Chapter Two Polar Reaction Under Basic Conditions a

Chapter Two Polar Reaction Under Basic Conditions a. Substitution and Elimination at C(sp 3)-X σ bonds b. Addition of Nuclephiles to Electrophilic π bonds c. Substitution at C(sp 2)-X σ bonds d. Base-promoted Rearrangements
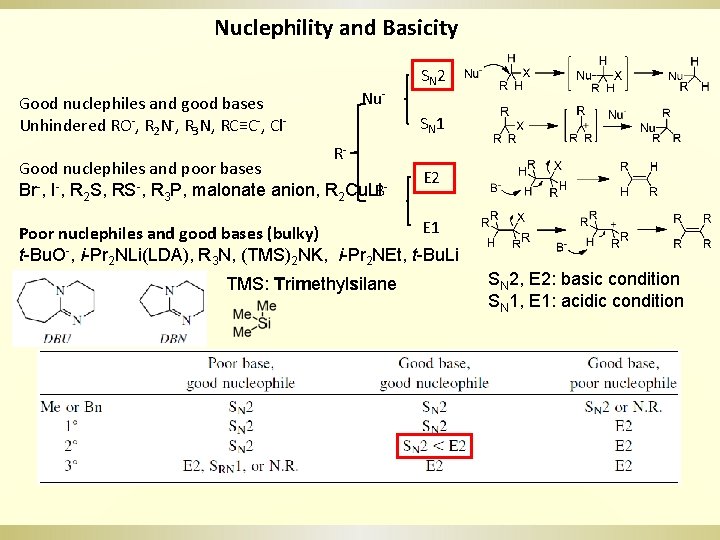
Nuclephility and Basicity Nu- Good nuclephiles and good bases Unhindered RO-, R 2 N-, R 3 N, RC≡C-, Cl- SN 2 SN 1 R- Good nuclephiles and poor bases Br-, I-, R 2 S, RS-, R 3 P, malonate anion, R 2 Cu. Li. B- E 2 E 1 Poor nuclephiles and good bases (bulky) t-Bu. O-, i-Pr 2 NLi(LDA), R 3 N, (TMS)2 NK, i-Pr 2 NEt, t-Bu. Li TMS: Trimethylsilane SN 2, E 2: basic condition SN 1, E 1: acidic condition
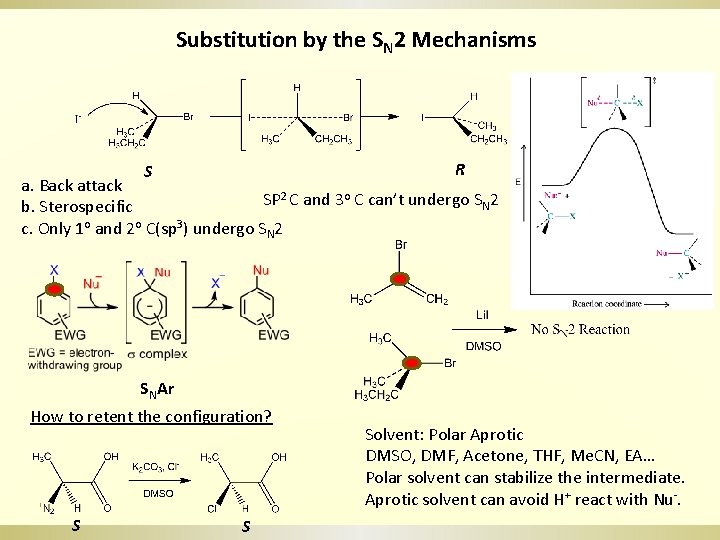
Substitution by the SN 2 Mechanisms R S a. Back attack 2 C and 3 o C can’t undergo S 2 SP N b. Sterospecific c. Only 1 o and 2 o C(sp 3) undergo SN 2 SNAr How to retent the configuration? S S Solvent: Polar Aprotic DMSO, DMF, Acetone, THF, Me. CN, EA… Polar solvent can stabilize the intermediate. Aprotic solvent can avoid H+ react with Nu-.
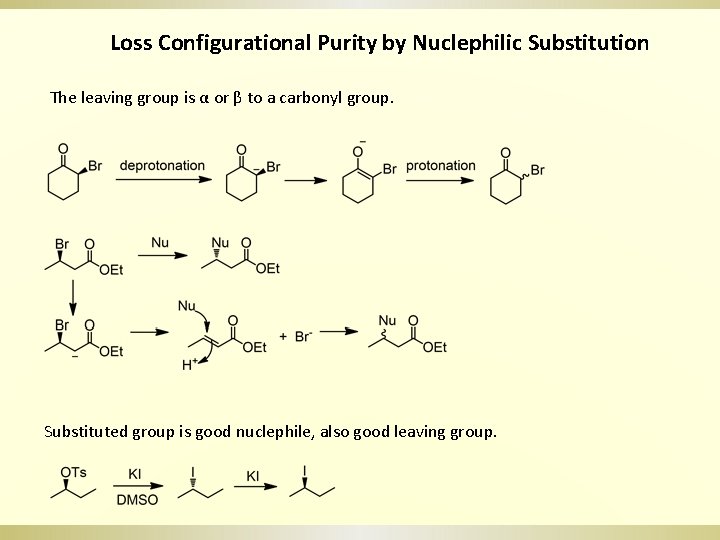
Loss Configurational Purity by Nuclephilic Substitution The leaving group is α or β to a carbonyl group. Substituted group is good nuclephile, also good leaving group.
