CHAPTER IV BACTERIAL STAINING ACKNOWLEDGMENT ADDIS ABABA UNIVERSITY

CHAPTER IV BACTERIAL STAINING

ACKNOWLEDGMENT • • ADDIS ABABA UNIVERSITY JIMMA UNIVERSITY HAWASSA UNIVERSITY HARAMAYA UNIVERSITY OF GONDAR AMERICAN SOCIETY OF CLINICAL PATHOLOGY CDC- Ethiopia

Learning objectives At the end of this chapter, the student will be able to: 1. Describe smear preparation for bacterial examination 2. Define stains and bacterial staining. 3. Describe the uses of staining solutions. 4. List the types of staining methods that are used to stain bacteria. 5. List the different types of bacterial staining techniques. 6. Discuss the principles of Gram’s stain and Ziehl Nelseen stain. 7. Describe the factors that contribute for false Gram’s stain and Zihel -Nelseen stain results. 8. Prepare the Gram’s stain and Zihel -Nelseen stain reagents.

PREPARATION OF SMEARS FOR BACTERIOLOGICAL DIAGNOSIS Ø Why we need to make smears? • Making smear is a precondition to facilitate staining and further observation of microorganisms under microscope. • Without making appropriate smear, which is thin enough to make a single layer of bacteria, it is difficult to observe and read different staining reactions of bacteria. .

Materials for bacteriological Smear preparation • Microscope slide • Swab, for making smear taking sample from body parts or culture media • Applicator stick- for taking sample from & make smear on slide • Lead pencil – (not grease pencil or ink) – for putting marks on slides for identification • Heat, alcohol or other chemical – for fixing the smear on the slide to prevent from being washed during staining procedure and for killing the bacteria on the slide.

Preparing smear • If smears are to provide reliable information they must be prepared, labeled and fixed correctly prior to being stained. Labeling Slides • Every slide must be labeled clearly with the date and the patients name and number. When ever possible, smears should be spread on slides which have one end frosted for labeling – A lead pencil should be used for writing on the frosted area. Because pencil marks unlike grease pencil marks, will not be washed off during the staining processes.

How to make smears • Smears should be spread evenly covering an area of about 15 – 20 mm diameter on a slide – NB. Careless handling of pathological material and cultures may result in the contamination of the out side of vessels, the laboratory bench, or floor. Microorganisms can then be transferred from these surfaces to the finger and hands, enter blood stream through cuts and scratches, or be transferred to the mouth and eyes. so when ever smears are made every precaution and aseptic technique should be followed strictly!

Ø Some of the techniques used to make smears from different specimens are as follows. • Purulent specimen – (Purulent – samples which contain pus /dead WBC) using a sterile wire loop, make a thin preparation. Do not centrifuge a purulent fluid, E. g. C. S. F containing pus cells • Non purulent fluid specimens - centrifuge the fluid and make a smear from a drop of the well mixed sediment. • Culture – Emulsify a colony in sterile distilled water and make a thin preparation on a slide. When a broth culture, transfer a loop full to a slide and make a thin preparation. • Sputum- Use a piece of clean stick to transfer and spread purulent and caseous material on a slide. Soak the stick in a phenol or hypochlorite disinfectant before discarding it. • Swabs- (are stick with a cotton roll at one tip)- Roll the swab on a slide. This is particularly important when looking for intracellular bacteria such as N. gonorrhoea (urethral, cervical or eye swab). Rolling the swab avoids damaging the pus cells. • Feaces (stool) – Use a piece of clean stick to transfer pus and mucus to a slide (decontaminate the stick before discarding it) spread to make a thin preparation.

The thickness of the smear should allow to read a text when placed under the smear.

Method of collecting and making skin smear of M. Leprea • A smear for the examination of M. leprae must be collected using on asceptic and safe technique. The site sampled should be the edge of a leprosy lesion. 1. Explain the procedure to the patient 2. Fit anew scalpel blade (surgical blade) in its scalpel holder. Sterilize the blade by wiping it carefully with a piece of absorbent cotton wool soaked in 70% v/v ethanol and flaming it for 2 -3 seconds in the flame of a sprit lamp. Allow the blade to cool.

3. Wearing protective rubber gloves, cleanse the area from where the smear is to be taken, using a cotton wool swab, moistened with 70% v/v ethanol. Allow the area to dry. 4. Pinch the skin tightly between the thumb and index finger until it becomes pale due to loss of blood. NB. The area should be kept bloodless. 5. Using the sterile blade, make a small cut through the skin surface, about 5 mm long and deep enough in to the dermis (2 -3 mm) where the bacteria will be found, continue to hold the skin tightly.

6. Turn the scalpel blade until it is at a right angle to the cut, using the blunt edge of the blade, scrape firmly two or three times along the edges and bottom of the cut to collect a sample of tissues juice and cell (not blood) 7. Transfer the sample to a slide. Make a small circular smear, covering evenly an area measuring 5 – 7 mm in diameter. 8. Cover the cut with small dressing.

