CHAPTER Thermodynamic Property Relations 1 A Little Math














- Slides: 29

CHAPTER Thermodynamic Property Relations 1. A Little Math – Partial Derivatives 2. The Maxwell Relations 3. The Clapeyron Equation 4. General Relations for du, dh, ds, Cv and Cp 5. The Joule-Thomson Coefficient 6. The Dh, Du and Ds of Real Gases 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. 12 Chung H. Jeon

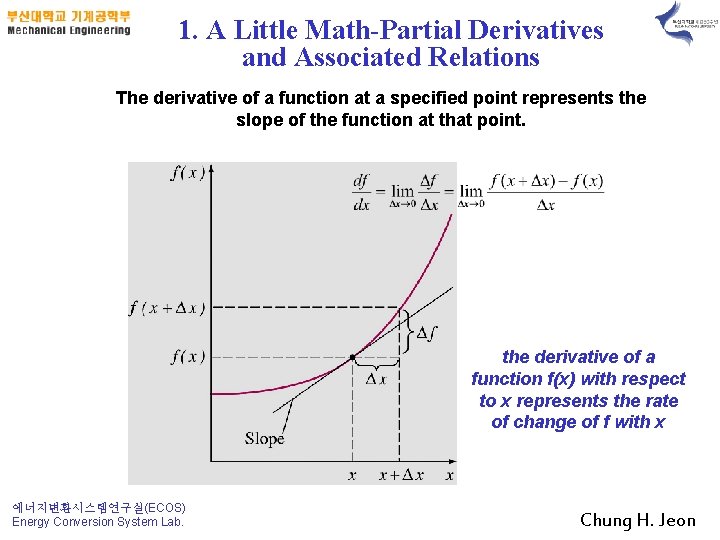
1. A Little Math-Partial Derivatives and Associated Relations The derivative of a function at a specified point represents the slope of the function at that point. the derivative of a function f(x) with respect to x represents the rate of change of f with x 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

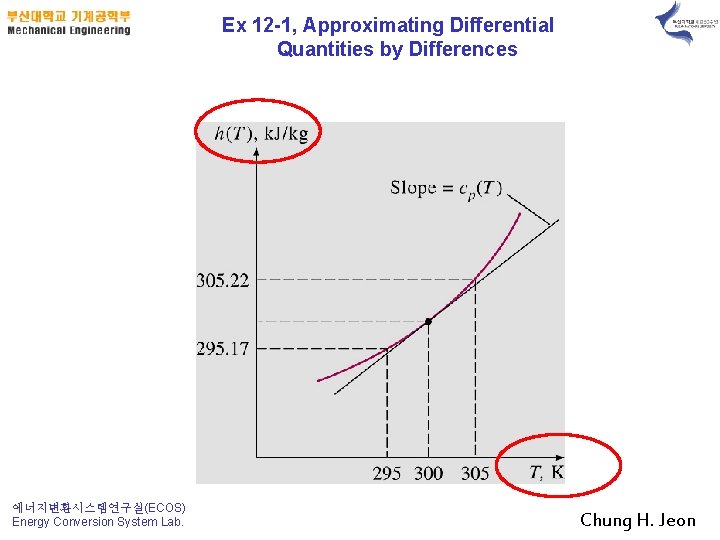
Ex 12 -1, Approximating Differential Quantities by Differences 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

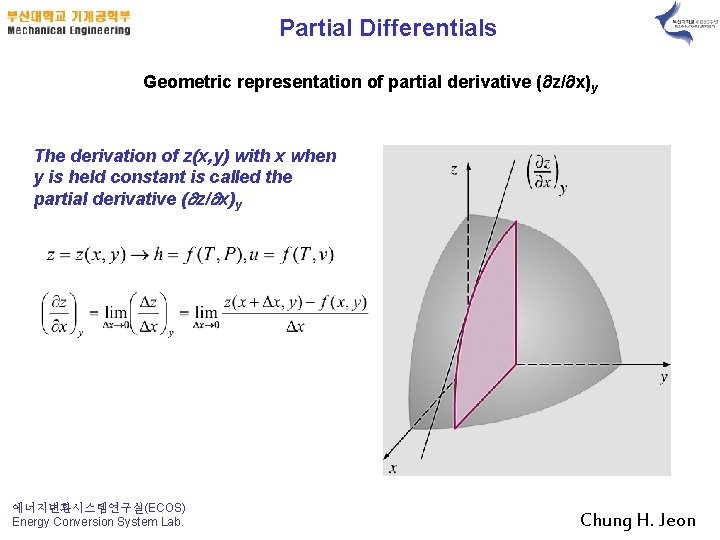
Partial Differentials Geometric representation of partial derivative ( z/ x)y The derivation of z(x, y) with x when y is held constant is called the partial derivative ( z/ x)y 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