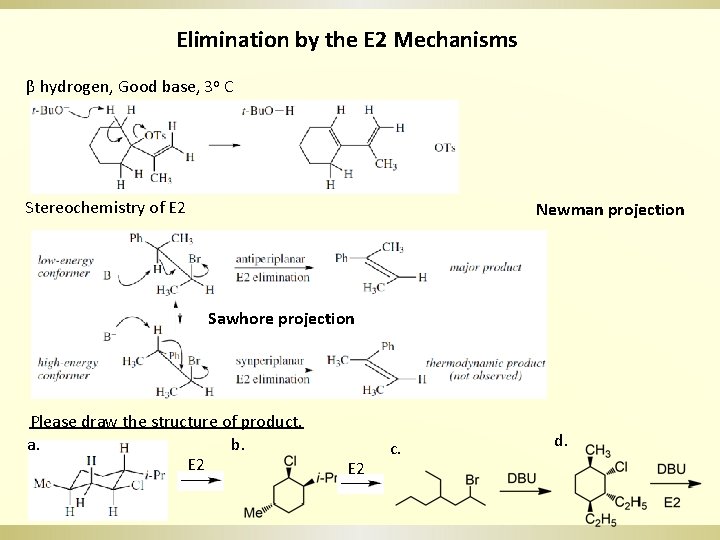
Elimination by the E 2 Mechanisms β hydrogen, Good base, 3 o C Stereochemistry of E 2 Newman projection Sawhore projection Please draw the structure of product. b. a. E 2 c. d.
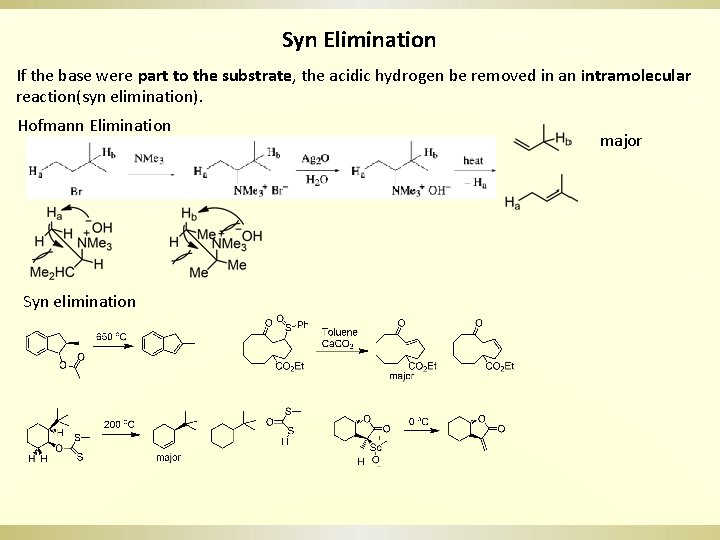
Syn Elimination If the base were part to the substrate, the acidic hydrogen be removed in an intramolecular reaction(syn elimination). Hofmann Elimination Syn elimination major
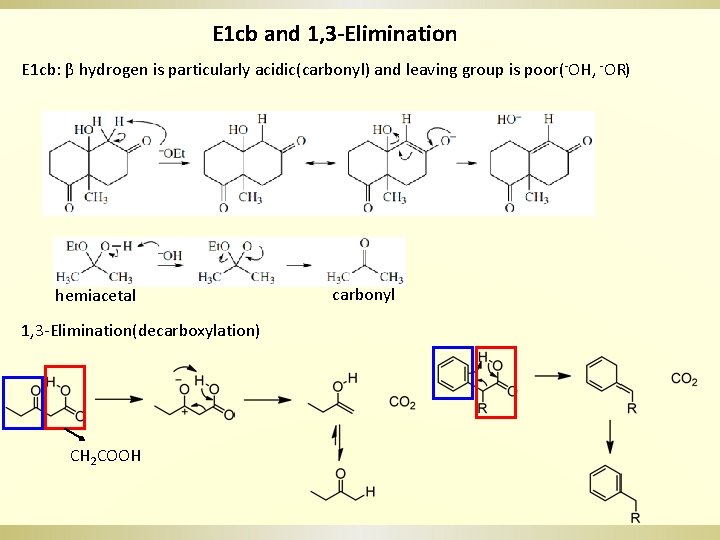
E 1 cb and 1, 3 -Elimination E 1 cb: β hydrogen is particularly acidic(carbonyl) and leaving group is poor(-OH, -OR) hemiacetal 1, 3 -Elimination(decarboxylation) CH 2 COOH carbonyl
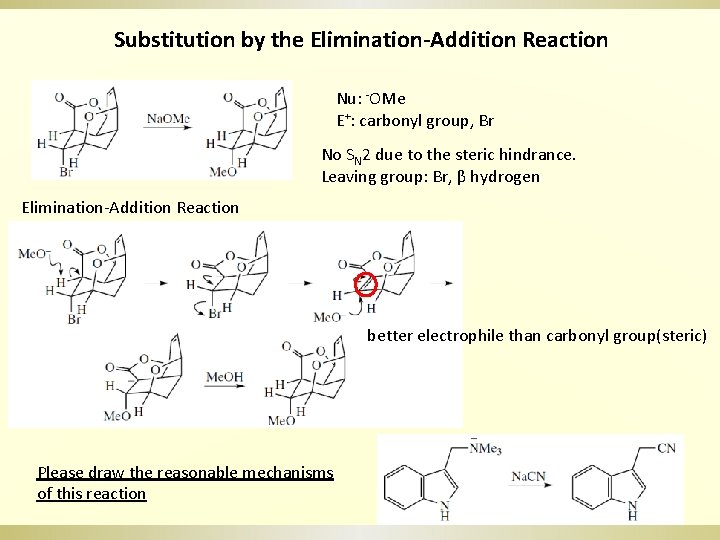
