CHAPTER II PATHOGENIC GRAM POSITIVE COCCI Objectives Upon

CHAPTER II PATHOGENIC GRAM POSITIVE COCCI

Objectives Upon completion of this unit of instruction, the student will be able to: • Discuss the basic characterisic of gram positive cocci • Describe the virulent factor of Gram positive cocci • Discuss pathogenicity, clinical manifestations, laboratory diagnosis, prevention & control of members of the Gram positive cocci

ü There are two most important genera of gram-positive cocci • The Genus staphylococcus • The Genus Streptococcus A. The Genus staphylococcus • Staphylococci make a very large contribution to man's Commensal flora and also account for a high proportion of his acute and chronic supportive lesions. ü General properties /characteristics – Gram-positive cocci arranged in Grape like clusters – Non – Motile, non – spore-forming – Produce catalase The genus: – Has grater than 30 Spp

– Contain 3 main Species of Clinical importance: • S. aureus • S. epidermis • S. saprophytius

a. Staphylococcus aureus – The most pathogenic of the group ü Coagulase production differentiate it from the others • Epidemiology • It is normal flora on human skin & nose (25 – 75% carriers) • Transmission is from person to person through direct contact or exposure to contaminated fomites • Risk factors include presence of a foreign body such as suture and catheter, previous surgical procedure and use of immuno-suppresser drugs

Antigenic composition ü S. aureus cell wall components & antigens: • Teichoic acid, protein A: – It binds to Fc portion of Ig. G, used in co-agglutination. • Capsule in some strains, make it more virulent surface receptors for bacteriophages that permit ‘phage typing’ for epidemiologic purposes • Coagulase - causes the clumping of non-capsulated strains when mixed with a solution containing fibrinogen, e. g. plasma. Protein A • Protein A is a surface protein of S. aureus which binds Ig. G molecules by their Fc region. • In serum, the bacteria will bind Ig. G molecules in the wrong orientation on their surface which disrupts opsonization and phagocytosis.

Mutants of S. aureus lacking protein A are more efficiently phagocytosed in vitro, and mutants in infection models have diminished virulence. Super antigens: • Superantigens: family of secreted proteins that share the ability to trigger massive activation of T cells, cytokine production produce intense polyclonal immune response - eg TSST-1 • Mos producing superantigens: G+ve bacteria – (staph, strept), Gram negative bacteria (E. coli), mycoplasma, viruses • Diseases Associated with Superantigen production • Toxic shock syndrome, psoriasis, rheumatoid arthrits, Diabetes mellitus, Scarlet fever, Eczema

Enzymes & toxins produced by S. aureus 1. Coagulase production – Most definitive virulence factor of S. aureus 2. Cytotoxins: α, β, γ toxins and P-V leukocidin, δ Haemolysins: – cause lysis of RBC of many animal spp demonstrated as β-hemolysis on blood agar. 3. Exfoliative toxin: – Cause the desquamation seen in SSSS in young children- produced by phage group II strains

4. Toxic shock syndrome toxin (TSST-1): – Produced by strains that cause the toxic shock syndrome (manifest by fever, shock, etc) 5. Enterotoxins: – 6 (A-F) soluble toxins produced by 50% of S. aureus strains – Toxins are heat stable and resistant to the action of gut enzymes – They cause diarrhoea & vomiting associated with staphylococcal food poisoning. 6. Other toxins and enzymes include: – Leukocidin - Staphylokinase – Proteinase - Lipase – Catalase - Hyaluronidase
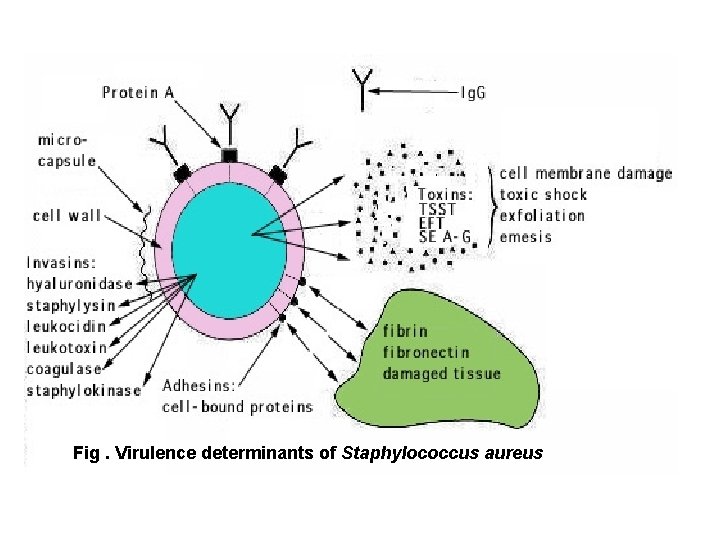
Fig. Virulence determinants of Staphylococcus aureus