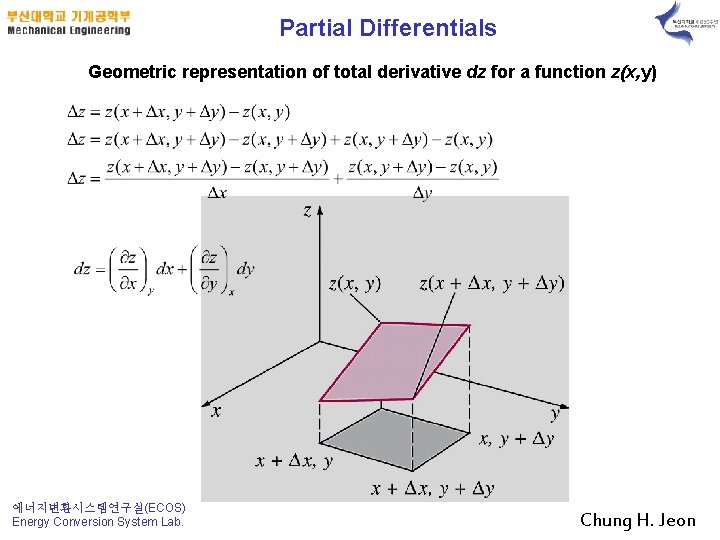
Partial Differentials Geometric representation of total derivative dz for a function z(x, y) 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

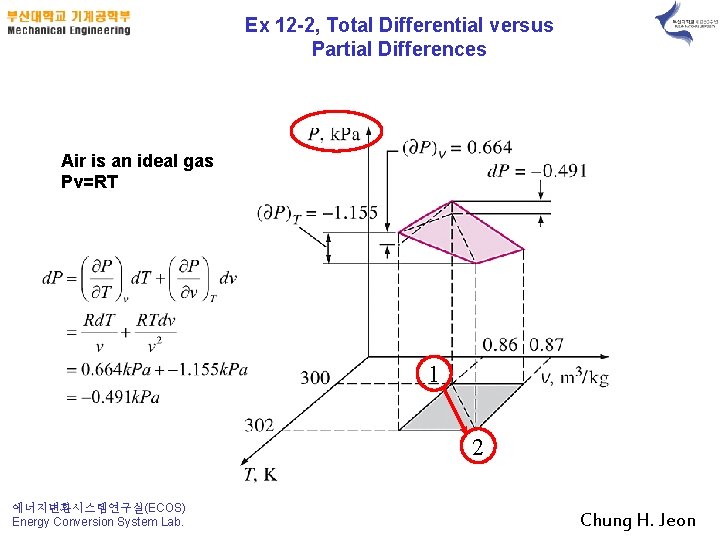
Ex 12 -2, Total Differential versus Partial Differences Air is an ideal gas Pv=RT 1 2 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Partial Differential Relations Reciprocity or cyclic relation 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Demonstration of the reciprocity relation for the function z + 2 xy - 3 y 2 z = 0. 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

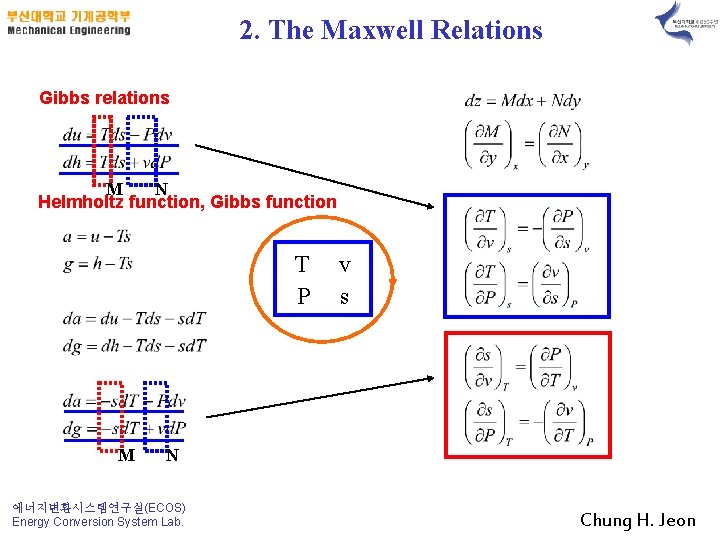
2. The Maxwell Relations Gibbs relations M N Helmholtz function, Gibbs function T P M v s N 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