Drying and fixing smears • After making a smear, leave the slide in a safe place for the smear to air – dry protected from direct sun light. • When a smear requires urgent staining, it can be dried quickly using gentle heat. • The purpose of fixation is to preserve microorganisms and to prevent smear being washed form slides during staining. Smears are fixed by heat, alcohol, or occasionally by other chemicals.

Heat Fixation - This is widely used but can damage organisms and alter their staining reactions especially when excessive heat is used. - Heat fixation also damages leukocytes and is therefore unsuitable for fixing smears which may contain intracellular organisms such as N. gonorrhoeae and N. meningitides. • When used, heat fixation must be carried out with care.

The following techniques are recommended: 1. Allow the smear to air – dry completely 2. Rapidly pass the slide, smear upper most, three times through the flame of a sprit lamp or pilot flame of a Bunsen burner. NB: do not heat smears excessively 3. Allow the smear to cool before staining.

Alcohol Fixation • This form of fixation is far less damaging to microorganisms than heat. Cells, especially pus cells, are also well preserved. - Alcohol fixation is therefore recommended for fixing smears when looking for Gram negative intracellular diplococcic. - Alcohol fixation is more bactericidal than heat. (E. g. M. tuberculosis is rapidly killed in sputum smears after applying 70% v/v alcohol.

The following technique is recommended. • Allow the smear to air dry completely • Depending on the type of smear, alcohol fix as follows. - For the detection of intracellular Gram negative diplococcic (N. gonorrhea or N. meningitides), fix with one or two drops of absolute methanol or ethanol. • Leave the alcohol on the smear for a minimum of 2 minutes or until the alcohol evaporates.

Other Chemical Fixation • Other chemicals are some times necessary to fix smears, which contain particularly dangerous organisms, to ensure all the organisms are killed. - 40 g/l potassium permanganate is recommended for fixing smears, which may contain Anthrax bacilli. - Formaldehyde vapor is sometimes recommended for fixing smears which may contain Mycobacterium species. Formaldehyde fixed smears, however, tend to stain poorly and the chemical it self is toxic with an injurious vapor.

Precautions to take when staining smears • Use a staining rack. Do not immerse slides in containers of stain because this can lead to contamination of stain and transfer of organisms from one smear to another. • Do not attempt to stain a smear that is too thick. This is one of the commonest causes of poor staining and incorrect reporting of smears. • When washing smears of C. S. F sediment and specimens which can be easily washed from a slide, direct the water from a wash bottle on the back of the slide, not directly on the smear. • After staining, place the slides at an angle in a draining rack for the smears to air dry. Do not blot smears to dry with filter or blotting paper.

PRINCIPLE OF STAINING IN BACTERIOLOGY • Even with the microscope, bacteria are difficult to see unless they are treated in a way that increases contrast between the organisms and their background. The most common method to increase contrast is to stain part or all of the microbe.

Definition Ø Stains (Dyes) are coloured chemical compounds that are used to selectively give (impart) colour to the colourless structures of bacteria or other cells. Ø Bacterial staining is the process of imparting colour to the colourless structures (cell wall, spore, etc) of the bacteria in order to make it visible under the microscope.

Uses of staining 1. To observe the morphology, size and arrangement of bacteria 2. To differentiate one group of bacteria from the other group. Ø Staining reactions are made possible because of the Physical phenomena of capillary osmosis, solubility, adsorption, and absorption of stains or dyes by cells of micro-organisms.

§ Basic principle: The cellular components of mammalian as well as microbial cell are different. For example the nuclei of cell is negatively charged because of the presence of acidic component (DNA) hence it combines with positively charged compounds, (basic dyes). and the cytoplasm parts of a cell is generally positively charged therefore combines with negatively charged compounds (acidic dyes).

Why are stains not taken up by every micro-organism? Factors controlling selectivity of microbial cells are: 1. number and affinity of binding sites 2. rate of reagent uptake 3. rate of reaction 4. rate of reagent loss (differentiation or regressive staining)

Type of stain in Microbiology There are three kinds of stains 1. Basic stains 2. Acidic stains 3. Neutral stains NB: This classification is not based on the p. H of stains

A. Basic Stains § Are stains in which the colouring substance is contained in the base part of the stain and the acidic part is colourless. § The bacterial cells can easily stain with basic dyes e. g. Methylene blue stain, Safranin, Genetian violet, Carbolfuchsin etc

B. Acidic Stains Ø Are stains in which the colouring substance is contained in the acidic part of the stain and the base part is colourless. Ø Acidic dyes are mainly used in histology laboratory. It is not commonly used in microbiology laboratory ; e. g. eosin.

C. Neutral Stains § Are stains in which the acidic and basic components of stains are coloured. § Neutral dyes stain both nucleic acid and cytoplasm. these stains are commonly used in hematology laboratory. e. g Giemsa’s stain, Wright’s stain.