Pathogenesis: • Pathogenesis of Stap. cocci is combined effect of: • Extra cellular factors • Toxins • Invasive properties of the strain • S. aureus causes suppurative and toxinoses in humans. • It causes superficial skin lesions, pneumonia, deepseated infections, food poisoning • It expresses many potential virulence factors ü Staphylococci produce disease through: • Their ability to multiply & invade tissue, • Production of extracellular enzymes & toxins • Enzyme-like protein that causes coagulation of plasma with fibrin deposition around the lesions render more resistant to phagocytosis

Diseases produced by S. aureus A. Focal suppuration and abscess formation: – Characteristic of S. aureus lesions due to coagulase which deposits fibrin around the lesions forming a wall which is reinforced by inflammatory cells. 1. Superficial infection: – Folliculitis, carbuncles, boils, stye, mastitis, abscess formation 2. Deep-seated lesions: – Osteomyelitis, septic arthritis, endocarditis, meningitis, endocarditis, bronchopneumonia, empyemia, etc. 3. Bacteraemia with multiple abscesses in tissues. – Outbreaks of hospital wound infections commonly occur due to antibiotic resistant staphylococci.

B. Toxin-mediated staphylococcal diseases 1. Food poisoning – Results from ingestion of preformed enterotoxin in contaminated food that is improperly cooked and kept unrefrigerated for some time. – Source of contamination of food: the hands or nose of a cook / food handlers /carriers. – Types of food involved in staphylococcal food poisoning are carbohydrate rich foods, e. g. cakes, pastry, milk, etc. – IP: short (1 -8 hrs) followed by nausea, vomiting, diarrhoea and general malaise with no fever.

2. Toxic shock syndrome (TSS): – This is associated with TSST-1, first described in menstruating women using tampons. The syndrome also occurs with wound or localized infections. – TSS has an abrupt onset of fever, vomiting, diarrhoea, muscle pains, rash – Hypotension, heart and renal failure may occur in severe cases.
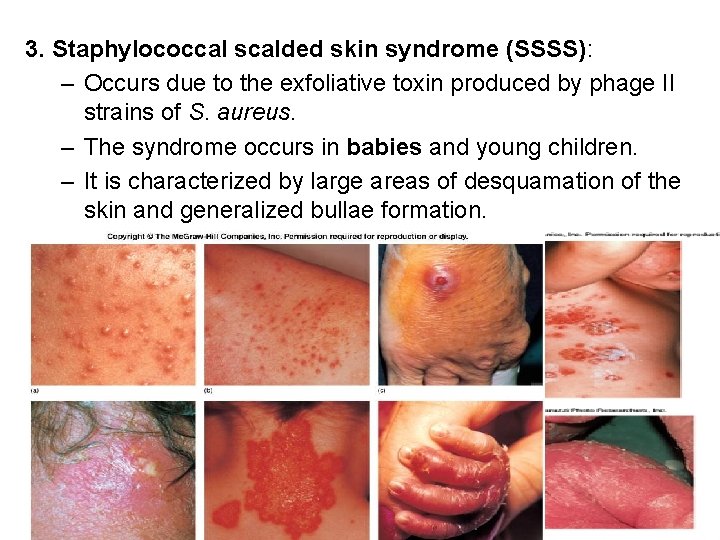
3. Staphylococcal scalded skin syndrome (SSSS): – Occurs due to the exfoliative toxin produced by phage II strains of S. aureus. – The syndrome occurs in babies and young children. – It is characterized by large areas of desquamation of the skin and generalized bullae formation.