Substitution by the Elimination-Addition Reaction Nu: -OMe E+: carbonyl group, Br No SN 2 due to the steric hindrance. Leaving group: Br, β hydrogen Elimination-Addition Reaction better electrophile than carbonyl group(steric) Please draw the reasonable mechanisms of this reaction
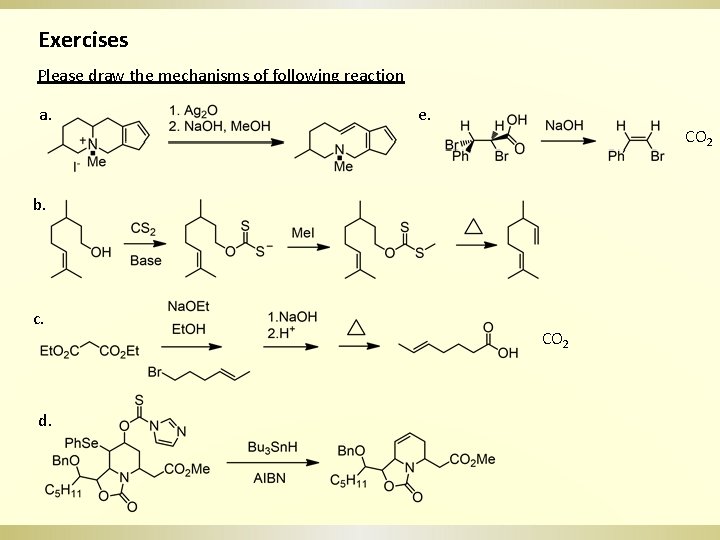
Exercises Please draw the mechanisms of following reaction a. e. CO 2 b. c. d. CO 2
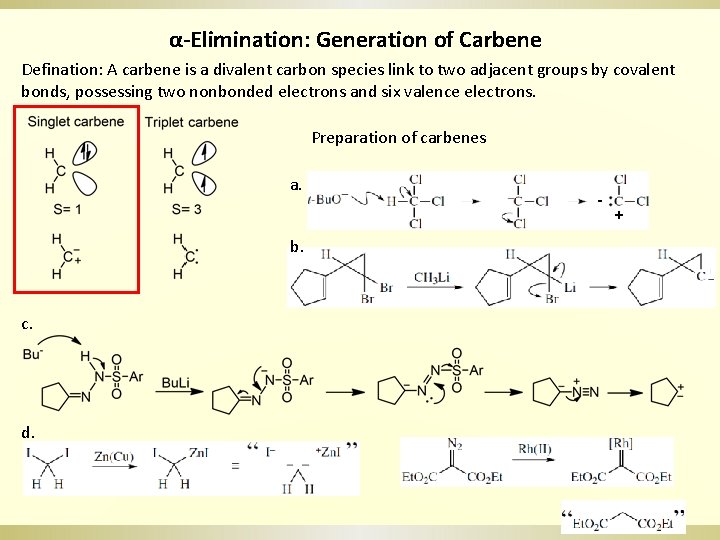
α-Elimination: Generation of Carbene Defination: A carbene is a divalent carbon species link to two adjacent groups by covalent bonds, possessing two nonbonded electrons and six valence electrons. Preparation of carbenes a. b. c. d. - +
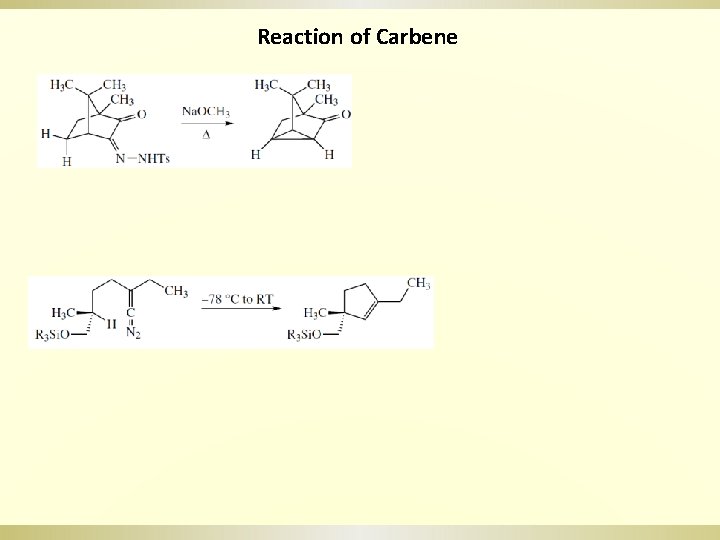
Reaction of Carbene
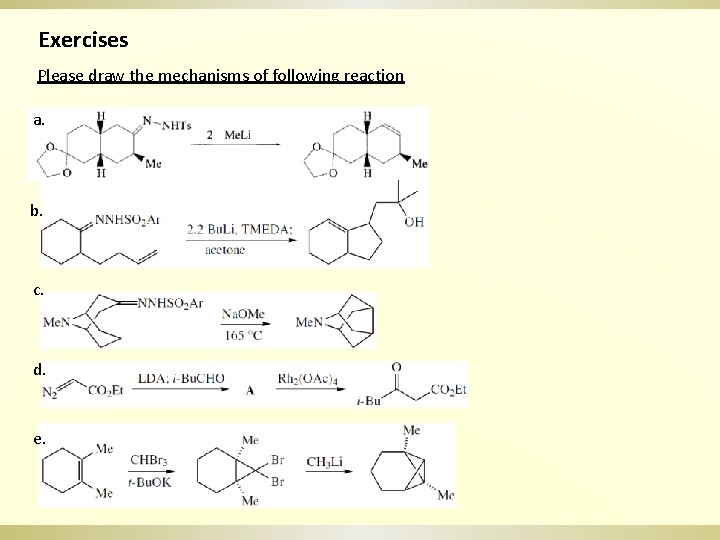
Exercises Please draw the mechanisms of following reaction a. b. c. d. e.

