Chapter TwentyOne Lipid Metabolism Lipids are Involved in

Chapter Twenty-One Lipid Metabolism

Lipids are Involved in Generation and Storage of Energy • The oxidation of fatty acids (FA)in triacylglycerols are the principal storage form of energy for most organisms • Their carbon chains are in a highly reduced form • The energy yield per gram of fatty acid oxidized is greater than that per gram of carbohydrate oxidized
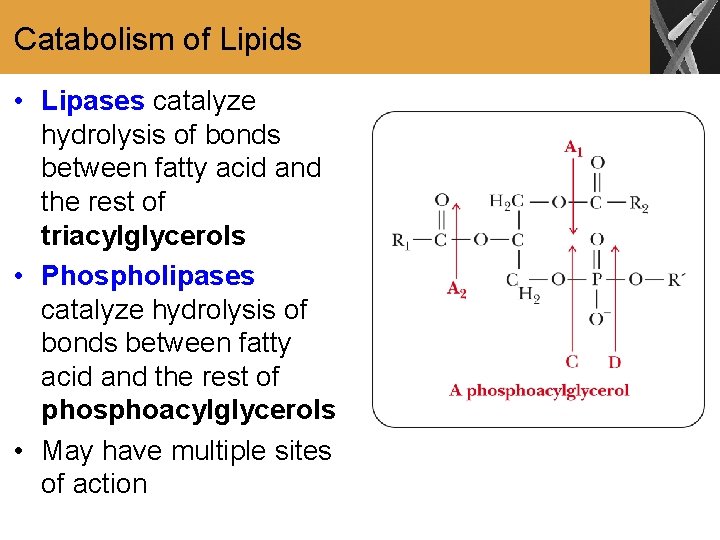
Catabolism of Lipids • Lipases catalyze hydrolysis of bonds between fatty acid and the rest of triacylglycerols • Phospholipases catalyze hydrolysis of bonds between fatty acid and the rest of phosphoacylglycerols • May have multiple sites of action
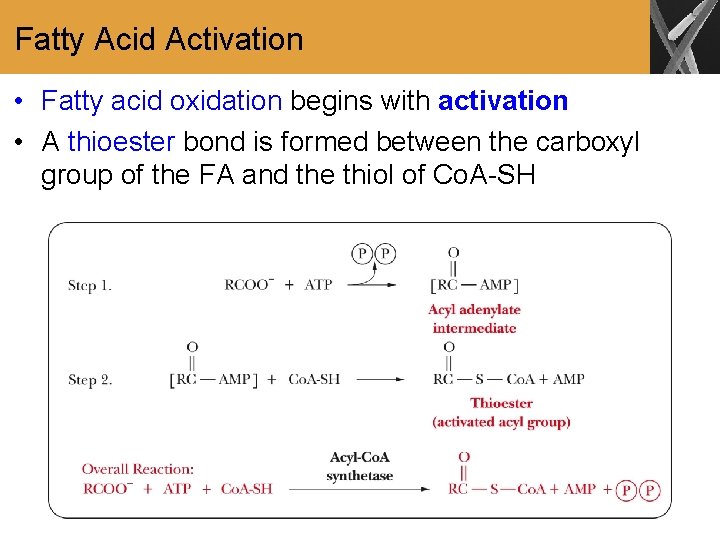
Fatty Acid Activation • Fatty acid oxidation begins with activation • A thioester bond is formed between the carboxyl group of the FA and the thiol of Co. A-SH
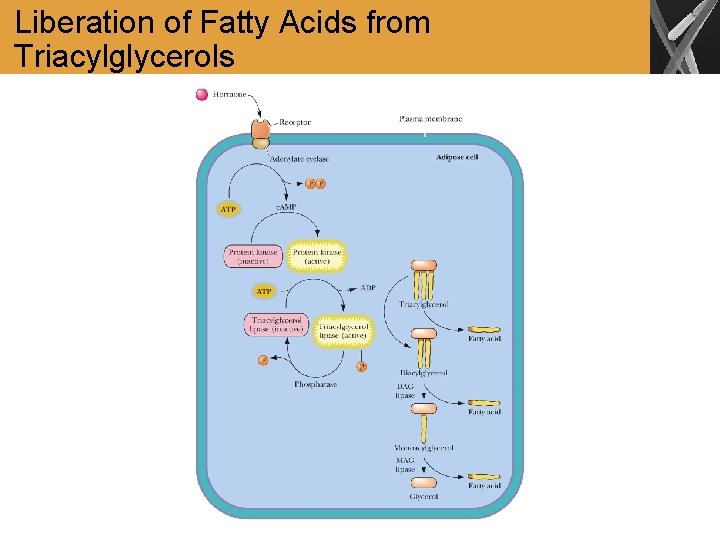
Liberation of Fatty Acids from Triacylglycerols

