monod equation) 7

Using Herbert-Pirt relation to calculate other rates Example: The case of aerobic growth on glucose, N-source is ammonia Suppose –q. S = 0. 3125 + 0. 0015 Growth reaction: -0. 3125 C 6 H 12 O 6 + a. NH 4+ + b. O 2 + c. H+ + d. H 2 O + C 1 H 1. 8 O 0. 5 N 0. 2 + e. CO 2 Use the yield and elemental balance to obtain a, b, c, d, e The catabolic reaction is: -C 6 H 12 O 6 - 6 O 2 + 6 CO 2 + 6 H 2 O What are (-q. NH 4+), (-q. O 2), q. CO 2, q. H+, q. H 2 O? 8

How to measure the kinetic parameters? Obtaining max and YSXmax. Using batch fermentation at constant volume where CS >> Hence = max (constant, during the exponential phase) q. S = q. Smax (constant), YSX = YSX max = (CX-CXo)/(CSo-CS) Obtaining KS and m. S. Using chemostats, is the variable to get -q. S. Apply Herbert. Pirt relation and plot –q. S vs. to get YSXmax and m. S. Plot –q. S vs. CS to obtain KS and q. Smax. 9

Further reading • The Herbert-Pirt relation including non catabolic product formation • Extended Herbert-Pirt relation • Different q. P - function • Effect of temperature and p. H. 10

Chapter VI Growth and product formation in bioreactors 11 11/24/2020
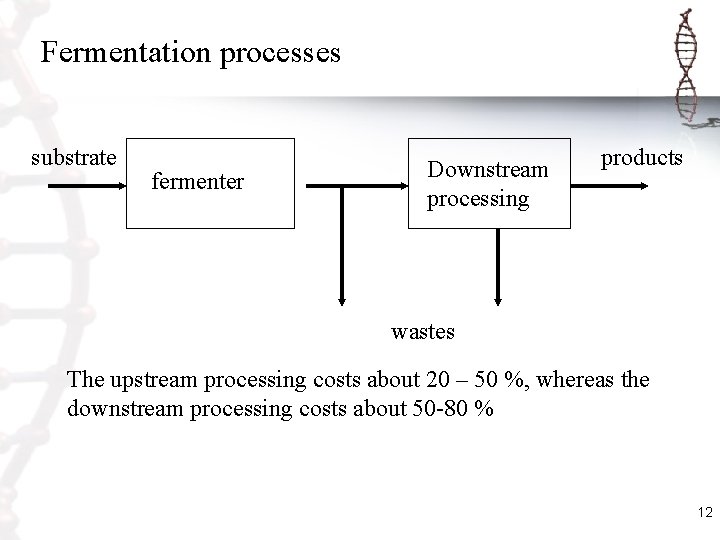
Fermentation processes substrate fermenter Downstream processing products wastes The upstream processing costs about 20 – 50 %, whereas the downstream processing costs about 50 -80 % 12

The type of reactors and processes The reactors systems: • Aerated stirred tank reactor 0. 1 – 1000 m 3 with cooling equipment • Bubble column 0. 1 – 10, 000 m 3 with airlift and cooling equipment • Bioreactors for immobilized cells/biocatalysts (packed bed, fluidized bed, trickle bed) The types of processes used are batch, continuous, and fed batch systems. Mostly, batch and fed batch reactors are used in industry whereas continuous reactors (chemostats) are used in laboratories. 13

Fermenter utilities Sterilization: 1. Autoclaving 2. Live steam batch 3. Membrane 4. Heat shock for continuous system (less odor, energy, degradation, dilution, time) 5. Mixing by stirrer 6. mix 20 – 200 seconds, power 1 -3 k. W/m 3. 7. Oxygen input 8. 50 – 200 mol/m 3 -h. 14

Fermenter utilities (cont’d) Air filtration, using membrane / bed filter with dry air about 60 m 3 air/m 3 broth-h. Compressor, 2 bar, 150 o. C. Heat removal • Cooling jacket • Coil With cool water temperature at 10 o. C and capacity is about 50, 000 k. J/m 3 reactor-h. 15

Laboratory chemostat and industrial fermenter Applikon laboratory chemostat at Kluyverlab, Delft University of Technology Labatt Breweries - London, Ontario New Vertical Fermenter 16

The world largest fermenter There may be biological waste treatment in larger vessels, but the world's largest fermenter is shown in these photos taken from Chemical and Engineering News. The fermenter is 200' high and 25 ft diam. The first photo (Chem. Eng. News, 10 -Apr-78) shows the fermenter being transported on vehicles with tank treads. 17
Packed bed fermenter 18

