Chapter Ten Quality assurance in Public health Microbiology

• Chapter Ten: Quality assurance in Public health Microbiology

• Acknowledgment • • Addis Ababa University Haramaya University Hawassa University Jimma University of Gondar ASCP CDC-Ethiopia

• Presentation Outlines • Session objectives • A review of Quality assurance • Components of quality assurance • Quality issues focusing on public health Microbiology • Safety issues in public health microbiology • Summary

• Session objectives At the end of this session students are expected to: • Revise the principle of quality assurance • Differentiate internal and external quality assurance • Describe the preanlytical analytical and postanlytical components of quality assurance from public health point of view. • Take remedial action during discrepant result

Quality Assurance Definition. • All planned and systematic actions necessary to provides adequate confidence that goods or services will satisfy the customer's needs. Or • The right result at , The right time on the right specimens; from the right patient with result interpretation based on correct reference data and at the right prices.

§ It Includes. • Quality control. • Quality assessment. • Quality improvement.

• Laboratory Testing Impacts Nearly Everyone • Accurate, reliable lab testing is essential to all aspects of health care including public health microbiology. • How do we achieve QUALITY ? excellent performance in public health microbiology laboratory?

• Quality is achieved by: • Determining the customer precise requirements • Ensuring that all resources, facilities and skills required to meet the customer requirements are available from the public health microbiology points. • Planning, documenting and implementing management procedures to ensure that the customer’s requirements are met consistently.

• Ensuring that staff are trained and provided the resources to do the job right the first time. • Undertaking regular reviews and audits of all process • Total and organized commitment from management.

Quality Management Systems Comprises: • Training and ongoing support of all staffs • Providing quality services to the users of public health microbiology laboratory • Management of equipments and supplies • Management of finance and budgets • Quality assurance, QC, EQA, Accreditation, SOPS and Good Communication

• Quality issues concerning public health microbiology could be unique in : • It focuses more on environmental samples • It should also assess the ability of the laboratory to detect agents of disease during emergency. • Sampling issues is also critical, say during water sampling, air sampling, soil sampling

• Quality assurance pertaining public health microbiology includes: • Preanlytical components • Analytical components • Post analytical components

• Preanlytical issues of public health microbiology laboratory § It includes the following points but not only limited to: • • Proper laboratory setup ( organization) Adequate trained personnel An up to date equipments Proper specimen management

• Laboratory Organization ( set up). • As clinical microbiology laboratory, the organization of public health microbiology lab. May be demanding • There must be adequate space / rooms for each sections: o Specimen receipt o Specimen analysis o Section for bacteriology, Mycobcateriology, Virology, toxins detection ( serology). o Media and reagent preparation ( sterilization room) o Adequate and appropriate stores for reagents, control materials

• Laboratory benches should be suitable for the intended purpose Properly designed Easily cleaned Resist chemicals : acids, basses, and other chemicals Have good electrical system that can fit with the various instruments • Could have compartments for inoculation, gram staining ( sinks), microscopic examination, etc. • •

• Adequate trained personnel : • • • Public health microbiology Virology Mycology/ toxins Laboratory quality officers / managers Data managers etc

• Adequate equipments and consumables: • • Incubators Autoclaves Centrifuges Homogenizers Mixers Shakers Safety cabinet Weighing balances

• Adequate equipments and consumables cont…: - o Distillers for distilled water preparation o Deionizers : ion free water o Specimen containers different types • Flasks • • Bottles Jerikan o various glass wares • Test tubes • Measuring cylinder • Flasks

• Consumables § Media of different types • Solid: blood agar, TCBS agar, chromagar • Semisolid: SS, • Liquid : TCBS, Alkaline peptone water etc. § Antimicrobial discs for AST § Calipers for measurements § Membrane filters

§ Mac. Farandstandards § Antiseptics and disinfectants • 75 % alcohol • Phenol • Bleach solution • Savlon » Buffer solutions of different types • Tris buffer • Phosphate buffered saline solutions ( PBS)

• Specimen management issues • Environmental samples such as food, water, milk and beverages are more common. • SOPs is the key for quality • Specimen receipt guidelines should be available • All specimens must be adequate in volume • Proper labeling is critical • There must be specimen transportation protocol

• Analytical aspects of public health microbiology o Once the sample is properly collected it has to be analyzed according to the required quality o It includes the following issues : • Calibration of equipments like pipettes, dispensers, • Running of quality control samples • Negative and positive quality control samples are required for : • Staining solutions • Culture media • AST • Result interpretation and verification

• Calibration is indicated • Whenever new equipment is installed • During change in spare parts • As it is required by the SOP for a particular tests • This is true for PH meter for every PH measurement

• Quality control issues concerning culture media • Any culture media should be prepared aseptically • Sterility testing is an acceptable quality control measures for culture media: – 5 % of new culture media should be incubated at 37 o c for 48 hrs. – If there is contamination another 5 % of the batch is repeated. If there is no contamination that is ok – If contamination persist , new batch of media will be prepared.

• In addition measures has to be taken to avoid contamination during : • Cleanliness of room • Preparation of distilled and de-ionized water • Limited access to microbiology laboratory

§ Proper use of refrigerator, incubator, etc • Store one item in one refrigerator/ different compartment • Never store samples, culture media in the same refrigerator • Volume of items stored/ kept must be proportional with the intended purposes.