The Role of Carnitine in Acyl-Co. A Transfer • The acyl-Co. A crosses the outer mitochondrial membrane, but not the inner membrane • The acyl group is then transferred to carnitine, carried across the inner mitochondrial membrane, and transferred to mitochondrial Co. A-SH • Carnitine Palmitoyltransferase I (CPT-I, carnitine acyltransferase I) has specificity for acyl groups between 14 and 18 carbons long
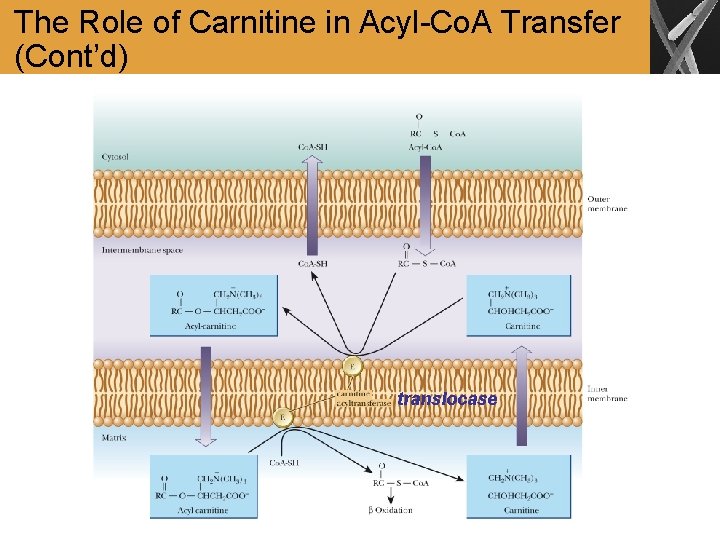
The Role of Carnitine in Acyl-Co. A Transfer (Cont’d) translocase

-Oxidation • -Oxidation: a series of reactions that cleaves carbon atoms two at a time from the carboxyl end of a fatty acid • The complete cycle of one -oxidation requires four enzymes • Reaction 1: Oxidation of the , carbon-carbon single bond to a carbon-carbon double bond • Reaction 2: Hydration of the carbon-carbon double bond • Reaction 3: Oxidation of the -hydroxyl group to a carbonyl group • Reaction 4: Cleavage of the carbon chain by a reverse Claisen reaction
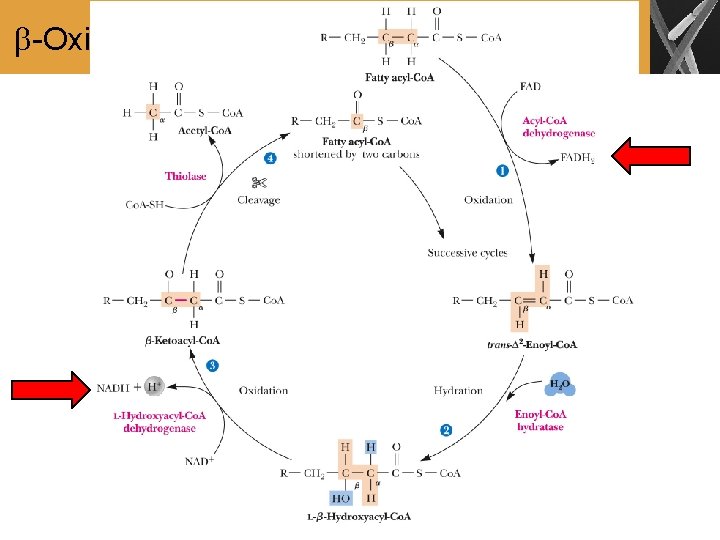
-Oxidation (Cont’d)
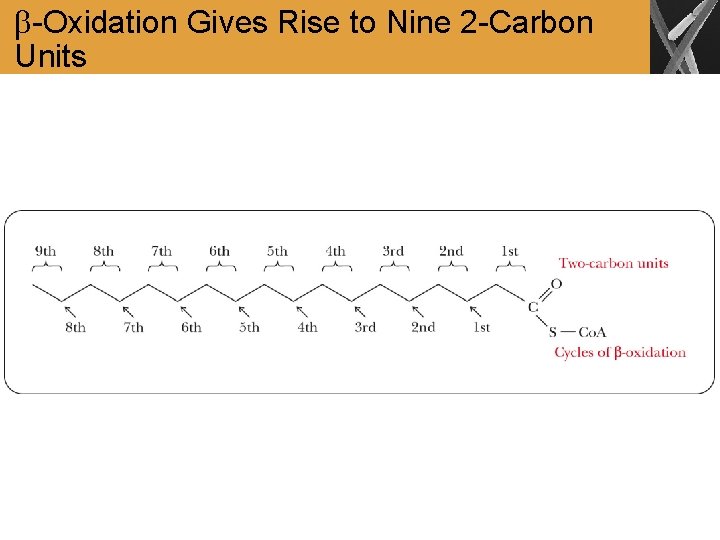
-Oxidation Gives Rise to Nine 2 -Carbon Units

Summary • Fatty acids are activated and transported to the mitochondrial matrix for further catabolism • The breakdown of fatty acids takes place in the mitochondrial matrix and proceeds by successive removal of two-carbon units as acetyl-Co. A • Each Cleavage of a two-carbon moiety requires a four-step reaction sequences called -oxidation

Energy Yield from FA Oxidation • The energy released by the oxidation of acetyl-Co. A formed by -oxidation of FA can be used to produce ATP • Eight cycles of -oxidation are required for the oxidation of Stearic acid to acetyl-Co. A