Type of staining methods 1. Simple staining method 2. Differential staining method 3. Special staining method

1. Simple staining method • It is a type of staining method in which only a single dye is used. • There are two kinds of simple staining methods A. Positive staining B. Negative staining A. Positive staining: The bacteria or its parts are stained by the dye. e. g. Methylene blue stain, Crystal violent stain

Procedure 1. Make a smear and label it 2. Allow the smear to dry in air 3. Fix the smear over a flame 4. Apply a few drops of positive simple stain 5. Wash off the stain with water 6. Air -dry and examine under microscope Result-all the bacterial surface is stained.

B. Negative staining. The dye stains the background and the bacteria component remain unstained. e. g. Indian ink stain. 2. Differential staining method A method in which multiple stains (dye) are used to distinguish different group of bacteria. e. g. Gram’s stain, Ziehl-Neelson stain.

GRAM’S STAIN This method was developed by the Danish bacteriologist Christian Gram in 1984. Basic concepts: • Most bacteria are differentiated by their gram reaction due to differences in their cell wall structure. • The surface of bacterial cell has got a negative charge due to the presence of polysaccharides and lipids (PG) this has made the surface of the bacteria to have affinity to cationic or basic dyes (when the colouring part is contained in the basic part. )

Principle Ø The principle of Gram’s stain is that cells are first fixed to slide by heat or alcohol and stained with a basic dye (e. g. crystal violate), which is taken up in similar amounts by all bacteria. ØThe slides are then treated with an Gram’s iodine (iodine KI mixture) to fix (mordant) the crystal violet stain on Gram positive bacteria, decolorized with acetone or alcohol, and finally counter stained with Safranin.

Ø Gram positive bacteria: - stain dark purple with crystal violet and are not decolorized by acetone or ethanol. The following are some important examples of gram positive bacteria. • • • Staphylococcus, Streptococcus, Clostridium Bacillus Corynebacterium … etc – N. B. The reason for the retention of the primary stain (CV) by the gram positive bacteria after decolorization is due to the presence of more acidic protoplasm (PG layer) of these organisms which bind to the basic dye.

Ø Gram negative bacteria: - stain red because after being stained with crystal violet they are decolorized by acetone or ethanol and take up red counter stain. (Neutral red, Safranin or dilute carbol fuchsin). • The following are some important gram negative bacteria: • Nesseria spp. • Haemophilus spp. • Salmonella, shigella, vibrio, Klebsilla, Coliforms …etc.

Required reagents • Crystal violet • Gram’s Iodine • Acetone-Alcohol or 95% Alcohol • Safranin or Neutral red

Gram’s stain reagents preparation 1. Crystal violet stain • Crystal violet ……………. 20 g • Ammonium oxalate ………… 9 g • Ethanol or methanol absolute ………. . 95 ml • Distilled water ……………. . to 1 litre

3. Acetone -alcohol To make 1 litre • Acetone ……………… 500 ml • Ethanol or methanol absolute …. . 475 ml • Distilled water ………………. 25 ml N. B: some workers prefer to use acetone by itself or ethanol 95% v/v as decolorizing solution. A mixture of acetone and alcohol is recommended because it decolorizes more rapidly than ethanol 95% v/v and is less likely to over decolorize smears than acetone with out alcohol added.
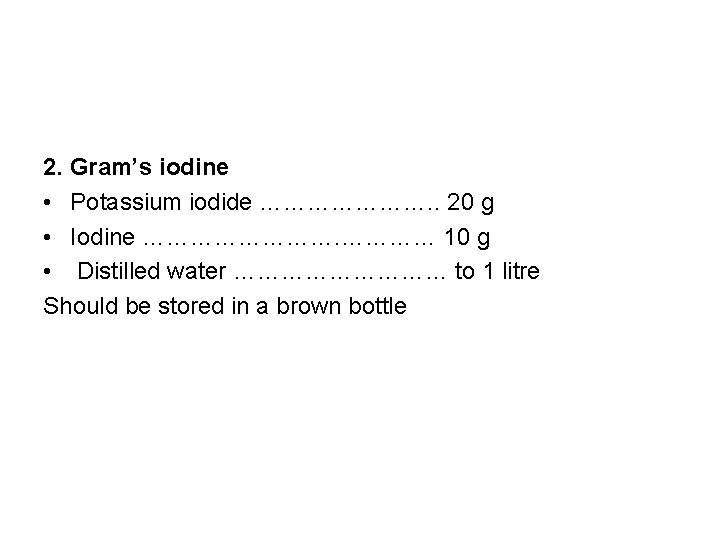
2. Gram’s iodine • Potassium iodide …………………. . 20 g • Iodine ………… 10 g • Distilled water …………… to 1 litre Should be stored in a brown bottle

4. Safranin 1. Prepare a stock solution § Safranine O ------------------------ 2. 5 g § Ethanol (95%) ----------------------100 ml Mix until all the safranine is dissolved. Transfer the solution to a glass – stoppered bottle. Label the bottle ( Safranine stock solution) and write the date. 2. Prepare a working solution in a glass stoppered bottle § Stock solution -----------------------10 ml § Distilled water -----------------------90 ml Label the bottle ( Safranine working solution) and write the date

5. Neutral red; 1 g/l (w/v) To make 1 litre • Neutral red ……………. 1 g • Distilled water ………… 1 litre N. B: Neutral red is selected as the counter stain because it stains well gonococci and meningococci. Safranin can also be used. The dilute carbolfuchsin (1 in 10) is recommended for staining Vincent’s organisms. Yersinia, Haemaophilus, campylobacter, and vibrio species.