Clinical findings – S. aureus infection results from direct contamination of a wound E. g. * Postoperative staphylococcal wound infection * Infection following trauma * Chronic osteomyelitis subsequent to open fracture * Meningitis following skull fracture – Food poisoning caused by enterotoxins • Characterized by: – Violent nausea – Vomiting – Diarrhea with out fever

– Toxic shock syndrome toxin is manifested by: • • • High fever Hypertension Vomitiny Cardiac & renal failure Diarrhea Myalgia

Virulence Factors of S. aureus • Toxins Ø Ø Cytotoxins- Toxic to many cells (leuc. , RBC, Plt, Mac. ) Exfoliative toxins- serine proteases Enterotoxin - superantigen TSST-1 - superantigen • Enzymes v Coagulase- fibrinogen to fibrin v Hyaluronidase- hydrolyzes Hyaluronic acid v Fibrinolysin- dissolves fibrin clots v Lipases – hydrolyzes lipid • Structural component – – Capsule Peptidoglycan Protien A Teichoic Acid 18

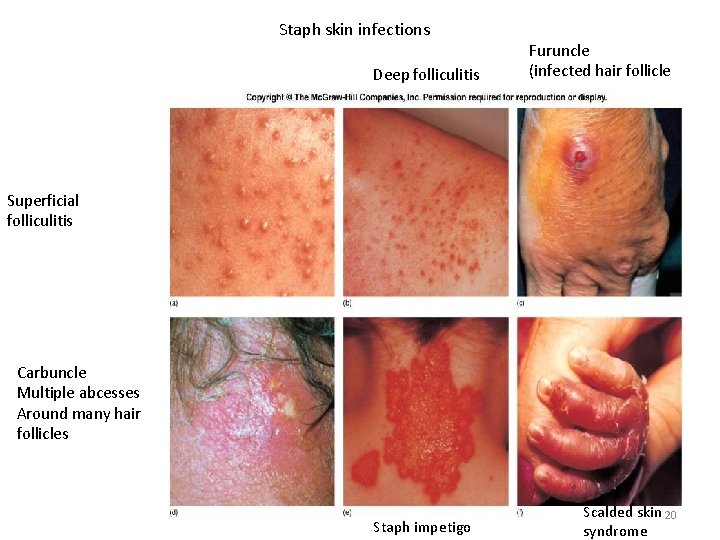
Staph skin infections Deep folliculitis Furuncle (infected hair follicle Superficial folliculitis Carbuncle Multiple abcesses Around many hair follicles Staph impetigo Scalded skin 20 syndrome

Lab. Diagnosis Potential specimens: depends on site of infection Pus , Tracheal aspirates , Blood , CSF Laboratory Diagnosis: Microscopy: gram-positive cocci in grape – like cluster Culture: produce beta-hemolytic colonies on blood agar Biochemically: Coagulase and catalase test – positive S. aureus is distinguished from the coagulase-negative staphylococci primarily by coagulase positivity. In addition, S. aureus colonies tend to be yellow and β-hemolytic, rather than gray and non hemolytic like the coagulase-negative staphylococci. S. aureus is also distinguished from most coagulase-negative staphylococci by being mannitol-positive. On Mannitol salt agar: - it forms Yellow colonies - MSA is selective media for Staph. (contain high salt conc. ) A. Smears Not helps to distinguish among different Spp.
Figure: Gram positive cocci
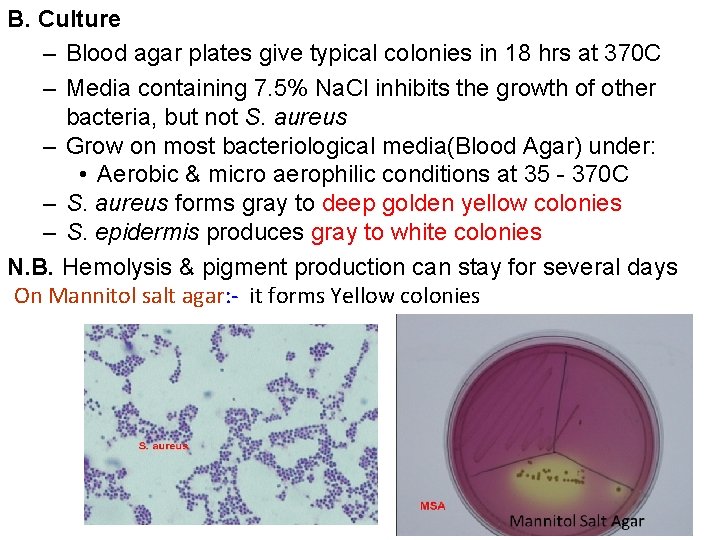
B. Culture – Blood agar plates give typical colonies in 18 hrs at 370 C – Media containing 7. 5% Na. Cl inhibits the growth of other bacteria, but not S. aureus – Grow on most bacteriological media(Blood Agar) under: • Aerobic & micro aerophilic conditions at 35 - 370 C – S. aureus forms gray to deep golden yellow colonies – S. epidermis produces gray to white colonies N. B. Hemolysis & pigment production can stay for several days On Mannitol salt agar: - it forms Yellow colonies

