Chapter One Introduction Chapter One Introduction What is











- Slides: 18

Chapter One Introduction

Chapter One / Introduction • What is Chemistry? • Chemistry is The study of Matter and changes it undergoes. • Matter is anything that occupies space and has mass • Thus Matter is everything around us.

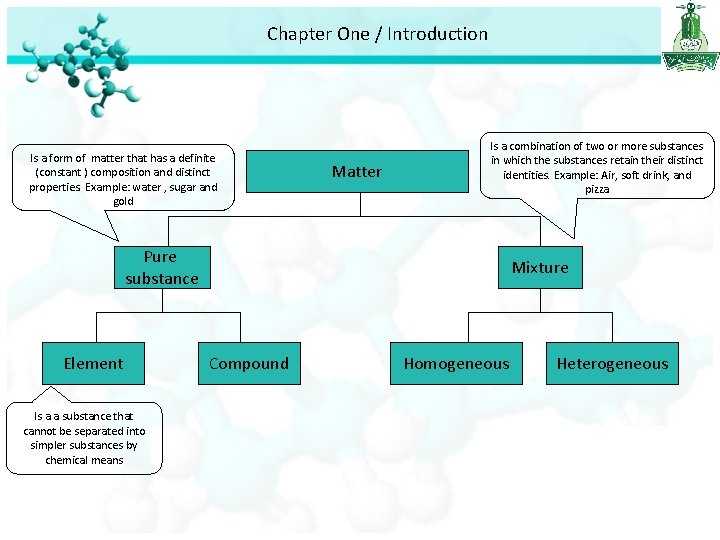
Chapter One / Introduction Is a form of matter that has a definite (constant ) composition and distinct properties. Example: water , sugar and gold Matter Is a combination of two or more substances in which the substances retain their distinct identities. Example: Air, soft drink, and pizza Pure substance Element Is a a substance that cannot be separated into simpler substances by chemical means Mixture Compound Homogeneous Heterogeneous

Chapter One / Introduction

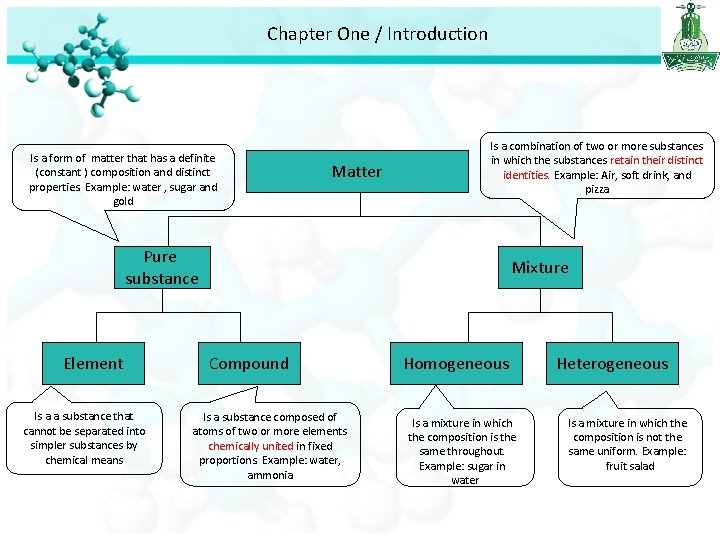
Chapter One / Introduction Is a form of matter that has a definite (constant ) composition and distinct properties. Example: water , sugar and gold Matter Is a combination of two or more substances in which the substances retain their distinct identities. Example: Air, soft drink, and pizza Pure substance Element Is a a substance that cannot be separated into simpler substances by chemical means Mixture Compound Is a substance composed of atoms of two or more elements chemically united in fixed proportions. Example: water, ammonia Homogeneous Is a mixture in which the composition is the same throughout. Example: sugar in water Heterogeneous Is a mixture in which the composition is not the same uniform. Example: fruit salad

Chapter One / Introduction Element compound Heterogeneous mixture magnet Homogeneous mixture