Procedure of Gram’s Stain 1. Fix the dried smear with heat by gently passing it over sprit lamp or Bunsen burner. – Note: When the smear is for the detection of gonococci or meningococci, it should be fixed with methanol for 2 minutes 2. Cover the fixed smear with crystal violet stain for 30 – 60 seconds 3. Rapidly wash off the stain with clean water 4. Tip of all the water, and cover the smear with Lugol’s iodine for 30 – 60 seconds 5. Wash off the iodine with clean water 6. Decolorize rapidly with acetone-alcohol for 30 seconds. wash immediately with clean water.

7. Cover the smear with Neutral red or Safranin for 2 minutes 8. Wash off the stain with clean water. 9. Wipe the back of the slide clean, and place it in a draining rack for the smear to air dry 10. Examine the smear microscopically, first with the 40 x objective to check the staining and to see the distribution of material, and then with oil immersion objective to report the bacteria and cells.

Results • Gram positive bacteria …. . …………………. . Purple(blue) • Yeast cells …………………. Dark purple • Gram negative bacteria ……. ………………. . Pale to red • Nuclei of pus cell ……. …………… Red • Epithelia cells …………………. Pale red
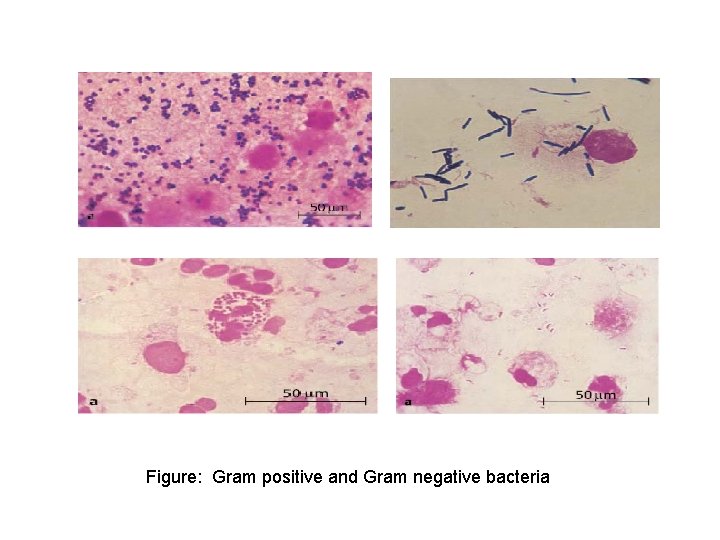
Figure: Gram positive and Gram negative bacteria

Reporting of Gram’s stained smear The report should include the following: 1. Number of bacteria present whether many, moderate, few or scanty 2. Gram reaction of the bacteria whether Gram positive or Gram negative 3. Morphology and arrangement of the bacteria whether cocci, diplococci, streptococci, rods, or coccobacili; also whether the organisms are intracellular.

4. Presence and number of pus cells. 5. Presence of yeast cells and epithelia cells. Example of a gram stain teport A Gram’s stain of urethral smear report might read as: – “Moderate number of gram negative intercellular diplococci and many pus cells “

Factors which contribute to false Gram’s staining result I. False Gram positive reaction • Preparation of too tick smear – the stain will not be fully washed. • Application of crystal violet - presence of sediment in crystal violet - this can be avoided by filtering the stain before use. • Decolourizing time - when insufficient decolourization time is used or ( when not fully decolorized).

II. False Gram Negative reactions • Making smear from old culture. • Cell wall damage due to antibiotic. • Excessive heat fixation of smear. • Over decolourization of smear (decolourization for longer time). • Use of an iodine solution which is too old (i. e. yellow in stead of brown in colour) (its mordant effect will be decreased). Solution for the above problem • Always check new batches of stain and reagent for correct staining reaction using a smear containing known gram positive and gram negative organism as a control.

Ziehl-Neelson (Acid fast Bacilli-AFB) staining method • Developed by Paul Ehrlich in 1882, and modified by Ziehl and Neelson • Ziehl-Neelson stain is used for staining mycobacteria which are hardly stained by Gram‘s staining method. • Once the Mycobacteria is stained with primary stain it can not be decolorized with acid, so named as acid-fast bacteria.

- Mycobacteria typically are slightly bent or curved slender rods. About 2 um- 4 um long and 0. 2 um – 0. 5 um wide. - The most striking chemical feature of mycobacteria is their extra ordinary high lipid content in the cell wall (up to 60% of its dry weight). This high lipid content probably accounts for some of the other unusual properties of mycobacteria. E. g Relative impermeability to stains, acid fastness, unusual resistance to killing by acid and alkali. • The cell wall of Mycobacteria also contains a peptidoglycan layer, glycolipids, protein and Mycolic acid(This is unique to mycobacteria, nocardiae and corynebacteria).

