Chapter Thirteen The Behavior of Gases 13 1


Chapter Thirteen: The Behavior of Gases Ø 13. 1 Gases, Pressure, and the Atmosphere Ø 13. 2 The Gas Laws

13. 1 What’s in Earth’s atmosphere? Ø Nitrogen (N 2) gas makes up about 78 percent of Earth’s atmosphere. Ø Nitrogen is released into the air by volcanoes and decaying organisms and is a vital element for living things.

13. 1 Comparing atmospheres ØAn atmosphere is a layer of gases surrounding a planet or other body in space.

13. 1 Life changed Earth’s atmosphere ØOver time, photosynthesis breaks down carbon dioxide, uses carbon to build the organism, and releases oxygen into the air.

13. 1 Life changed Earth’s atmosphere ØEarth stores carbon as calcium carbonate so it doesn’t return to the atmosphere. Ø“Fossil fuels” (oil, coal, and natural gas) are carbon from decaying plants and animals in the ground.

13. 1 Atmospheric pressure ØAtmospheric pressure is a measurement of the force of air molecules in the atmosphere at a given altitude. ØYour ear drum is one way you can detect changes in pressure.

13. 1 Pressure in the atmosphere ØAt sea level, the weight of the column of air above a person is about 9, 800 newtons (2, 200 pounds)! ØThis is equal to the weight of a small car. ØWhy aren’t we crushed by this pressure?

13. 1 Measuring Pressure ØA barometer is an instrument that measures atmospheric pressure. ØMercury barometers were common until we discovered their vapors were harmful.

13. 1 Measuring Pressure ØToday we use aneroid barometers. ØThey have an airtight cylinder made of thin metal. ØThe walls of the cylinder respond to changes in pressure.

13. 1 Pressure in the atmosphere ØThe gas molecules closest to Earth’s surface are packed together very closely. ØThis means pressure is lower the higher up you go into the atmosphere.
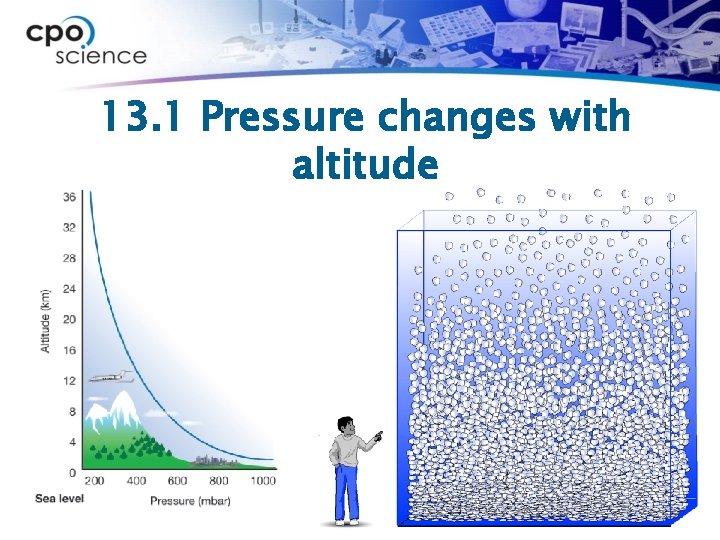
13. 1 Pressure changes with altitude

13. 1 Units of pressure

13. 2 Boyle’s Law Ø When you squeeze a fixed quantity of gas into a smaller volume the pressure goes up. Ø This rule is known as Boyle’s law.

13. 2 Boyle’s Law

Solving Problems ØA kit used to fix flat tires consists of an aerosol can containing compressed air and a patch to seal the hole in the tire. ØSuppose 5 liters of air at atmospheric pressure (1 atm) is compressed into a 0. 5 liter aerosol can. What is the pressure of the compressed air in the can? ØAssume no change in temperature or mass.

Solving Problems 1. Looking for: Ø …final pressure in atmospheres (P 2) 2. Given Ø …V 1 = 5 L , P 1= 1 atm, V 2 =. 5 L 3. Relationships: Ø Boyle’s Law: P 1 V 1 = P 2 V 2 4. Solution Ø Rearrange equation so P 2 = P 1 V 1 / V 2 Ø P 2 = 1 atm x 5. 0 L/ 0. 5 L = 10 atm.

13. 2 Pressure and Temperature ØThe pressure of a gas is affected by temperature changes. ØIf the mass and volume are kept constant, the pressure goes up when the temperature goes up, and down when the temperature goes down.

13. 2 Gay-Lussac’s Law

13. 2 Pressure and Temperature ØAny time we apply gas laws, the temperature must be in Kelvins. ØOnly the Kelvin scale starts from absolute zero, when energy of particles is theoretically zero.

13. 2 Charles’ Law ØAccording to Charles’ law, the volume of a gas increases with increasing temperature. ØVolume decreases with decreasing temperature.

13. 2 Charles’ Law ØA hot-air balloon floats because the air inside is less dense than the air outside.

Solving Problems ØA can of hair spray has a pressure of 300 psi at room temperature 21°C. ØThe can is accidentally moved too close to a fire and its temperature increases to 295°C. ØWhat is the final pressure in the can? (Round answer to nearest whole number. )

Solving Problems 1. Looking for: Ø …final pressure in atmospheres (P 2) 2. Given Ø …P 1= 300 atm, T 1 = 21 C , T 2 = 295 C 3. Relationships: Ø Convert temps using K = C + 273 Ø Charles’ Law: P 1/T 1 = P 2/T 2 4. Solution Ø Rearrange equation so P 2 = P 1 x. T 2 / T 1 Ø P 2 = 300 atm. x 568 K / 294 K = 580 atm.
- Slides: 24