What do we want? Grow on low cost substrate Thus, it is important to analyze the kinetics of growth and product formation, designing the optimal feed profile, role of transport processes, maybe use bigger reactors. High q. P at low Remember! Extra cellular products q. S or completely determines the microbial behavior. It must be controlled at an optimal value High yield of biomass High max High temperature tolerant Low viscous biomass Generally regarded as safe (GRAS) Mineral medium 19
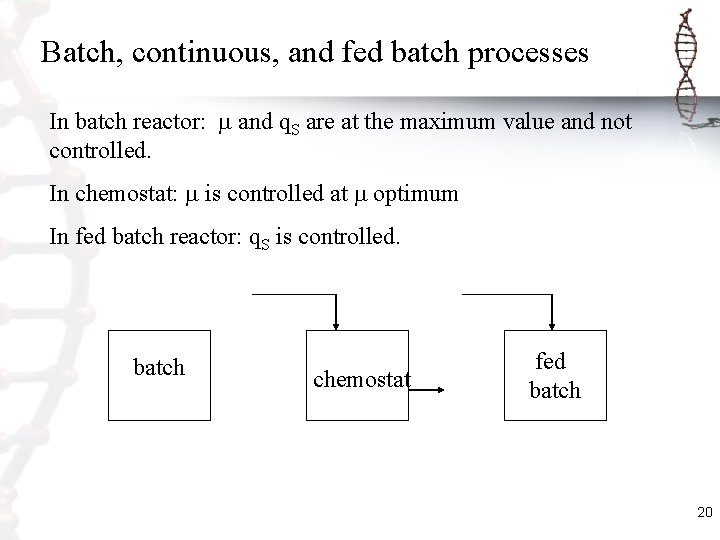
Batch, continuous, and fed batch processes In batch reactor: and q. S are at the maximum value and not controlled. In chemostat: is controlled at optimum In fed batch reactor: q. S is controlled. batch chemostat fed batch 20

Constant volume batch reactor The steps: 1. Add sterile growth medium solution (substrate consist of C, N, P source, K+, Mg+, salts, vitamin, trace elements) and choose the right T and p. H. 2. Add electron acceptors (O 2, NO 3 -, etc) 3. Inoculate microorganism at initial time with concentration CXo. 4. Run the fermentation and harvest the broth, proceed to the downstream processing. 21

Constant volume batch reactor What is the plot CS and CX with time? Derive from the mass balance, one can obtain that the growth is exponential, the slope of the curve CX vs. time is r. X. Parameters State variables reactor operator micro organism V CS 0 CX 0 q. Smax YSXmax m. S KS CS CX 22

Substrate concentration profile At constant volume: For batch reactor: t = 0, CS = Cso ; Hence, by putting CS = 0, t end of batch is obtained The slope in CS vs. t plot is r. S. 23

Try to sketch the curve CS, CX, , -q. S, YSX, r. X, -r. S versus time for batch reactor! Conclusion for the batch reactor • , YSX, and q. S are constant at the maximum values • Microbial model parameters are determined by combining the exponential equation with CS and CX data versus time. • , the most important rate, can not be controlled 24

Chemostat Steady state: constant volume, constant in and out flow rates. Constant CS and CX. Parameters State variables reactor operator micro organism V CS, in L, in q. Smax YSXmax m. S KS L, out CS CX 25

Steps in operating continuous culture • Sterilized reactor is filled with sterile growth medium solution containing substrate at CS, in • Air sparging starts to provide electron acceptor • Add a small amount of microorganism (inoculum) • Start the medium in flow (often L, in and L, out are nearly equal, not always) • Wait until steady state is achieved The main property of chemostat: 1. CS and CX are independently manipulated by the user by manipulating transport (to and from the reactor) of substrate and biomass. 2. Excellent experimental tool to study microbial kinetics, stoichiometry, under controlled conditions. 26

Biomass balance in chemostat In – out + gen = acc The µ is chosen by manipulating exit flow rate Hence, all other qi-values are set (stoichiometric coupling) Does CS depend on CS, in? Use µ(CS) to prove it. 27

Critical dilution rate The maximal value for D (=µ) attainable in the chemostat, is achieved when the maximal CS in achieved. At critical dilution rate, CS CS, in. Note: CS, in normally in order 10000 mg/l, CS, min order 1 mg/l, KS order 10 mg/l. Wash out If D > Dcrit, the cell will be washed out Then, sketch the graph CS vs. D. 28

Substrate mass balance This equation together with Herbert-Pirt relation provide r. S and ( -q. S) from measured concentration, flows, and volume. What about CX? 29