Photogenic Mycobacteria 1. Tubercle bacilli - M. tuberculosis (human tubercle type) - M. bovis (bovine type) 2. Leprosy bacilli - M. lepraemurium 3. Environmental mycobacteria (atypical, anonymous) mycobacteria - M. avium- intercellulare - M. xenopi etc

M. tuberculosis • Most infections with M. tuberculosis are caused by inhaling cough droplets or dust particles containing tubercle bacilli which become lodged in the lung forming a small inflammatory lesion and cause Pulmonary tuberculosis. • Infected droplets remain air born for considerable time, and may be inhaled by susceptible persons • Tuberculosis is a disease of global importance. One third of the world’s population is estimated to have been infected with M. tuberculosis and eight million new cases of tuberculosis arise each year.

M. bovis • Is found mainly as a pathogen in a cattle and occasionally in other animals. • Humans become infected by close contact with infected cattle or by ingesting the organisms in raw un treated milk. • Person to person transmission of bovine strains may also occur.

Other forms of tuberculosis • Tuberculous meningitis – when tubercle bacilli reach the meninges through blood and affect the meninge. • Miliary tuberculosis: - Wide spread miliary infection (liver, spleen & lymph glands) • Renal and urogental tuberculosis • Bone and joint tuberculosis

M. Leprae • causes leprosy, a chronic infectious disease that affects the skin, peripheral nerves, mucosa of the upper respiratory tact and the eye. • It is mainly transmitted via the respiratory tract or skin and has a long period of incubation(2 – 5 years) or latency Environmental Mycobacteria – • are the most frequent causes of pulmonary infections resembling tuberculosis, and disseminated disease and also cause lymphadenitis (mainly in children). • Such species are acid fast but differ from the M. tuberculosis complex by being opportunistic pathogens, with limited distribution and acquired from the environment. E. g Soil or water.

Principle of Ziehl-Neelson (Acid fast) staining method § Sputum smear is heat –fixed, flooded with a solution of carbilfusin (a mixture of basic fuschin and phenol) and heated until steam rises. The heating which facilitate penetration (entrance) of the primary stain into the bacterium. § After washing with water, the slide is covered with 3% HCl (decolourizer). Then washed with water and flooded with methylene blue ( Mycobacterium tuberculosis) and malachite green (Mycobacterium leprae).

Materials for AFB staining • Sputum container (for M. tuberculosis) – for sample collection • Wire loop or applicator stick – to spread sputum on the microscope slide • Microscope slide – for making smears • Marking pen – to put identification number on the microscope slide • Forceps- to hold smeared slide • Bunsen burner or sprit lamp – to fix the smeared slide and to flame the smear during staining. • Staining racks (staining rods) – for staining. • Slide rack – to place stained slide to dry in the air • Ziehel – Neelson (AFB) stain - Carbolfuchsin - 3% Acid alcohol - Methylene blue (malachite green)

Ziehl Neelson stain reagents preparation A. Carbolfuschin - Solution A (saturated solution of basic fuchsin) Basic fuchsin …………. . 3 gm Ehanol …………………. . 100 ml - Solution B ( phenol aquous solution, 50 g/L ( 5%) Phenol …………. . . 10 gm Distilled water ……………… 200 ml - Mix 10 ml of solution A with 90 ml of solutin B. - Transfer resulting mixture to a glass stopperd amber bottle and label.

B. 3% Acid –alcohol (decolorizer) Hydrocloric acid ( conc) …………… 3 ml Ehanol ………………. . 97 ml Label the bottle. N. B It is recommended to use 25 % H 2 SO 4 as decolorizing solution • Content per liter: – sulfuric acid (minimum 95%) 250 ml – water 750 ml

Preparation: 1. Measure 750 ml of water into a 2 -liter flask 2. Measure 250 m. L of concentrated sulphuric acid in a cylinder 3. Pour it slowly into the flask containing the water, directing the flow of acid gently along the inner side of the flask 4. Stop and swirl flask regularly as a lot of heat is generated until all acid is added 5. Mix well and allow to cool before use

C. Methylene Blue Stain • Methlene blue chloride. . . . 0. 5 g • Distilled water. . . . . 100 ml D. Malachite green …………………. 0. 5 gm Distilled water …………. 100 ml - Using a pestle and mortar, grind the malachite green crystal to a powder. Dissolve the grind powder in 100 ml distilled water and store in a dark brown bottle.

Hot and cold Ziehel Nelson technique • In the hot Zn technique the phenolic – carbol fuchsin stain is heated to enable the dye to penetrate the waxy mycobacterial cell wall. • Techniques that do no heat the stain are referred to as ‘cold’ techniques. In these, a penetration of the stain is usually achieved by increasing the concentration of basicfuchsin and phenol. • Comparison between the ‘hot’ and ‘cold’ method has shown that both M. leprae and M. tuberculosis stain less well by the cold method.