Polar Reaction Under Basic Conditions a. Substitution and Elimination at C(sp 3)-X σ bonds b. Addition of Nuclepphiles to Electrophilic π bonds c. Substitution at C(sp 2)-X σ bonds d. Base-promoted Rearrangements
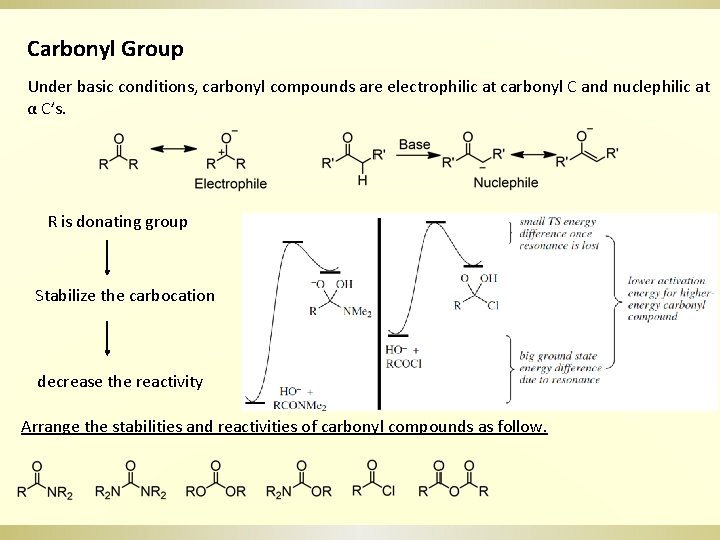
Carbonyl Group Under basic conditions, carbonyl compounds are electrophilic at carbonyl C and nuclephilic at α C’s. R is donating group Stabilize the carbocation decrease the reactivity Arrange the stabilities and reactivities of carbonyl compounds as follow.
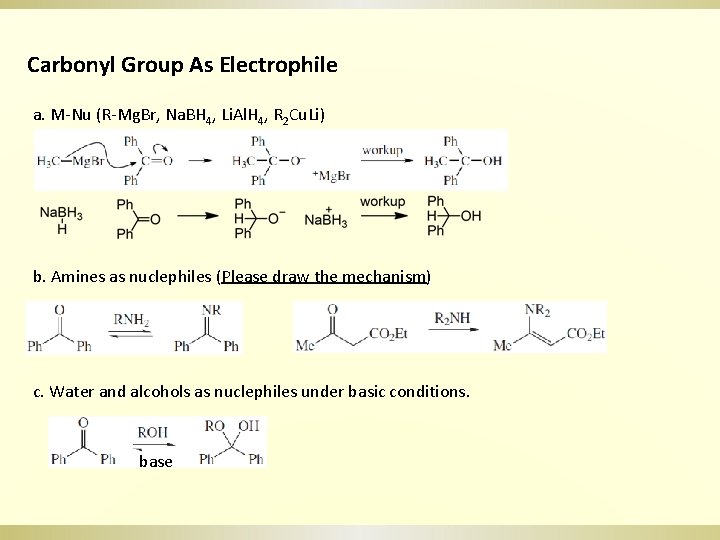
Carbonyl Group As Electrophile a. M-Nu (R-Mg. Br, Na. BH 4, Li. Al. H 4, R 2 Cu. Li) b. Amines as nuclephiles (Please draw the mechanism) c. Water and alcohols as nuclephiles under basic conditions. base
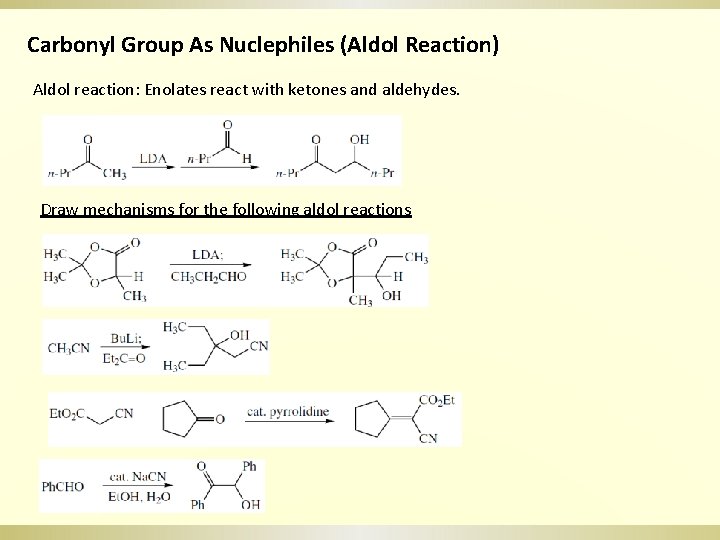
Carbonyl Group As Nuclephiles (Aldol Reaction) Aldol reaction: Enolates react with ketones and aldehydes. Draw mechanisms for the following aldol reactions