• Quality control measures related to staining solution • Staining solution must be free from contaminants • It has to be filtered • Known gram negative and gram positive control slides/ materials must be run for new batch of stain and according to SOP of each procedures. • It should be with in the expiry date

• Quality control issues concerning AST • Once microbes are isolated from samples drug resistance pattern can be done • Pure culture must be prepared • The AST methods must be standardized ( WHO recommendation) • Mc. Farland standard solution is very useful to prepare standard in oculum / suspensions.

• Standard organisms with known sensitivity pattern must be used : • ATCC ( American Type Culture Collection system) • A new reference culture should be purchased annually. • Working culture can be maintained at 4 degree centigrade for four weeks.

• Recommended antimicrobial discs must be used. • Antimicrobial discs should be stored at refrigerator temprature( 2 -8 degree centigrade). • It has to be used with in the expiration date. • Caliper for measurements for zone of inhibition is vital.

• Result interpretation and verification • Result must be interpreted according to SOP • Any result that is sent to requester must be verified by the laboratory manager / head responsible for that unit. • Screening tests should be confirmed

• External quality assessment issues • This is a system that allow participation of particular laboratory to compare it’s performance with others. • It should be given by an external laboratories ( national and international) • It can be achieved by : • Onsite supervision • Proficiency testing • Blind rechecking of the laboratory results

• Onsite supervision of public health laboratory • It gives chance to observe all laboratory stet up and activities • It is best but expensive. • Required by accreditation bodies

§ Proficiency testing • Unknown water / milk sample could be sent to laboratories • The laboratory can analyze for the requested tests • The performance is compared with PT providers o It does not assess the over all performance of the lab o give chances for improvement o it should be continious

• Blind rechecking • Samples analyzed for microbiological purpose will be repeated by the external bodies. • Results will be communicated. • Remedial actions will be taken by the participant labs. • It is demanding • Limited EQA systems are available for public health microbiology labs.

• Post-analytical microbiology issues in – It includes matters such as: • • Recording Documentation communication sample retention issues. public health

• Safety in laboratory public health microbiology • All necessary safety measures should be taken • Safety measures must be available related to infectious and non infectious substances • Waste management issues. • Hazard related to sample processing transportation must be assessed. and sample

• Safety and personal protective equipment must be available • Gloves, gowns , biohazard bags • Goggles, Masks in case of epidemics and routine activities • Shower must be there • First aid measures should be available • Good attitudes towards hand hygiene practice is essential

• Summary • A good public health microbiology service is highly dependent on a sound quality assurance system. • Quality assurance is the over all system that control the laboratory activities. • It can be broadly classified in to internal and external components.
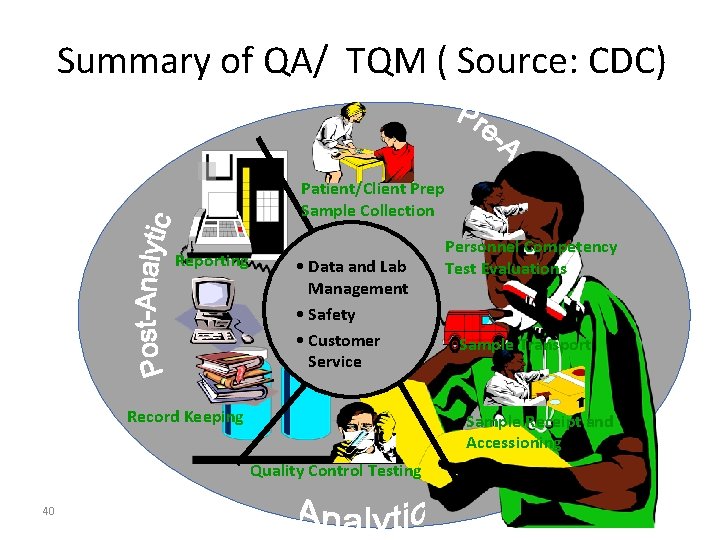
Summary of QA/ TQM ( Source: CDC) Patient/Client Prep Sample Collection Reporting • Data and Lab Management • Safety • Customer Service Record Keeping Sample Transport Sample Receipt and Accessioning Quality Control Testing 40 Personnel Competency Test Evaluations

• References • Jane Hudson , Principle of Clinical Laboratory Management. A study guide and Workbook. Printice Hall, Clinical Laboratory Science Series, upper Saddle River, New Jersy, 2004. • David Burnett, Understanding Accreditation In Laboratory Medicine. ACB Venture Publications. 1996. • Niel T. Constantine , Rebecca D. Saville , Elzabeth M. Dax. Retroviral Testing and Quality Assurance Essential for Laboratory Diagnosis. , 2005. , Med Mira Laboratories. • Guidance on regulations for the Transport of Infectious Substances , World Health Organization 2007. • Wytze P. Oosterhuis, David E. Bruns, Joseph Watine, Sverre Sandberg, and Andrea R. Horvath. 2004. Evidence-Based Guidelines in Laboratory Medicine: Principles and Methods. Clinical Chemistry 50: 5 , 806– 818. Special report. • Laboratory Biosaftey Guidelines, 3 rd , 2004, Minister of Health, Population and Public Health Branch Centre for Emergency Preparedness and Response, Canada
- Slides: 41