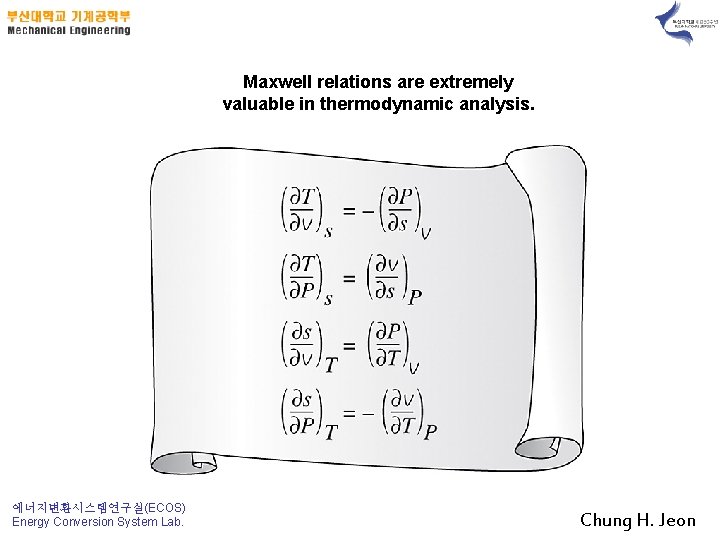
Maxwell relations are extremely valuable in thermodynamic analysis. 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

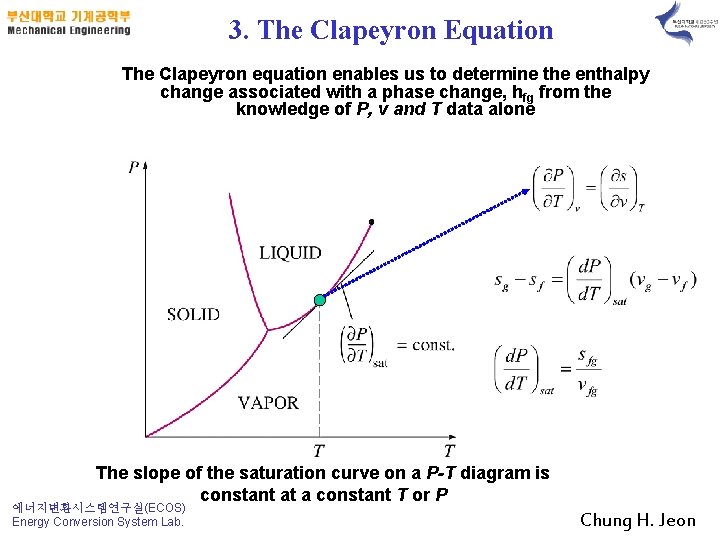
3. The Clapeyron Equation The Clapeyron equation enables us to determine the enthalpy change associated with a phase change, hfg from the knowledge of P, v and T data alone The slope of the saturation curve on a P-T diagram is constant at a constant T or P 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

At low pressure vg>>vf, vfg=vg vapor is ideal gas, vg=RT/P Clapeyron equation 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Clapeyron-Clausius equation Chung H. Jeon

4. General Relations for du, dh, ds, Cv, and Cp Internal Energy Changes 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Enthalpy Changes 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Entropy Changes 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Specific Heats Cv and Cp M N substitution 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

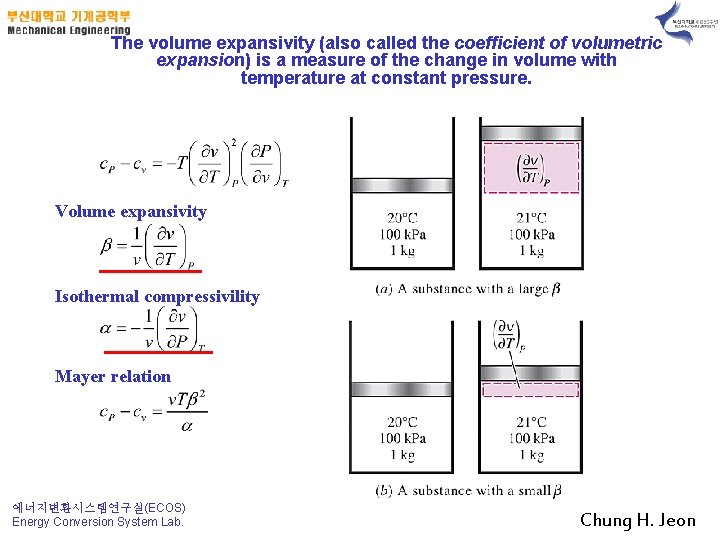
The volume expansivity (also called the coefficient of volumetric expansion) is a measure of the change in volume with temperature at constant pressure. Volume expansivity Isothermal compressivility Mayer relation 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