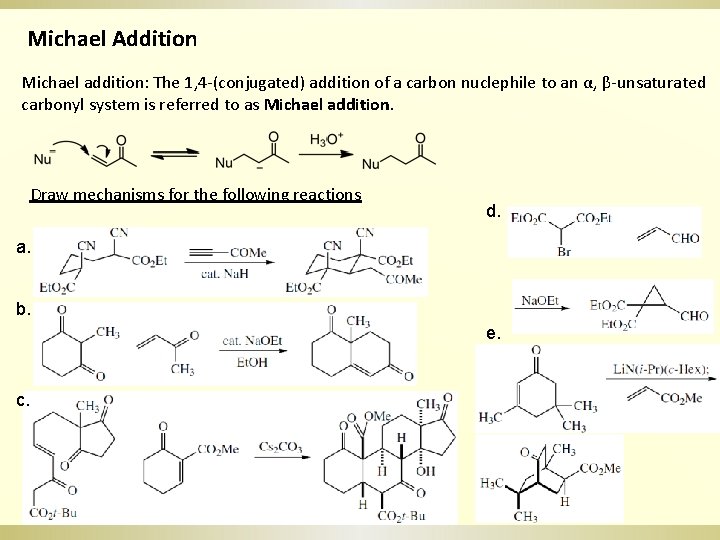
Michael Addition Michael addition: The 1, 4 -(conjugated) addition of a carbon nuclephile to an α, β-unsaturated carbonyl system is referred to as Michael addition. Draw mechanisms for the following reactions d. a. b. e. c.
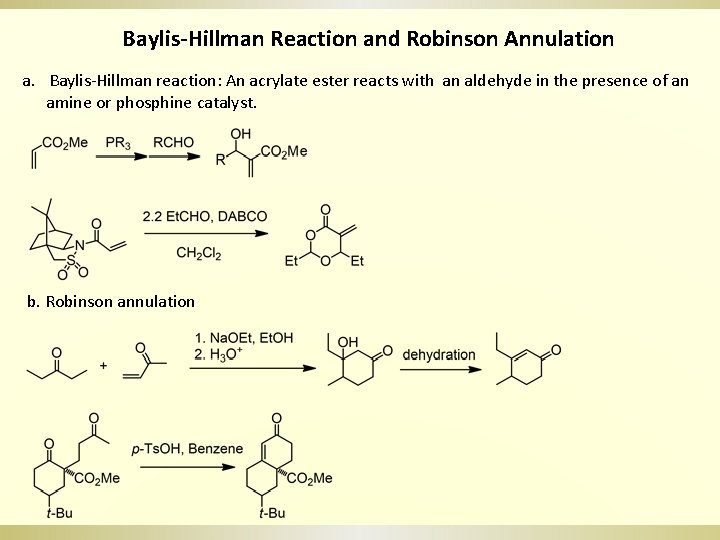
Baylis-Hillman Reaction and Robinson Annulation a. Baylis-Hillman reaction: An acrylate ester reacts with an aldehyde in the presence of an amine or phosphine catalyst. b. Robinson annulation

Polar Reaction Under Basic Conditions a. Substitution and Elimination at C(sp 3)-X σ bonds b. Addition of Nuclephiles to Electrophilic π bonds c. Substitution at C(sp 2)-X σ bonds d. Base-promoted Rearrangements
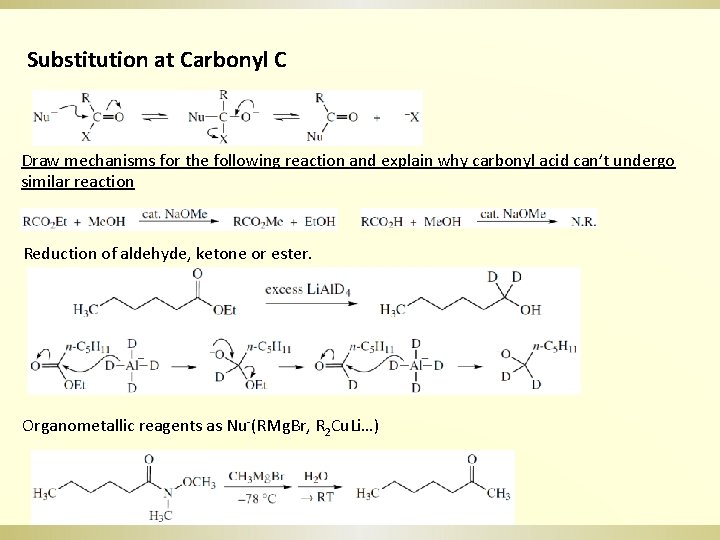