Biochemical Test C. Catalase Test – A drop of H 2 O 2 + small amount of bacterial growth on a slide – Formation of bubbles (release of O 2) ----- Positive test D. Coagulase Test – Human / citrated rabbit / plasma diluted 1: 5 (+) equal volume of growth from colonies from broth culture – Incubated at 370 C • Positive test: If clots form in 1 - 4 hrs N. B. All coagulase positive staphylococci are considered pathogenic to humans Susceptibility Testing – Disk disffusion susceptibility testing to penicillin detected by a pos. test for ß -lactamase Serological Testing • Anti-bodies to teichoic acid (Chronic infections)

b. Coagulase negative staphylococci (Co. NS) • Slime production is an important virulence factor for Co. NS. 1. S epidermidis – Inhabitant of the skin. – It is involved in indwelling catheters, prosthetic materials, shunts, surgery – It is a common cause of prosthetic heart valve endocarditis – It also causes nosocomial bacteremia – Often multiple antibiotic resistance - Methicllin

Co. NS 2. S. saprophyticus • Commonly isolated from animals and their carcasses. • Coagulase and phosphatase -ve, urease & Lipase +ve ü S. saprophyticus is resistant to the antibiotic Novobiocin, a characteristic used in Lab. to distinguish it from S. epidermidis • It is a 2 nd most common cause of UTI, after E. coli in young women accounting for 10 -20%. • It also causes soft tissue infections • Quinolones are commonly used in treatment of S. saprophyticus UTI.
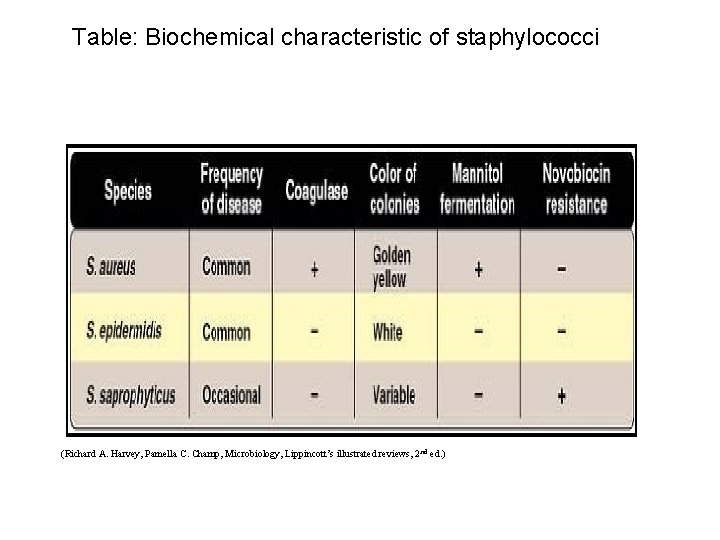
Table: Biochemical characteristic of staphylococci (Richard A. Harvey, Pamella C. Champ, Microbiology, Lippincott’s illustrated reviews, 2 nd ed. )
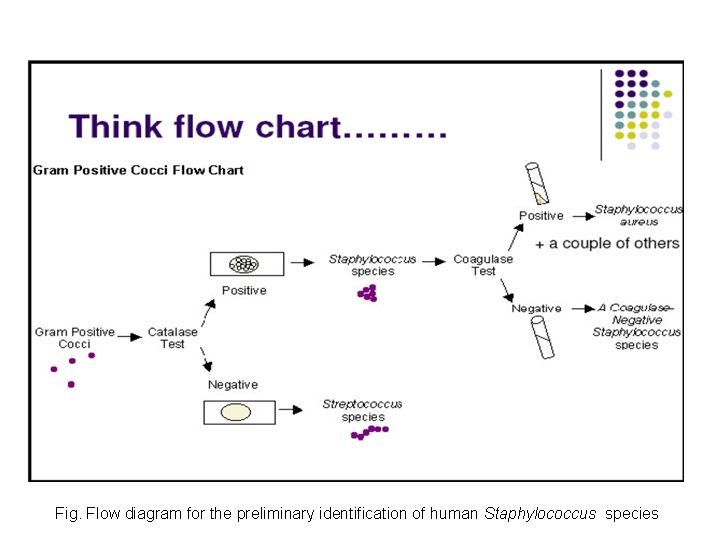
Fig. Flow diagram for the preliminary identification of human Staphylococcus species