Difference between Acid fastness of Mycobacterium species • M. tuberculosis and M. ulcerance are strongly acid fast, when staining specimens for these species, a 3% v/v acid solution or 25%H 2 SO 4 is used to decolorize the smear. • M. lepreae is only weakly acid fast. A 1% v/v acid or 5% H 2 SO 4 decolorizing solution is therefore used for M. lepreae smears and also different staining and decolorizing times.

Procedure 1. Use blood – specked, purulent and caseous sputum for smear preparation (do not use saliva) 2. Heat fix the dried smear, Alcohol fixation: This is recommended when the smear has not been prepared form sodium Hypochlorite (bleach) - Heat fixation of untreated sputum will not kill M. tuberculosis where as alcohol – fixation is bactericidal. * Since M. tuberculosis can infect almost any organ in the body, the laboratory could receive a variety of extra – pulmonary specimen E. g. body fluid, tissue, pus, CSF, urine etc. NB: The benefit of microscopy on the specimens is limited and it is recommended that extra pulmonary specimens be referred for culture. However if there is no option the fluid (non purulent) should be centrifuged and smears prepared form the sediment.

3. Cover the smear with carbolfuchsin stain 4. Heat the stain until vapor just begins to rise. Do not over heat – Allow the heated stain to remain on the slide for 5 minutes. 5. Wash off the stain with clean water 6. Cover the smear with 3% acid alcohol for 5 minutes or until the smear is sufficiently decolorized. i. e. pale pink. 7. Wash well with clean water 8. Cover the smear with methylene blue for 1 – 2 minutes. 9. Wash off the stain with clean water.

10. Wipe the back of the slide clean, and place it in a draining rack for the smear to air dry (do not blot dry) 11. Examine the smear microscopically, using the 100 x oil immersion object. 12. Examine a minimum of 300 fields systematically before the smear is reported as negative.
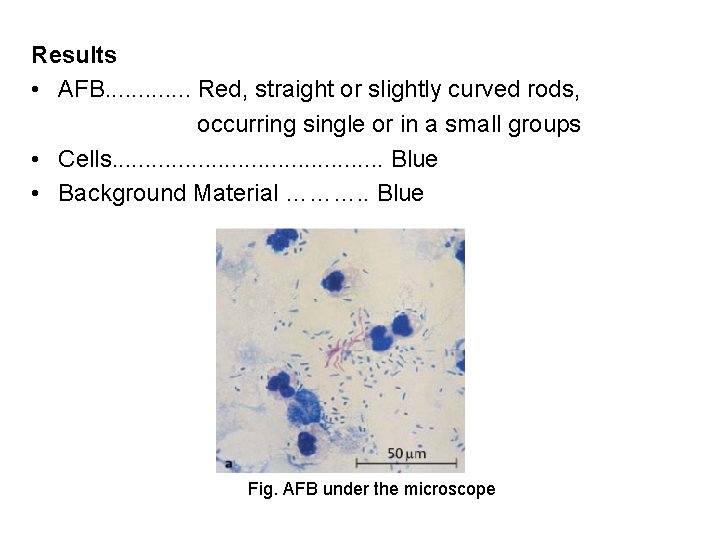
Results • AFB. . . Red, straight or slightly curved rods, occurring single or in a small groups • Cells. . . . . Blue • Background Material ………. . Blue Fig. AFB under the microscope

REPORTING OF SPUTUM SMEAR • When any definite red bacilli are seen report the smear as AFB positive and give an indication of the number of bacteria present as follows: When 1 - 9 AFB /100 fields -------- report the exact number 10 – 100 AFB/100 fields --------- report + 1 – 10 AFB/field -------------- report ++ More than 10 AFB/field --------- report +++ (source : Manual basic techniques for a health lab WHO, 2003) N. B: AFB means number of acid fast bacilli seen. • • when very few AFB are seen E. g When only one or two AFB are seen, request further specimen for examination. When no AFB are seen after examining 300 fields report the smear as ‘ No AFB seen’ do not report as “Negative” because organisms may be present but not seen in those field examined.

Sensitivity of direct microscopy • Studies have shown that between 5000 to 10, 000 tubercle bacilli per millilitre of sputum are required for direct microscopy to be positive. So sufficiently large number of acid fast bacilli should be present to be detected by direct microscopy • The sensitivity can further be improved by examination of more than one smear from a patient. So up to three specimens (spot– morning- spot) are required and this can significantly increase the sensitivity. • Other way of increasing sensitivity is sodium hypochlorite (bleach) centrifugation technique to concentrate AFB. NB: Studies have shown that centrifugation of bleach treated sputum increased the sensitivity of direct microscopy from 43. 4% to 76. 3%, increasing the number of smear-positive patients detected. Sensitivity is significantly increased in HIV/AIDS patients.

Sodium hypochlorite centrifugation technique to concentrate AFB 1. Transfer 1 -2 ml of sputum to a screw - cap container of 15 m-20 ml capacity 2. Add an equal volume of concentrated sodium hypochlorite (bleach) solution and mix well. 3. Leave at room temperature for 10 -15 minutes, shake at intervals to break down the mucus in the sputum.