Substitution at Carbonyl C Draw mechanisms for the following reaction and explain why carbonyl acid can’t undergo similar reaction Reduction of aldehyde, ketone or ester. Organometallic reagents as Nu-(RMg. Br, R 2 Cu. Li…)
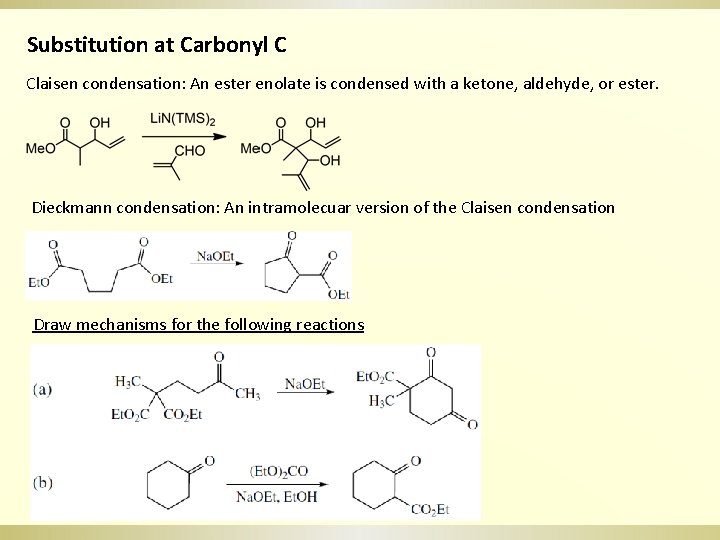
Substitution at Carbonyl C Claisen condensation: An ester enolate is condensed with a ketone, aldehyde, or ester. Dieckmann condensation: An intramolecuar version of the Claisen condensation Draw mechanisms for the following reactions
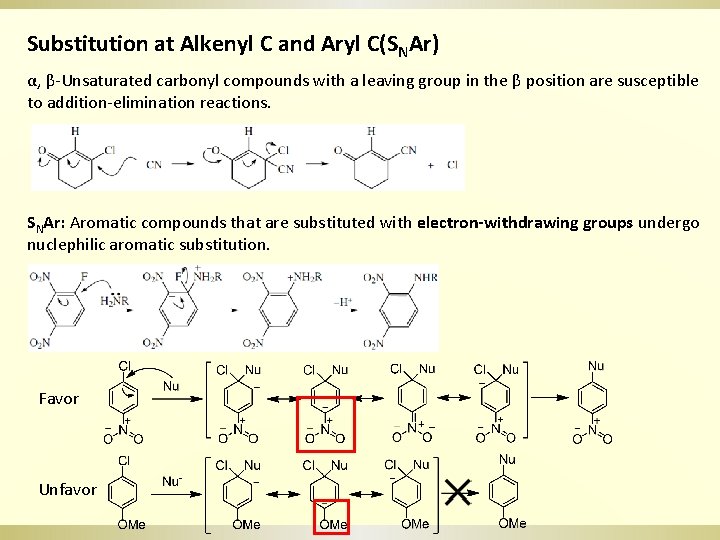
Substitution at Alkenyl C and Aryl C(SNAr) α, β-Unsaturated carbonyl compounds with a leaving group in the β position are susceptible to addition-elimination reactions. SNAr: Aromatic compounds that are substituted with electron-withdrawing groups undergo nuclephilic aromatic substitution. Favor Unfavor
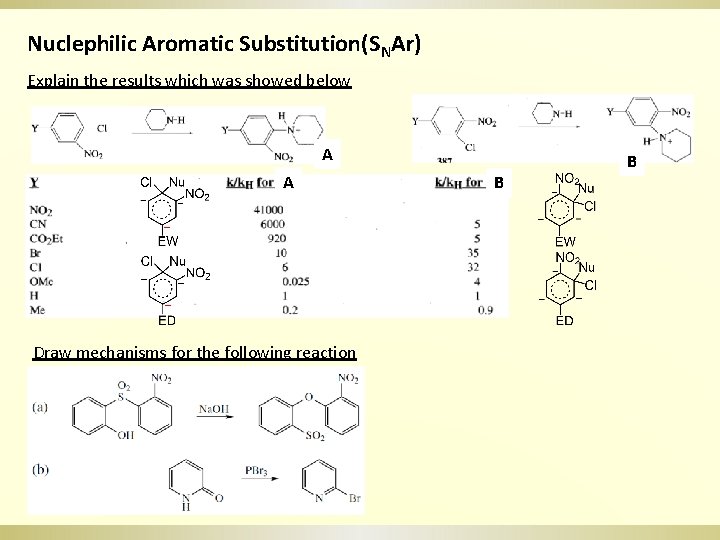