Treatment – Penicillin G for sensitive isolates • Tetracycline, Amino glycosides, Cephalosporins, Erythromycins, Vancomycin ü MRSA (ORSA): methicillin (oxacillin)-multiresistant S. aureus, resulting from acquisition of mec. A, which encodes a novel PBP (PBP 2’) that is not bound by -lactams but retains its normal activities. Prevention: – No effective immunization with toxoid or bacterial vaccines – Cleanliness, Hand washing & aseptic management of lesions – Treat shaders in skin & nose – Remove carriers from high- risk areas: E. g. operating Rooms

Genus Streptococcus Characteristics • Gram positive cocci arranged in chain • S. pneumonia is exceptions (lanceolat and diplococci) • Most are facultative anaerobes • Grow best on enriched media and increased CO 2 con. • Some members are encapsulated • Catalase (-): distinguish from Staphylococcus(+) 30

Species Classification System Species differentiation with in the genus is complicated and they could be classified based on: 1. Hemolytic Reactions (Growth Media = Blood Agar) – – – α-Hemolysis: partial lysis of RBCs β-Hemolysis: complete lysis of RBCs γ-Hemolysis: no lysis of RBCs 2. Lancefield Classification – Grouping system based on cell wall antigens Ø Stimulate Ab formation – – Divided from group A U Groups A, B and D are common human pathogens 3. Biochemical Properties used for species not be classified into the Lancefield classification – – – Growth in 6. 5% Na. Cl soln. (high salt/adverse conditions) Sensitivity to Bacitracin or Optochin Bile solubility: some Step. bacteria can grow in bile 31
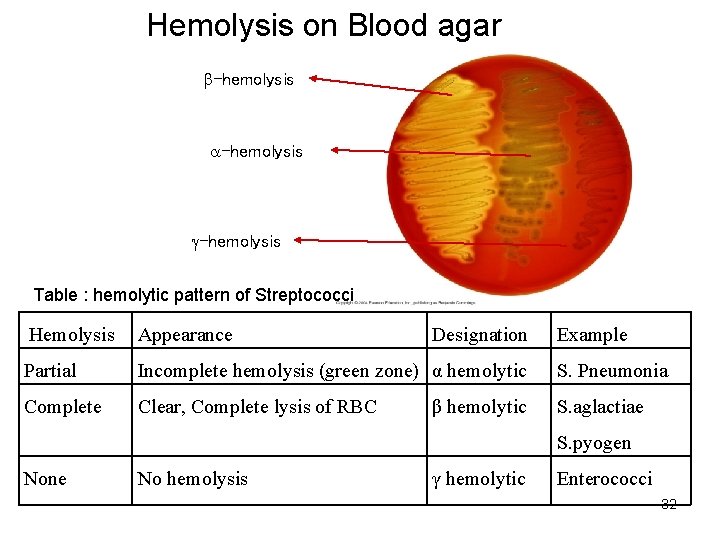
Hemolysis on Blood agar -hemolysis Table : hemolytic pattern of Streptococci Hemolysis Appearance Designation Partial Incomplete hemolysis (green zone) α hemolytic S. Pneumonia Complete Clear, Complete lysis of RBC S. aglactiae β hemolytic Example S. pyogen None No hemolysis γ hemolytic Enterococci 32

33

Medically important streptococci included: - 34

Streptococcus pyogens (Group A) General characteristics • Is most pathogenic among the streptococci group Gram positive cocci which occurs commonly in chain Grow best on Blood agar (beta hemolytic) Catalase negative and facultative anaerobes 35