Energy Yield from FA Oxidation (Cont’d) • The overall equation for oxidation of stearic acid can be obtained by adding the equations for -oxidation, the citric acid cycle, and oxidative phosphorylation
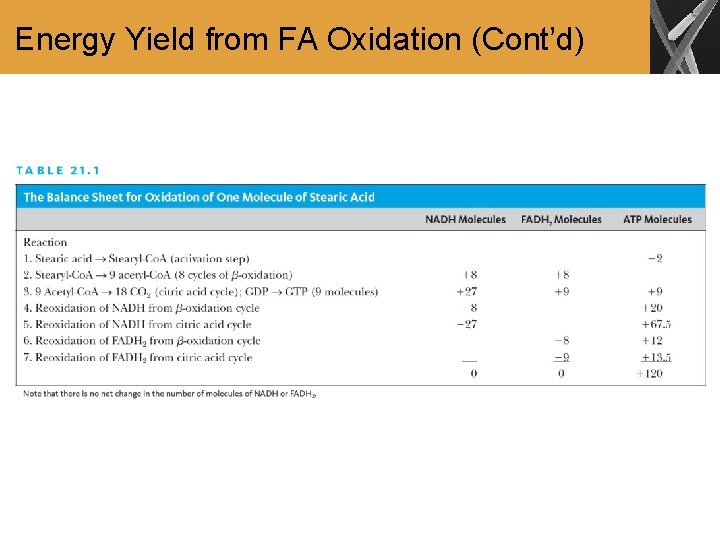
Energy Yield from FA Oxidation (Cont’d)

Summary • The complete oxidation of FA by the citric acid cycle and the electron transport chain releases large amounts of energy • When we include the reoxidation of NADH and FADH 2 from -oxidation and the citric acid cycle, we obtain a net yield of 120 ATP for a single molecule of stearic acid
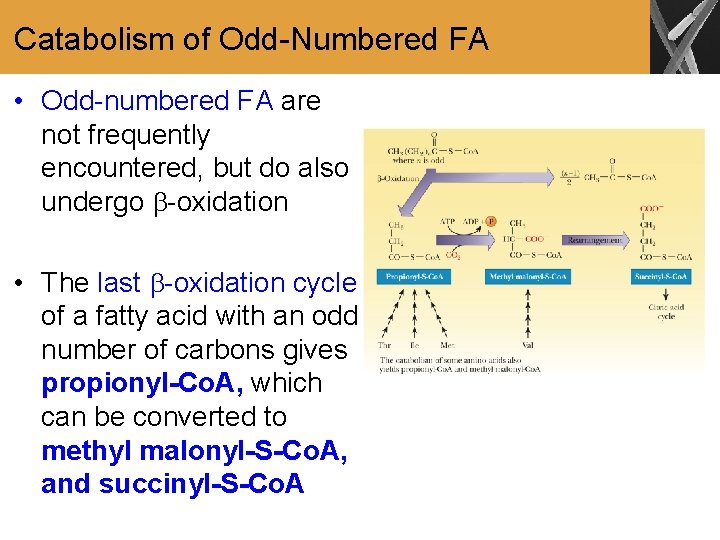
Catabolism of Odd-Numbered FA • Odd-numbered FA are not frequently encountered, but do also undergo -oxidation • The last -oxidation cycle of a fatty acid with an odd number of carbons gives propionyl-Co. A, which can be converted to methyl malonyl-S-Co. A, and succinyl-S-Co. A

Oxidation of an Unsaturated FA • A cis-trans isomerization is needed to convert unsaturated FA to acetyl-Co. A • This enzyme is known as an isomerase • Oxidation of unsaturated FA does not generate as much ATP relative to saturated FA with the same # of carbons
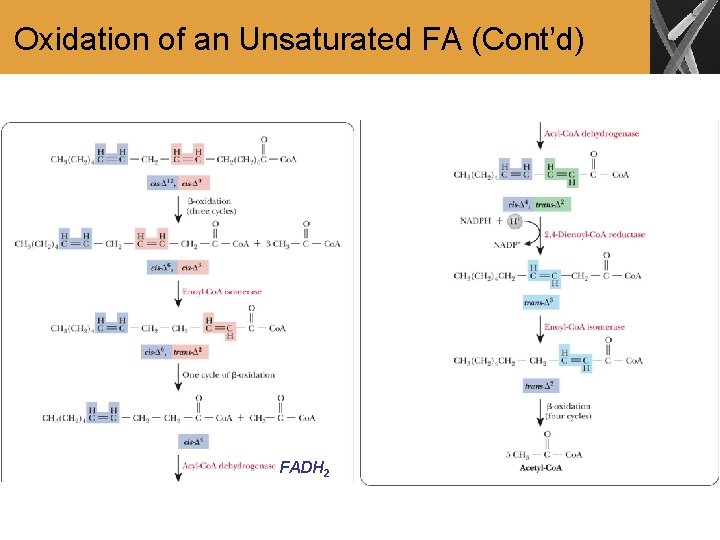
Oxidation of an Unsaturated FA (Cont’d) FADH 2

Summary • FA with odd number of carbons produce propionyl. Co. A in the last step of the oxidation • Propionyl-Co. A can be converted to succinyl-Co. A, which plays a role in the citric acid cycle • The oxidation of unsaturated FA requires enzymes that catalyze isomerization around the double bonds so that oxidation can proceed