Nuclephilic Aromatic Substitution(SNAr) Explain the results which was showed below A A Draw mechanisms for the following reaction B B
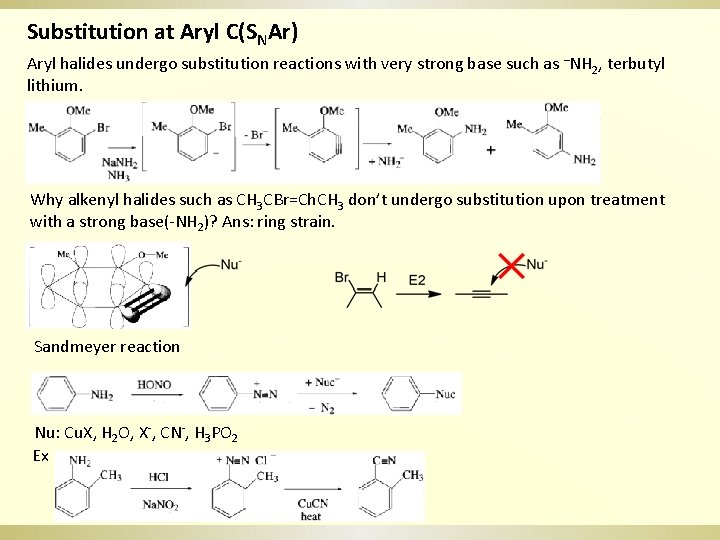
Substitution at Aryl C(SNAr) Aryl halides undergo substitution reactions with very strong base such as –NH 2, terbutyl lithium. Why alkenyl halides such as CH 3 CBr=Ch. CH 3 don’t undergo substitution upon treatment with a strong base(-NH 2)? Ans: ring strain. Sandmeyer reaction Nu: Cu. X, H 2 O, X-, CN-, H 3 PO 2 Ex

Polar Reaction Under Basic Conditions a. Substitution and Elimination at C(sp 3)-X σ bonds b. Addition of Nuclepphiles to Electrophilic π bonds c. Substitution at C(sp 2)-X σ bonds d. Base-promoted Rearrangements
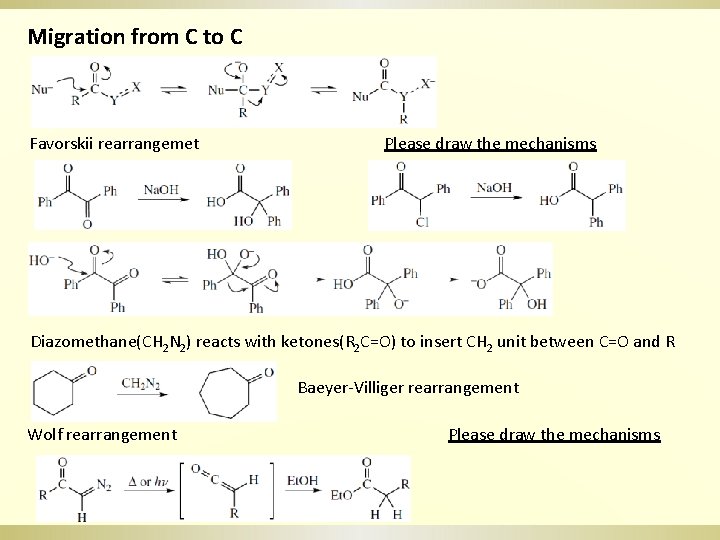
Migration from C to C Favorskii rearrangemet Please draw the mechanisms Diazomethane(CH 2 N 2) reacts with ketones(R 2 C=O) to insert CH 2 unit between C=O and R Baeyer-Villiger rearrangement Wolf rearrangement Please draw the mechanisms