4. Add about 8 ml of distilled water, and mix well. 5. Centrifuge at 3000 g for 15 minutes. N. B: When centrifugation is not possible, leave the Na. OCl treated sputum to sediment overnight. 6. Using a glass Pasteur pipette, remove and discard the supernatant fluid. Mix the sediment. 7. Transfer a drop of the well mixed sediment to a clean scratch free glass slide. Spread the sediment to make a thin preparation and allow to air dry. 8. Heat fix the smear and stain it using Ziehl Neelson technique and examine microscopically.

Factors which bring false positive results • Utilization of slides that have been positive. These should be discarded. • Using scratched slides on which deposits of stain may look like bacilli • Using unfiltered fuchsin which may contain crystals. • Carelessness in heating the fuchsine, allowing it to dry and crystallize on the smear • Inadequate decolorizing of the smear.

Fluorescence staining for Mycobacteria • Fluorescence microscopy uses illumination from either a quarth halogen lamp or a high pressure mercury vapor lamp. • The advantage of fluorescence microscopy is that a low magnification is used to scan smears, allowing a much larger of the smear to be seen and resulting in more rapid examination.

Reagents • Auramine O Auramine-------------------------0. 1 gm 95% Ethanol-----------------------10 ml Dissolve Auramin with Ethanol -----------Solution 1 • Phenol crystal---------------------3. 0 gm Distilled water---------------------87 ml Dissolve phenol with Distiied water--------Solution 2 Mix solution 1 and 2 and store in amber bottle away from light and heat.

Decolorizing solution 3% acid alcohol Counter stain Either Potassium permanganate or acridine orange can be used • Potassium permanganate (KMn. O 4) ---------0. 5 g Dis. Water-------------------------100 ml Dissolve and store in amber bottle • Acridine orange----------------------0. 01 g Anhydrous dibasic sodium phosphate (NA 2 HPO 4) -0. 01 g Distilled water-------------------------100 ml Dissolve sodium phosphate in distilled water. Add Acridine orange. Store in amber bottle.

Procedure 1. Place the dried labeled smear on staining rack 2. Flood entire smear with Auramine O and allow to stain for 15 min ensuring that staining solution remains on smear 3. Rinse with distilled water and drain. Tap water contains chlorine which may interfere with fluorescence. 4. Decolorize with 3% acid alcohol for two minutes 5. Rinse with distilled water and drain

6. Flood the smear with Potassium permanganate or acridine orange and allow to counter stain for two minutes. Time is critical with potassium permanganate because counter staining for longer time may quench the fluorescence of acid fast bacilli. 7. Rinse with distilled water and drain 8. Allow the smear to air dry. Do not blot. Read as soon as possible. Examine fluorescence stained smear within 24 hours as the fluorescence may fade with time.
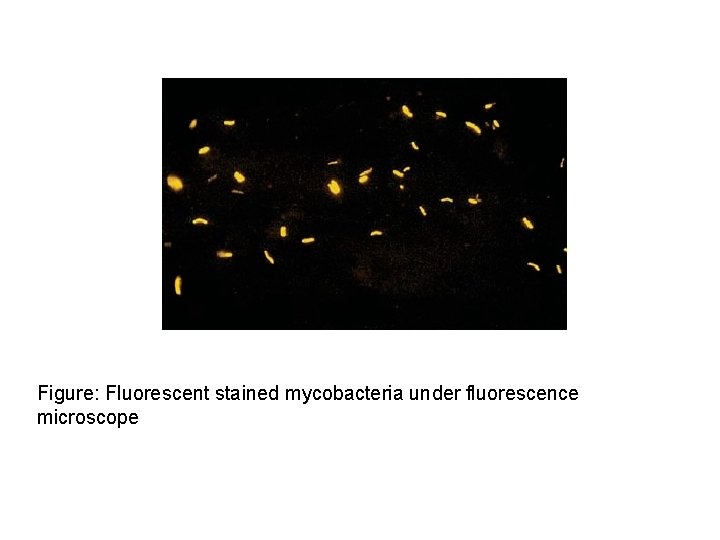
Figure: Fluorescent stained mycobacteria under fluorescence microscope

3. Special Staining method • These are stains, which are used to stain capsules and spores. There are two types of special staining methods A. Capsule Staining method B. Spore staining method

A. Capsule staining method • This technique is used for showing the presence of capsules around bacteria Required reagents • Crystal Violet ……………. . 10 g/l (1% w/v) stain • Copper sulphate………… 200 g/l (20%w/v)