Ketone Bodies • Formation of ketone bodies occurs when the amount of acetyl-Co. A produced is excessive compared to the amount of oxaloacetate available to react with it • Intake high in lipids and low in carbohydrates • Diabetes not suitably controlled • Starvation • Ketone bodies are: are acetone, -hydroxybutyrate, and acetoacetate • Formed principally in liver mitochondria • Can be used as a fuel in most tissues and organs
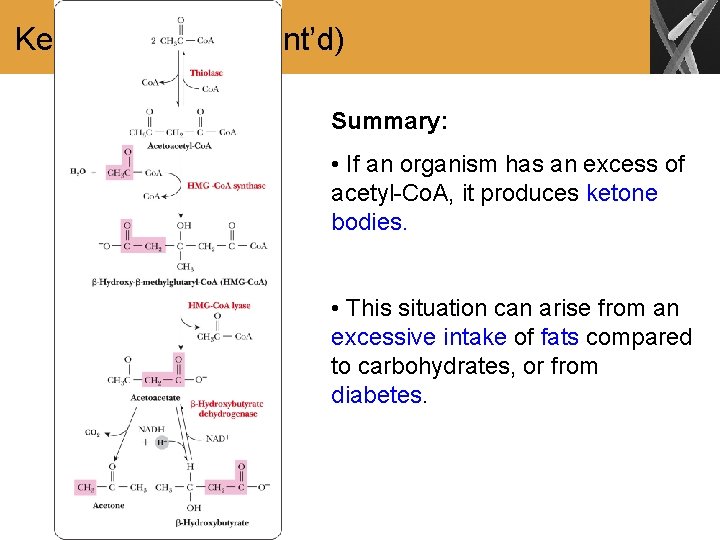
Ketone Bodies (Cont’d) Summary: • If an organism has an excess of acetyl-Co. A, it produces ketone bodies. • This situation can arise from an excessive intake of fats compared to carbohydrates, or from diabetes.
Fatty Acid Biosynthesis • Biosynthesis is not exact reversal of oxidation • Biosynthetic reactions occur in the cytosol
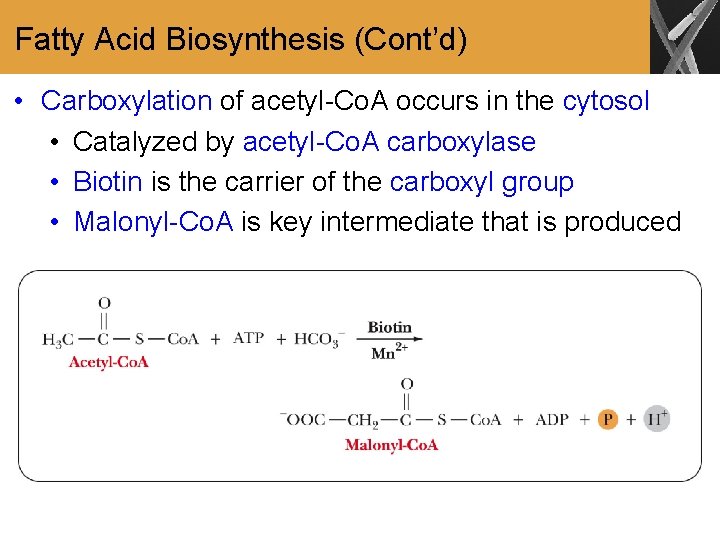
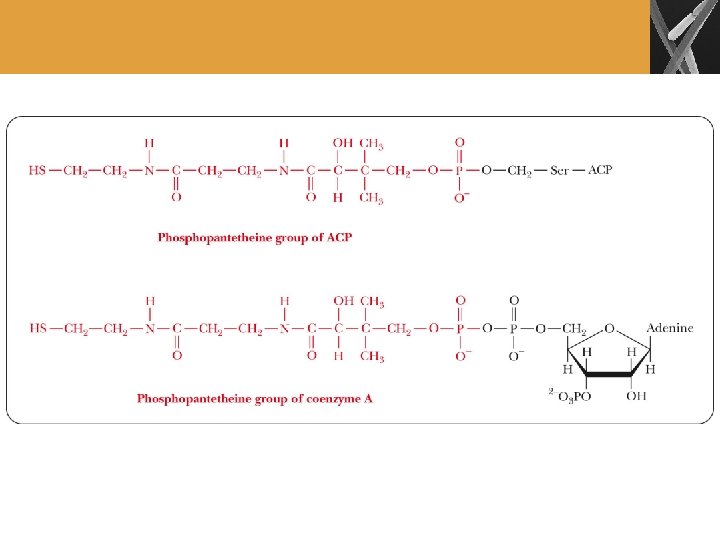
Fatty Acid Biosynthesis (Cont’d) • Carboxylation of acetyl-Co. A occurs in the cytosol • Catalyzed by acetyl-Co. A carboxylase • Biotin is the carrier of the carboxyl group • Malonyl-Co. A is key intermediate that is produced
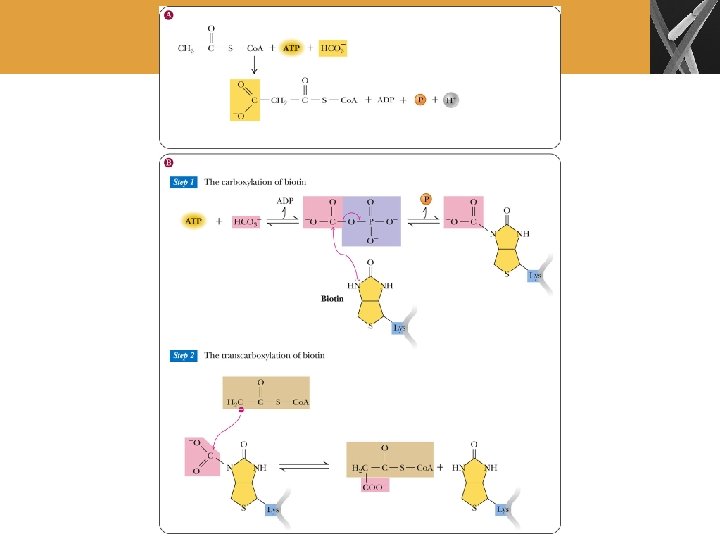
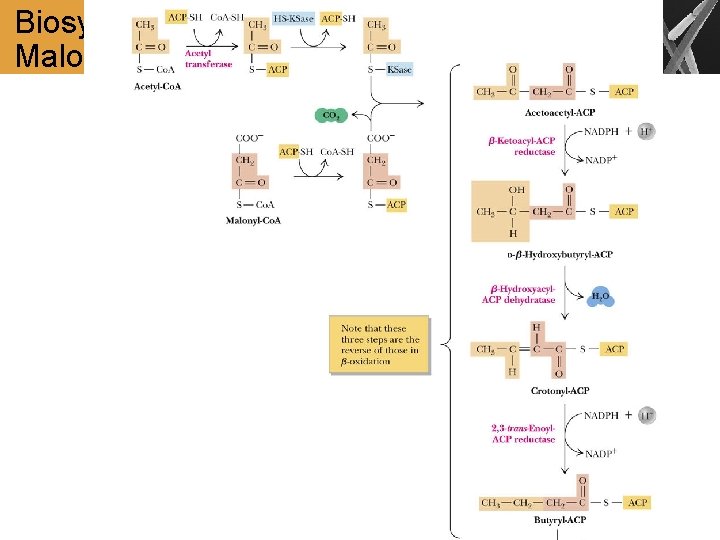
Biosynthesis of Palmitate from Acetyl- and Malonyl-Co. A
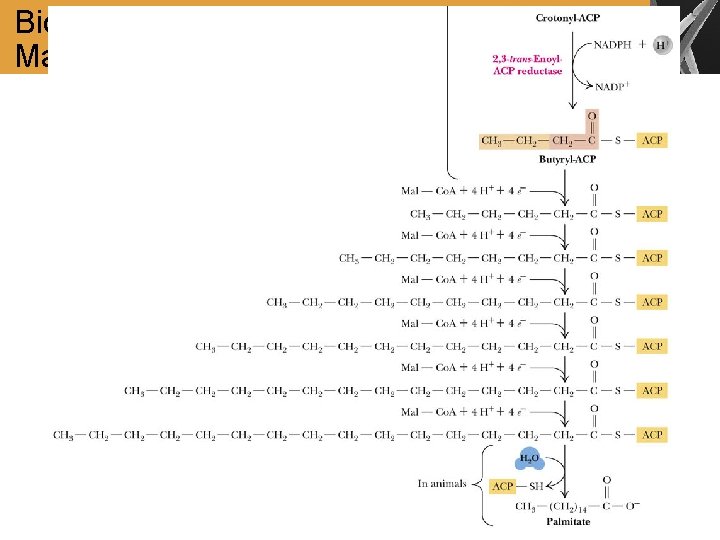
Biosynthesis of Palmitate from Acetyl- and Malonyl-Co. A
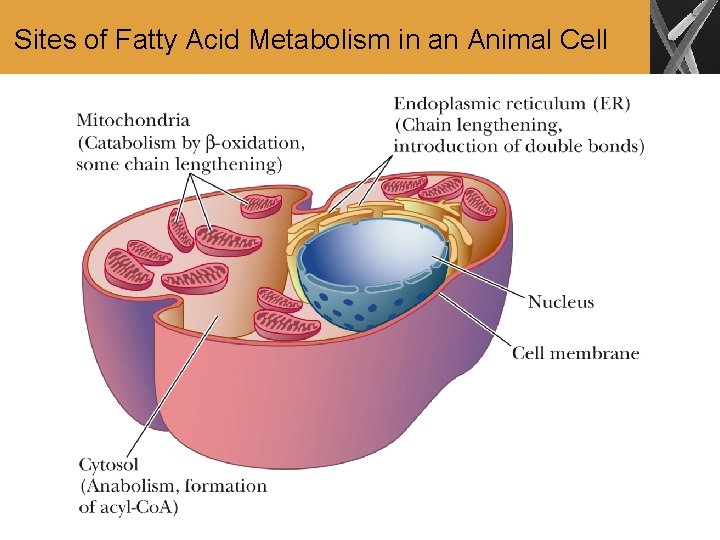
Sites of Fatty Acid Metabolism in an Animal Cell