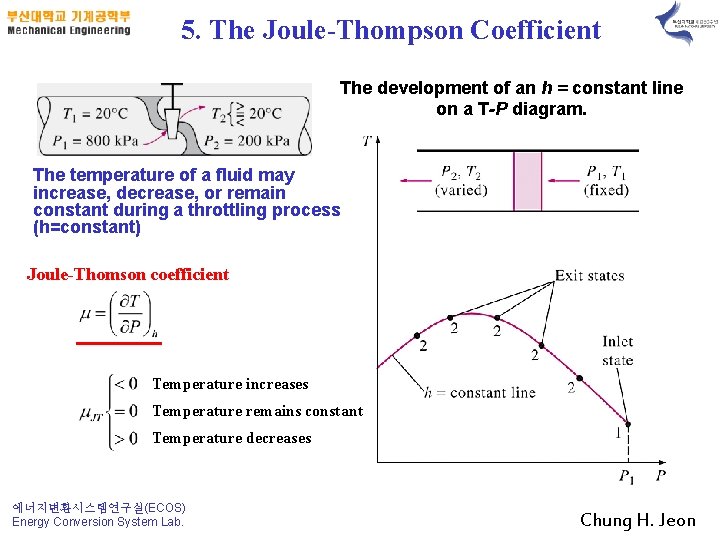
5. The Joule-Thompson Coefficient The development of an h = constant line on a T-P diagram. The temperature of a fluid may increase, decrease, or remain constant during a throttling process (h=constant) Joule-Thomson coefficient Temperature increases Temperature remains constant Temperature decreases 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

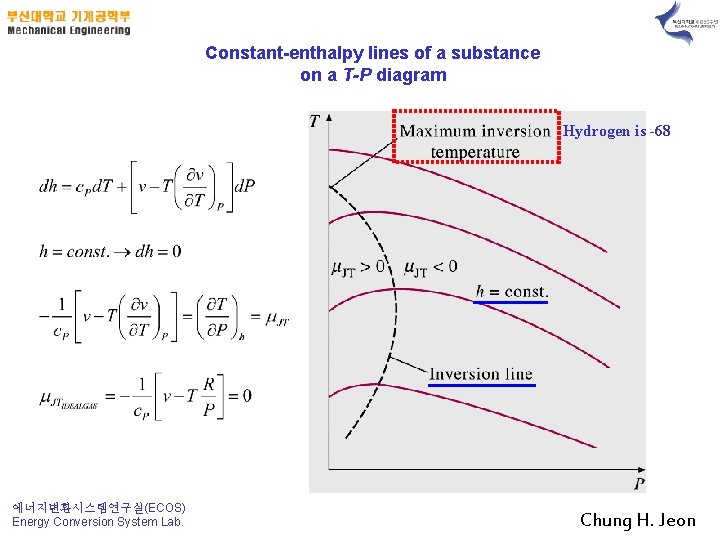
Constant-enthalpy lines of a substance on a T-P diagram Hydrogen is -68 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