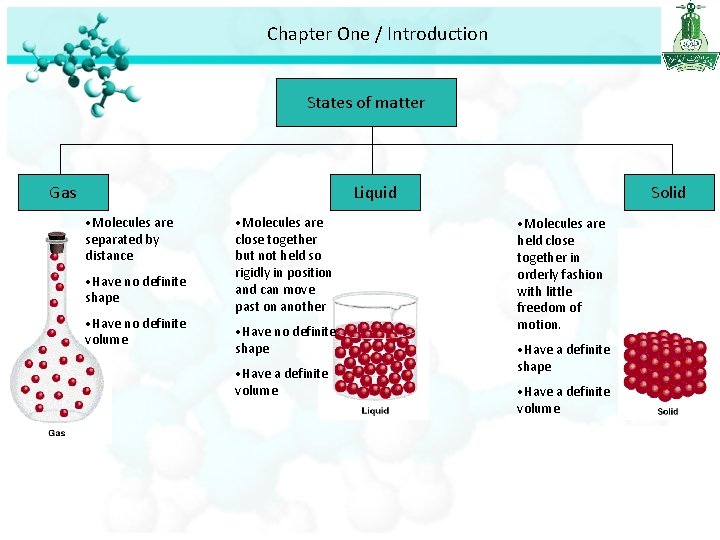
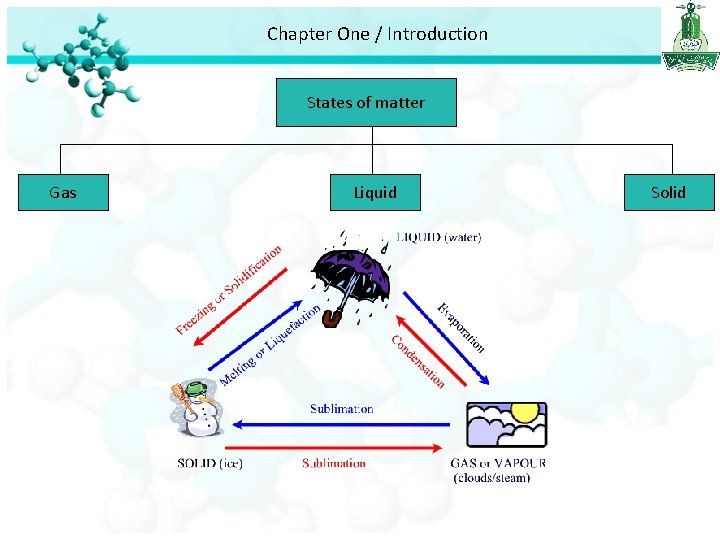
Chapter One / Introduction States of matter Gas Liquid • Molecules are separated by distance • Have no definite shape • Have no definite volume • Molecules are close together but not held so rigidly in position and can move past on another • Have no definite shape • Have a definite volume Solid • Molecules are held close together in orderly fashion with little freedom of motion. • Have a definite shape • Have a definite volume

Chapter One / Introduction States of matter Gas Liquid Solid

Chapter One / Introduction Measurement in chemistry • • In every measurement there is a number followed by a unit. Units are essential to stating measurements correctly. The international system of units (SI) is used world wide to reports result. There are seven SI base units as following: Base Quantity Name of unit Symbol Length meter m Mass kilogram kg Time second s Electrical current ampere A Temperature kelvin K Amount of substance mol Luminous intensity candela cd

Chapter One / Introduction Measurement in chemistry • • • Derived units are unit made up of combination of SI base units such as: Properties unit symbol Definition force Newton N Kg m/s 2 Pressure Pascal pa N/m 2 or Kg /m s 2 Energy Joule J Kg/m 2 s 2 or N m SI units are modified in decimal fashion by a series of prefixes. A prefix may be added to a unit to produce a multiple of the original unit.