Summary • Acetyl-Co. A is transported to the cytosol and converted to malonyl-Co. A • The biosynthesis of FA proceeds by the addition of 2 -carbon units to the hydrocarbon chain. The process is catalyzed by the fatty-acid synthase complex Comparison of FA Degradation and Biosynthesis
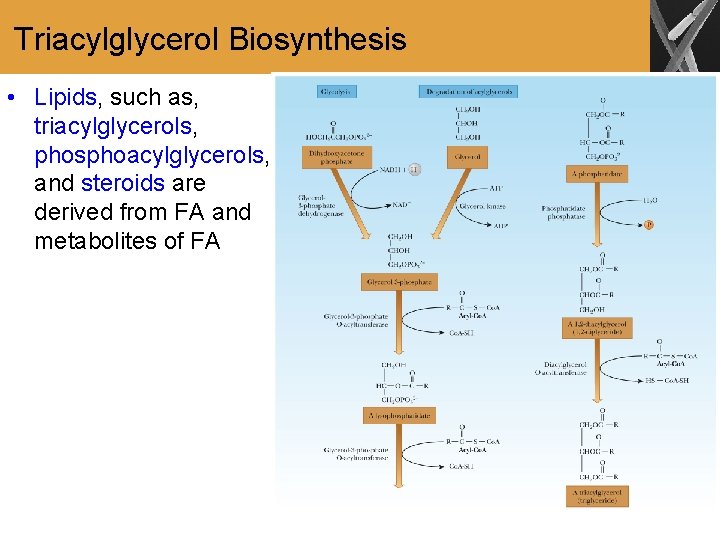
Triacylglycerol Biosynthesis • Lipids, such as, triacylglycerols, phosphoacylglycerols, and steroids are derived from FA and metabolites of FA
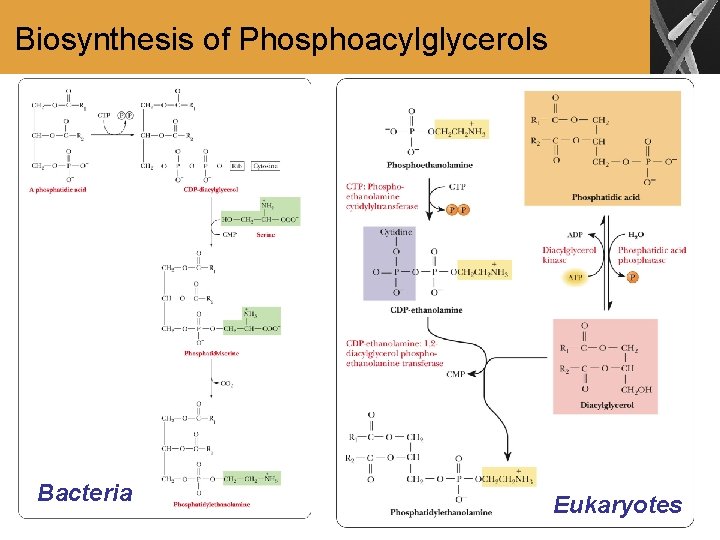
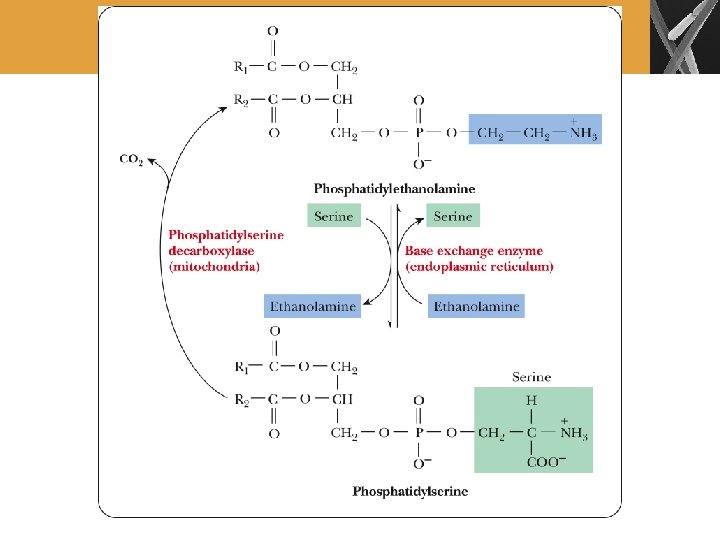
Biosynthesis of Phosphoacylglycerols Bacteria Eukaryotes
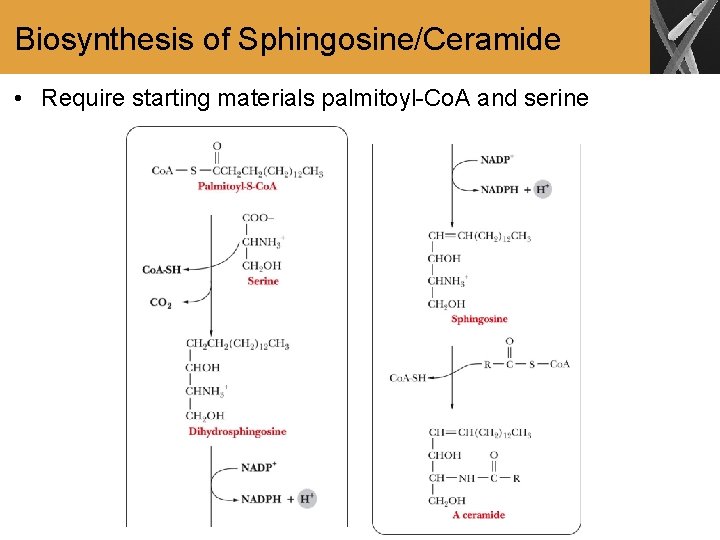
Biosynthesis of Sphingosine/Ceramide • Require starting materials palmitoyl-Co. A and serine