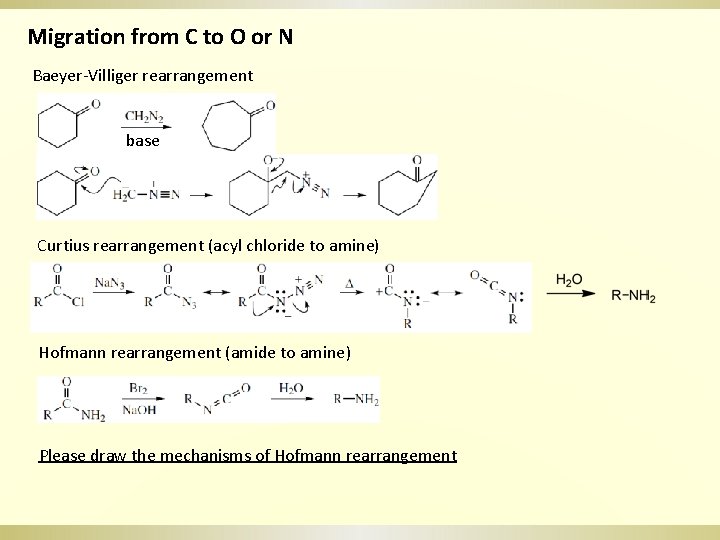
Migration from C to O or N Baeyer-Villiger rearrangement base Curtius rearrangement (acyl chloride to amine) Hofmann rearrangement (amide to amine) Please draw the mechanisms of Hofmann rearrangement
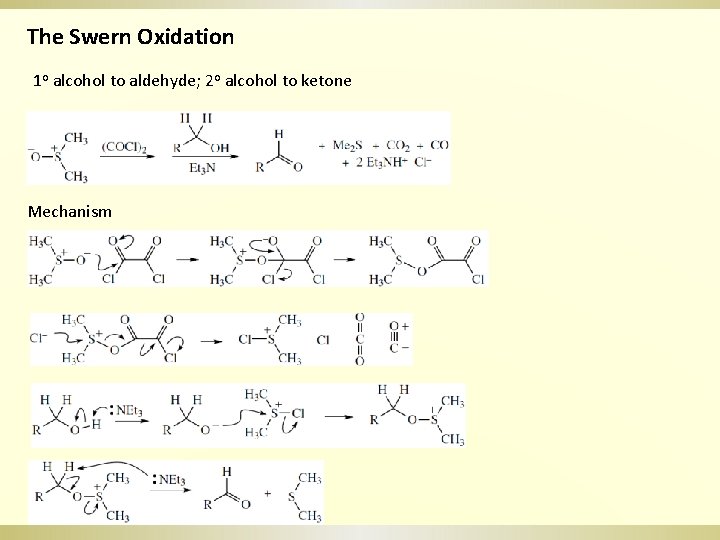
The Swern Oxidation 1 o alcohol to aldehyde; 2 o alcohol to ketone Mechanism
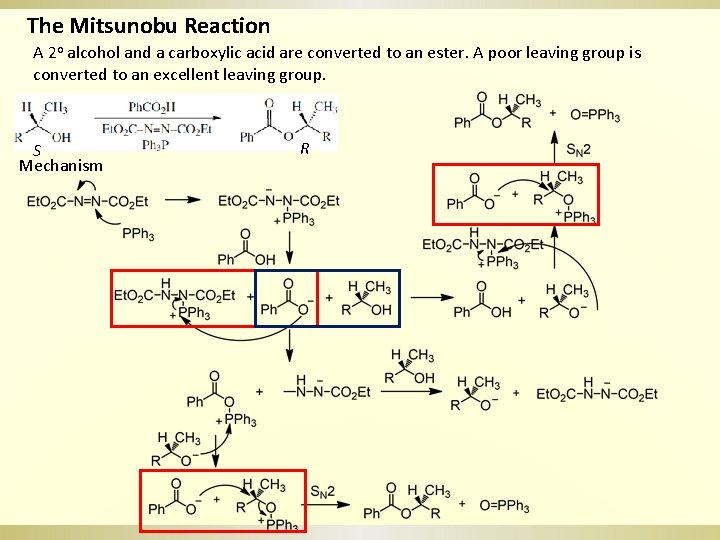
The Mitsunobu Reaction A 2 o alcohol and a carboxylic acid are converted to an ester. A poor leaving group is converted to an excellent leaving group. S Mechanism R
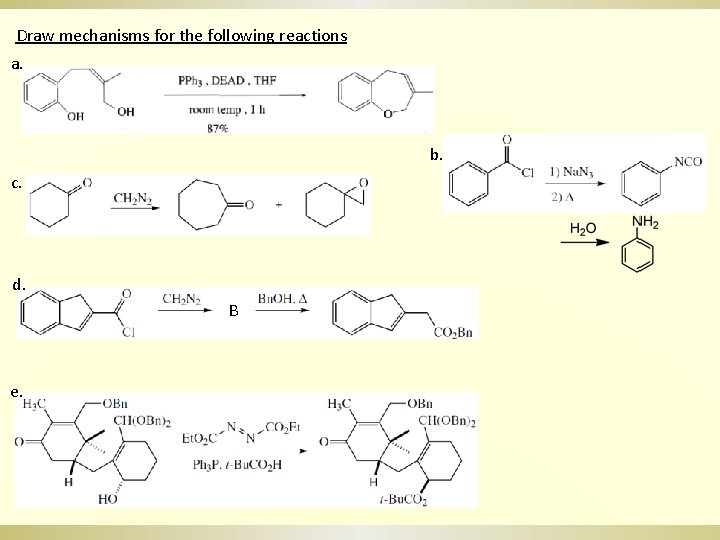
Draw mechanisms for the following reactions a. b. c. d. B e.
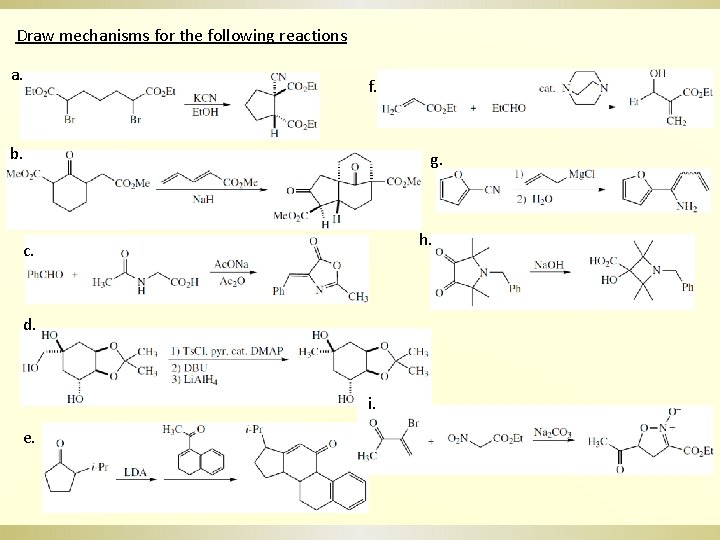
Draw mechanisms for the following reactions a. f. b. g. h. c. d. i. e.

Thanks For Your Attention

- Slides: 33