The application of chemostat 1. Kinetic studies, at different µ = D one can varies q. S, CS to obtain YSXmax, m. S, q. Smax, KS. 2. Physiological and genomic micro array studies of microorganisms under defined steady state, I. e. substrate, electron acceptor, N-source, type of limitation for growth. 3. Waste water treatment 4. Industrial fermentation, which is not widely applied 30

Chemostat optimization Normally, the maximal r. X and –r. S are needed as the economic parameter Try to find the optimum D where dr. S/d. D and dr. X/d. D = 0. Note that r. S has a maximum at higher D. Chemostat wash out dynamic What happen when D is close to Dcrit? What happen to CX when D = D’>Dcrit? 31

Pro and cons of chemostat (+) Excellent experimental tool because µ is defined (-) Low biomass and product concentration Loss of biomass in outflow Relatively prone to be contaminated compare to batch or fed batch reactors Microbial selection for non-producing mutants 32

Fed batch fermentation In batch reactor, CS and CX are high. No transport of S or X and no control on µ. In chemostat, CS and CX are low. Transport of S or X and control on µ. In fed batch reactor. Substrate transport in, not out. No biomass transport. Why fed batch? 1. Low CS no toxicity / osmotic problem 2. High CX high CP easier downstream processing 3. Control of µ? 33
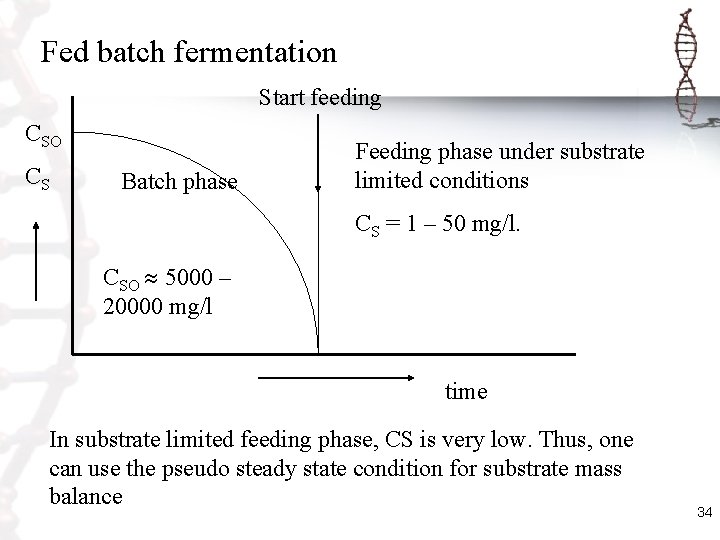
Fed batch fermentation Start feeding CSO CS Batch phase Feeding phase under substrate limited conditions CS = 1 – 50 mg/l. CSO 5000 – 20000 mg/l time In substrate limited feeding phase, CS is very low. Thus, one can use the pseudo steady state condition for substrate mass balance 34

Substrate mass balance in fed batch reactor Hence, in a fed batch reactor the substrate conversion (-r. S) is controlled by the operator. Through controlled r. S, µ is controlled. It is obvious that the reactor volume changes with time. However, since the change is very small, for simplicity we can assume constant volume, constant density. 35

Rates in fed batch reactor We start with an assumption that volume is a variable Biomass balance Pseudo steady state substrate mass balance Product mass balance 36

Possible substrate feeding strategies in a fed batch fermentation 1. Substrate input ( S, in. CS, in) is constant 2. µ is maintained at µ optimum that gives maximal q. P or maximal YSP. 3. Substrate input is determined by other known reactor limitations (oxygen, heat, etc) 4. Apply Herbert-Pirt equation and biomass balance to calculate r. X and CX. 37

Biomass balance in fed batch reactor Assume constant volume (DV <<) Combine with Herbert-Pirt relation and eliminate –r. S using its fed batch relation: This is homogeneous differential equation in CX. 38

Biomass balance in fed batch reactor The first explains growth, the later is about maintenance. Initially CX is low in a fed batch reactor. By neglecting maintenance: CX increases linearly with time Later, CX increases, more and more substrate needed for maintenance d. CX/dt decreases. End, all substrate needed for maintenance, d. CX/dt = 0 39

Fed batch reactor with constant feeding rate The analytical solution for CX(t) in feeding phase. t = time after start feeding CXf = biomass concentration at start feeding Note: For t >>> 0, For t <<< (YSXmax(-m. S))-1, linear growth 40

Fed batch reactor with constant feeding rate r. X decreases µ sharply decreases q. S decreases CS decreases slowly 41

Fed batch reactor with constant optimum µ • µ = µopt = constant qi, Yij are all constant. • q. S constant at q. Sopt CS constant. • Biomass concentration increases exponentially • All rates increases exponentially • Substrate feeding rate increases exponentially tf = time when feeding starts 42
- Slides: 42