Cholesterol Biosynthesis • All carbon atoms of cholesterol and steroids synthesized from it are derived from the two-carbon acetyl group of acetyl-Co. A • Involves many reaction steps • Involvement of isoprene units are key to the biosynthesis of steroids and other biomolecules known as terpenes
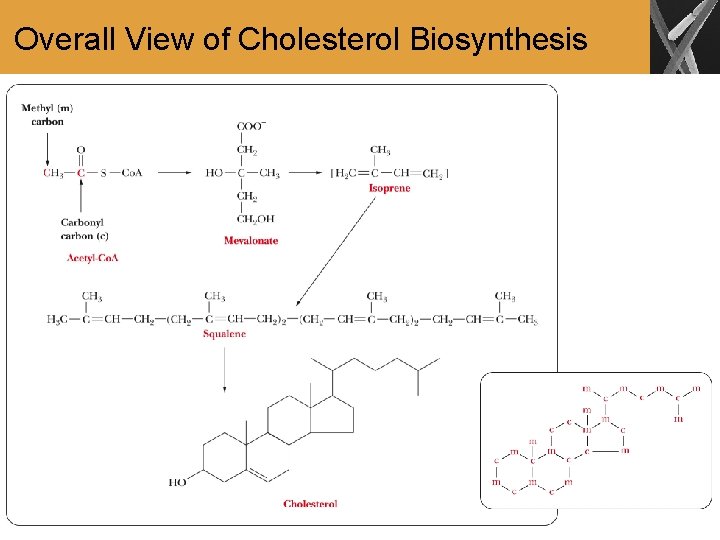
Overall View of Cholesterol Biosynthesis