Procedure 1 Fix the direct smear using alcohol Note: when preparing the smear, mix the organism suspension with a drop of normal serum this will help to show the capsules 2 Cover the smear with crystal violet stain and heat gently until the steam just begins to raise, leave to stain for 1 minute. 3 Wash off the stain with the copper sulfate solution N. B don’t use water 4 Wipe the back of the slide clean and place in a draining rack for smear to air dry 5 Examine the smear microscopically first with the 40 x, then with the 100 x oil immersion objective to look for capsulated bacteria.
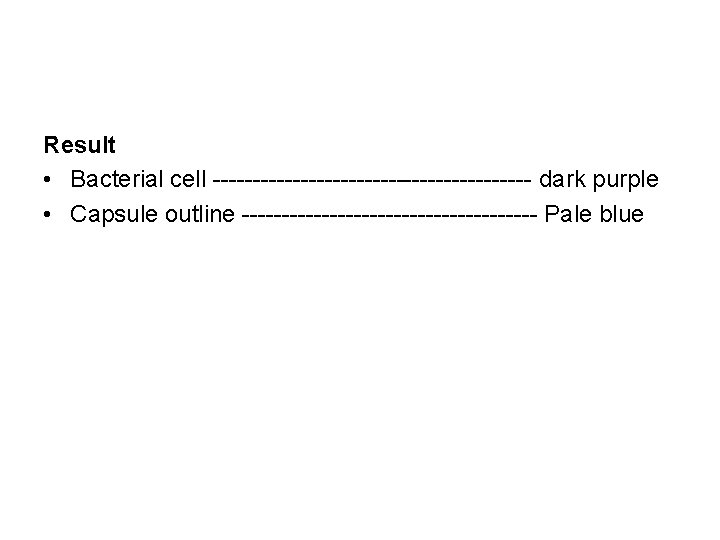
Result • Bacterial cell -------------------- dark purple • Capsule outline ------------------- Pale blue

B. Spore staining method § It is based on the binding of the malachite green and the permeability of the spore vs. cell wall. The steaming helps the malachite green to permeate the spore wall. • The primary dye malachite green is a relatively weakly binding dye to the cell wall and spore wall. In fact, if washed well with water, the dye comes right out of the cell wall. That is why there does not need a decolourizer in this stain.

Materials needed: – – Dye kit (malachite green, Safranin) Stain rack Hot plate Paper towel (cut the size of the slide) Procedure 1. Make a smear, air-dry and heat-fix. 2. Put a beaker of water on the hot plate and boil until steam is coming up from the water. Then turn the hot plate down so that the water is barely boiling. 3. Place the wire stain rack over the beaker which now has steam coming up from the boiled water. 4. Cut a small piece of paper towel and place it on top of the smear on the slide. The towel will keep the dye from evaporating too quickly, thereby giving more contact time between the dye and the bacterial walls.

5. Flood the smear with the primary dye, malachite green, and leave for 5 minutes. Keep the paper towel moist with the malachite green. DO NOT let the dye dry on the towel. 6. Remove and discard the small paper towel piece. 7. Wash really WELL with water and move the slide and wire rack from the boiling water to the regular stain tray to finish up the last step in the procedure. 8. Place the smear in the stain jar or flood the smear with the counter stain dye, Safranin, and leave for 1 minute.

The cold method spore stain • Without heat you have to really rough up the spore wall to get in the dye. • Heat fix the smear by running the slide through the flame about 20 times, and leave malachite green on for 20 minutes during the stain process.

Interpretation • Spores will be a light green: • Vegetative cell walls will pick up the counter stain safranin and become red(Red) • Notice: the position of the spore could be --terminal, central, or subterminal and this may be helpful in the identification of the unknown species of bacteria.

REFERENCE 1. 2. 3. 4. 5. 6. 7. 8. Mackie and Mc. Cartney, Practical medical microbiology 13 th ed. 1989 Monica Cheesbrough. Medical Laboratory Manual for Tropical Countries, Microbiology, volume II, 2 nd edition. Tropical Health Technology and Butter Worth-Heinemannith, 2006. Geo. F. Brooks, Janet s. Butel, Staphen A. Morse. Jawetz, Malnick and Adelberg's Medical Microbiology. 21 st edition. Appelton & Langh, 1998. T. D. Sleight, M. C. Murphy. Notes on Medical bacteriology, 2 nd edition. Churchill livingstone, Medical division of Longman group UK limited, 1986. Rajesh Bhatia, Rattan Lal Ichhpujmai, Essentials of Medial Microbiology, 1 st edition. Jaypee brothers Medical Publishers Ltd. 1994. Salle(1981). Fundamental principles of bacteriology, Ta. Ta Mc. Graw – Hill publishing Company Ltd, New Dalhi Mackie and Mc. Cartney(1989). Practical medical microbiology 13 th edition. Churchill Livingston Bernand D. Davis, Renanto Dulbecco, Herman N. Eisen and Harold S. Ginsberg(1990). Microbiology 4 th edition. Lipinocott Company. 9. Richard A. Harvey, Pamella C. Champ, Microbiology, Lippincott’s illustrated reviews, 2 nd ed. 10. Benson’s microbiological application, Laboratory manual in general microbiology, 8 th ed. 2001 11. Sherris, Medical microbiology, an introduction to infectious disease. 4 th ed. 2004. 12. Baron's Medical Microbiology 4 th edition, 2000 13. Jawetz, Melnick, & Adelberg's Medical Microbiology, 24 th Ed, 2007 14. Abilo Tadesse, Meseret Alem, University of Gondar. , Ethiopia Public Health Training Initiative, The Carter Center, Minstry of Health, and the Ethiopia Ministry of Education, 2006
- Slides: 90