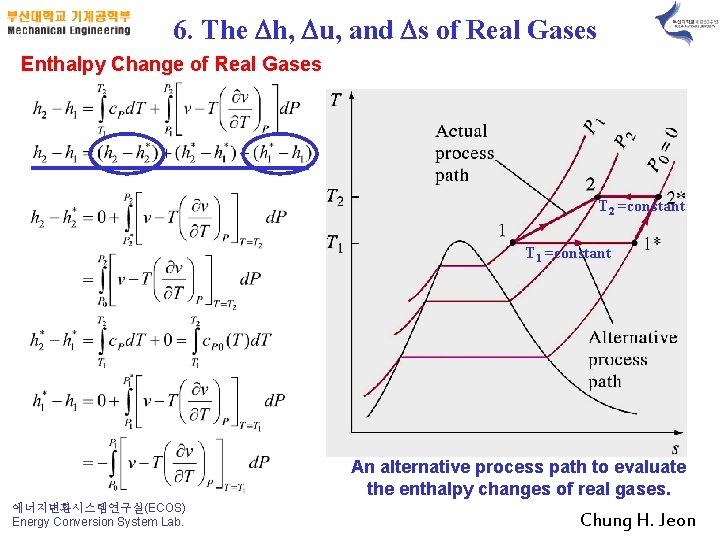
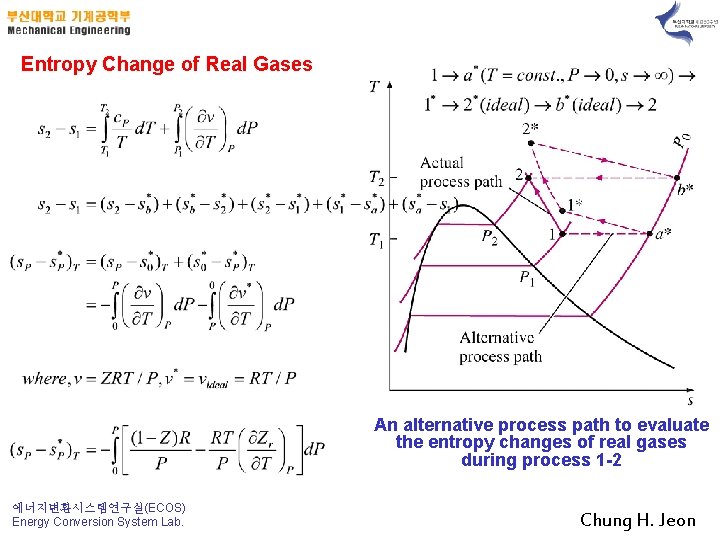
6. The Dh, Du, and Ds of Real Gases Entha. Ipy Change of Real Gases T 2 =constant T 1 =constant An alternative process path to evaluate the enthalpy changes of real gases. 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

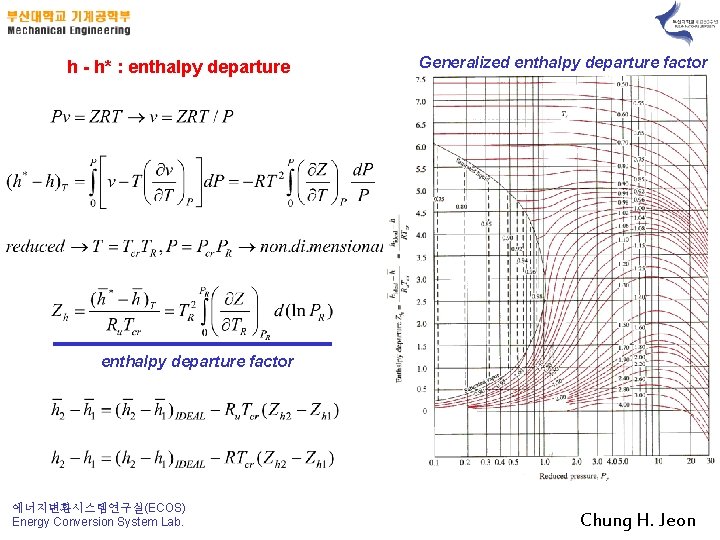
h - h* : entha. Ipy departure Generalized entha. Ipy departure factor 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Internal Energy Change of Real Gases 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

Entropy Change of Real Gases An alternative process path to evaluate the entropy changes of real gases during process 1 -2 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Chung H. Jeon

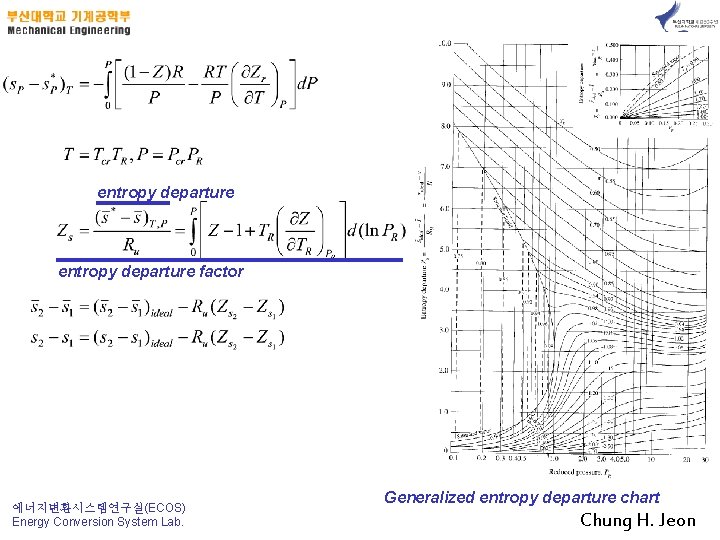
entropy departure factor 에너지변환시스템연구실(ECOS) Energy Conversion System Lab. Generalized entropy departure chart Chung H. Jeon