Cholesterol Biosynthesis (Cont’d) • Synthesis begins with the condensation of two molecules of acetyl-Co. A • Next, condensation with a third molecule of acetyl-Co. A • The formation of mevalonate is completed by reduction of the thioester that gives a 1° alcohol Committed step See ketone body at p 601

Mevalonate to Squalene • The pyrophosphorylation of the 1° alcohol of mevalonate (two moles of ATP) is followed by phosphorylation of the 3° alcohol (one mole of ATP), then the concerted decarboxylation and elimination of phosphate ion gives isopentenyl pyrophosphate • Then there is an enzyme-catalyzed isomerization of the carbon-carbon double bond that gives dimethylallyl pyrophosphate • Dimethylallyl pyrophosphate is then reacted with isopentyl pyrophosphate, which is followed by H+ loss to give farnesyl pyrophosphate • The joining together of two units of farnesyl pyrophosphate (C 15) units by a two-electron oxidation gives squalene (C 30)
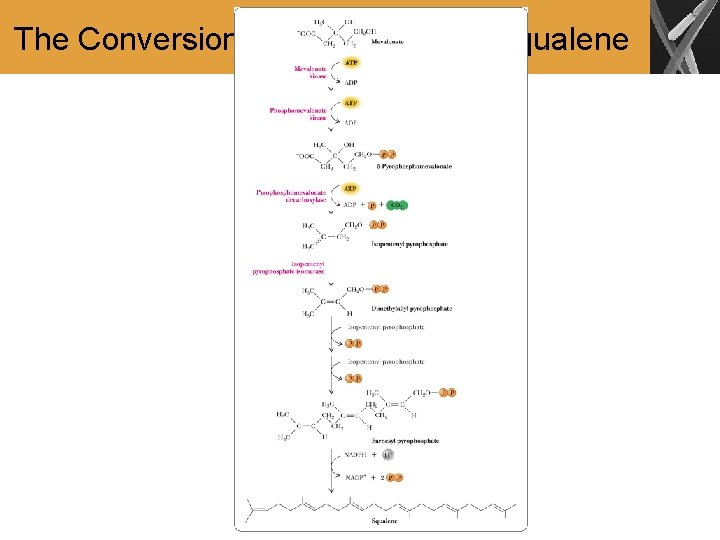
The Conversion of Mevalonate to Squalene
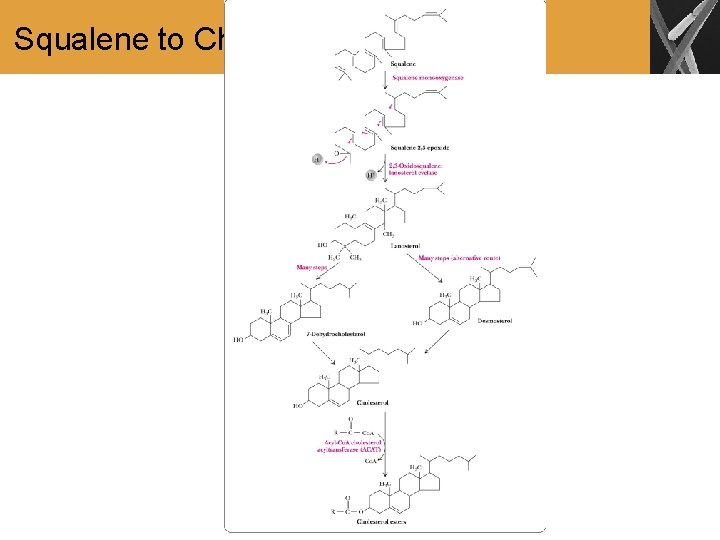
Squalene to Cholesterol
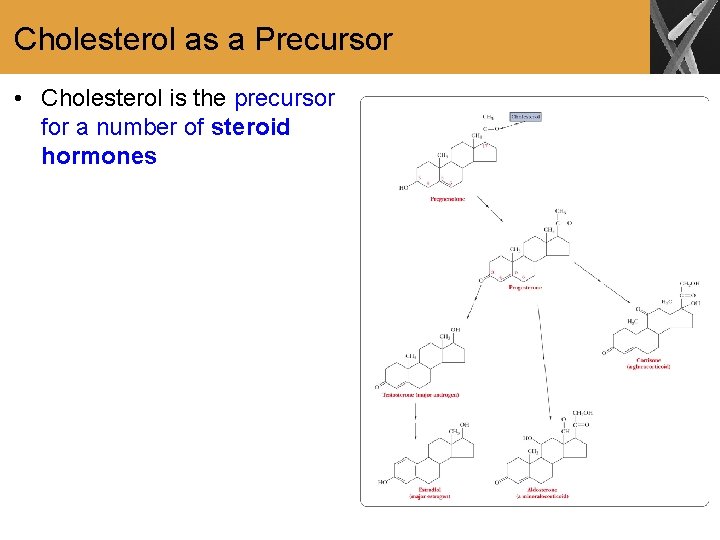
Cholesterol as a Precursor • Cholesterol is the precursor for a number of steroid hormones