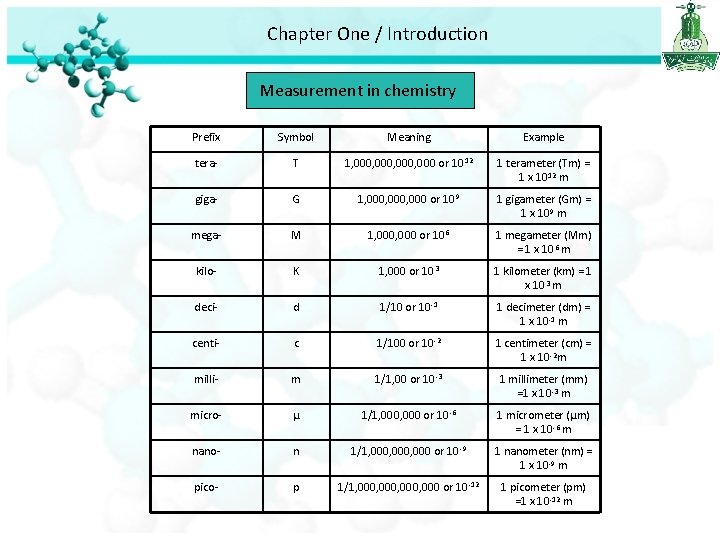
Chapter One / Introduction Measurement in chemistry Prefix Symbol Meaning Example tera- T 1, 000, 000 or 10 12 1 terameter (Tm) = 1 x 1012 m giga- G 1, 000, 000 or 10 9 1 gigameter (Gm) = 1 x 109 m mega- M 1, 000 or 10 6 1 megameter (Mm) = 1 x 106 m kilo- K 1, 000 or 10 3 1 kilometer (km) = 1 x 103 m deci- d 1/10 or 10 -1 1 decimeter (dm) = 1 x 10 -1 m centi- c 1/100 or 10 -2 1 centimeter (cm) = 1 x 10 -2 m milli- m 1/1, 00 or 10 -3 1 millimeter (mm) =1 x 10 -3 m micro- µ 1/1, 000 or 10 -6 1 micrometer (µm) = 1 x 10 -6 m nano- n 1/1, 000, 000 or 10 -9 1 nanometer (nm) = 1 x 10 -9 m pico- p 1/1, 000, 000 or 10 -12 1 picometer (pm) =1 x 10 -12 m

Chapter One / Introduction Measurement in chemistry Unit conversion: 1 - prefix to Base unit eg. Km =====> m 2 - Base unit to prefix eg. m =====> km 3 - prefix to prefix eg. km =====>nm

Chapter One / Introduction Measurement in chemistry 1 - prefix to Base unit To remove a prefix from a value, insert the numerical value of the prefix in place of the symbol. Example : convert 8. 53 pm to meters? Replace p with x 10 -12, therefore the answer is 8. 53 x 10 -12 m • 2 - Base unit to prefix To insert a prefix into a value, insert both the prefix and the inverse of its numerical number. Example: convert 0. 000462 g to milligrams ? 0. 000462 x 103 mg = 0. 462 mg • 3 - prefix to prefix Example : convert 6 km to nm? First convert km to m Replace k with x 103, therefore the answer is 6 x 103 m Then convert m to nm 6 x 103 x 10+9 = 6 x 1012 nm

Chapter One / Introduction Measurement in chemistry How many second are in a microseconds ? (a) 1 x 10 -1 (b) 1 x 10 -6 (c) 1 x 10 -15 (d) 1 x 10 6 Which of the following is the smallest distance? (a) 21 m Put all of them in the same unit (b) 2. 1 x 102 cm (c) 21 mm (d) 2. 1 x 104 pm

Chapter One / Introduction Measurement in chemistry The diameter of an atom is approximately 1 10 -7 mm. What is this diameter when expressed in nanometers? (a) 1 10 -18 nm (b) 1 10 -15 nm (c) 1 10 -9 nm (d) 1 10 -1 nm Which of these quantities represents the largest mass? (a) 2. 0 102 mg Put all of them in the same unit (b) 0. 0010 kg (c) 1. 0 105 g (d) 2. 0 102 cg

Chapter One / Introduction Measurement in chemistry There is also a set of non-SI units accepted for use with SI such as litter. One common unite of volume is litter (L). A litter is the volume occupied by one cubic decimeter. 1 dm = 10 cm 1 dm 3 = 1000 cm 3 1 dm 3= 1 L 1000 cm 3 = 1 L 1 cm 3 = 1 m. L • • 1 L=1000 ml 1 m. L = 10 -3 L 1 m. L=1 cm 3 1 L=1 dm 3

Chapter One / Introduction Density • Density = mass / volume d = m/V Mass (g) Density (g/ml) Volume (ml) • Example: A student determines that a piece of an unknown material has a mass of 5. 854 g and a volume of 7. 57 cm 3. What is the density of the material? d = m/V = 5. 854 (g) / 7. 57 (cm 3) = 0. 773 g/cm 3 = 0. 773 g/m. L • Example A piece of silver (Ag) metal weighing 194. 3 g is placed in a graduated cylinder containing 242. 0 m. L of water. The volume of water now reads 260. 5 m. L. From these data calculate the density of silver? volume of silver = volume of water (after) – volume of water ( before ) = 260. 5 – 242. 0 = 18. 5 ml d = m/V = 194. 3 / 18. 5 = 10. 5 g/ml