Role of Cholesterol in Heart Disease • Lipids are transported in the blood stream by lipoproteins • Cholesterol and its fatty acid esters are packaged into several classes of lipoproteins for transport
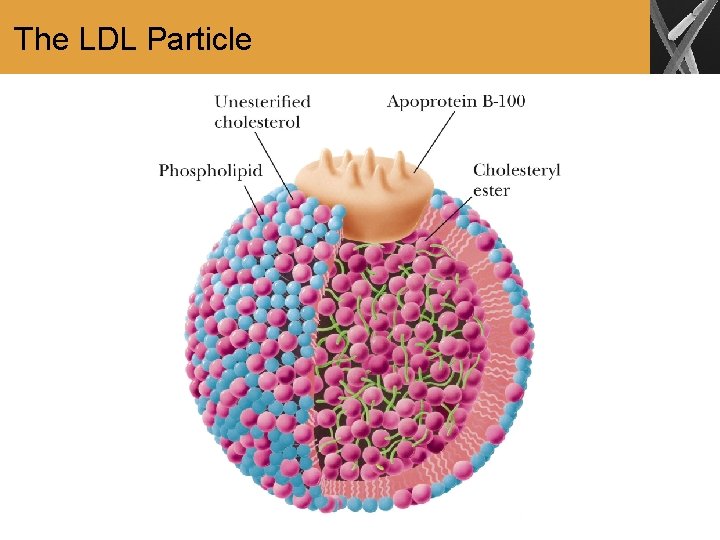
The LDL Particle
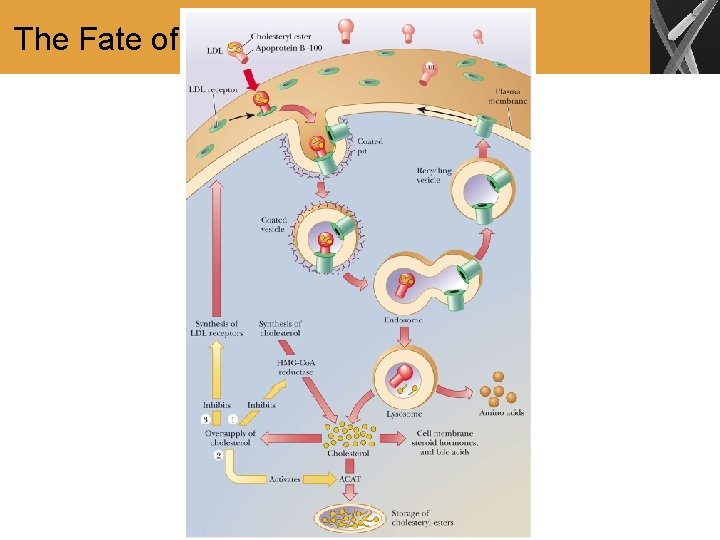
The Fate of Cholesterol

Summary • The biosynthesis of cholesterol proceeds by the condensation of five-carbon isoprenoid units • Isoprenoid units in turn are derived from the reaction of three acetyl-Co. A units • Once cholesterol is formed, it serves as a precursor for other steroids • Cholesterol must be packaged for transport in the bloodstream. Some of these forms of cholesterol play a role in heart disease

• Problem set • 1, 3, 5, 11, 12, 14, 18, 20, 21, 24, 25, 28, 30, 37, 38, 44, 47, 48 • Homework (Due day: Nov, 26 th) • Figure 21. 3, Figure 21. 4, Figure 21. 5, Figure 21. 6, Figure 21. 11, Figure 21. 15, Figure 21. 24
- Slides: 45