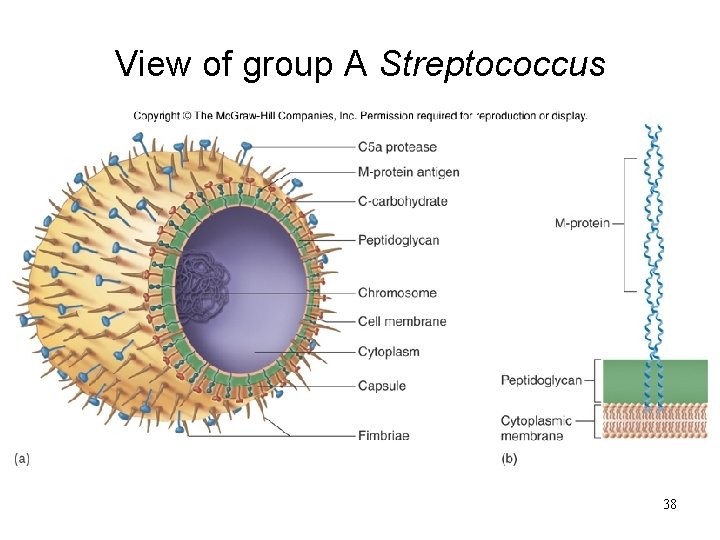
• Has group-specific carbohydrate (Lancefield group A antigen) and type specific protein (M protein) • M protein is the major type-specific protein associated with virulent strains • The protein is anchored in the cytoplasmic membrane, extends through the cell wall, and protrudes above the cell surface • The carboxyl terminus is anchored in the CM • The amino terminus, which extends above the cell surface, is responsible for the antigenic variability observed among the unique serotypes of M proteins. • M proteins are subdivided into class I and class II molecules 36

• The epidemiologic classification of S. pyogenes is based on sequence analysis of the emm gene that encodes the M proteins • Other important components in the cell wall of S. pyogenes include M-like surface proteins, lipoteichoic acid, and F protein. • Lipoteichoic acid and F protein facilitate binding of host cells by complexing with fibronectin, which is present on the host cell surface 37
View of group A Streptococcus 38

ü The virulence of group A streptococci is determined by the ability of the bacteria Ø to avoid opsonization and phagocytosis, Ø to adhere to the surface of host cells, Ø to invade into the epithelial cells, Ø to produce variety of toxins and enzymes or Virulence: Is associated with surface Ag’s, toxins and enzymes 39

1. Cell Surface Ag’s Ø Hyaluronic Acid Capsule • Antiphagocytic factor • Not immunogenic b/c hyaluronic acid is normal component of human connective tissue Ø M Protein • Component of fimbriae • Antiphagocytic • Attachment factor Ø C-CHOs (Cell Wall CHO’s) • Protection – protects bacterium from being dissolved by lysozyme defense of host Ø Lipoteichoic Acid: cell surface molecule • Adherence factor – attachment to epithelial cells of skin & pharynx (lipoteichoic acid, M proteins, and F protein the most 40 important mediate adhesion)

2. Extracellular Toxins Ø Hemolysins (destroy RBC, WBC, and platelet) v Streptolysin O: ü O 2 -labile ücauses hemolysis(beta) deep in blood agar plates. ü Antigenic ü ASO (antistreptolysin O) titer >160 -200 units suggests recent infecn. v Streptolysin S: üO 2 -stable. ünot Causes Beta-hemolysis on the surface of blood agar plates. üNot antigenic Ø Can also kills phagocytes by releasing the lysosomal contents after engulfment. 41

Ø Erythrogenic Toxin (pyrogenic toxin) v Toxin induces a fever with alteration of temperature regulatory system v Toxin responsible for bright red rash typical of scarlet fever 3. Enzymes: Streptokinase (fibrinolysin) Ø Can lyse blood clots and may be responsible for the rapid spread of the organism. Streptodornase (DNases A to D) • Decreases viscosity of DNA suspension. 42

Hyaluronidase (spreading factor): v Destroys connective tissue and aids in spreading of bacteria. C 5 a peptidase • Destroy C 5 a which is important as chemotactic signals • Prevents streptococci from C 5 a-mediated recruitment and activation of phagocytes, and is important for survival of S. pyogenes in tissue and blood. Transmission – Person to person from droplets during coughing, on the hand of health personnel, or from fomites like towels 43

Pathogenesis and Clinical disease • Responsible for suppurative diseases (pharyngitis, soft tissue infections, streptococcal toxic shock) and non suppurative diseases (rheumatic fever, glomerulonephritis) Pharyngitis/tonsillitis – bacteria colonize mucous membranes of pharynx and tonsils – Sx’s: redness, edema, enlargement and tenderness of throat (severe sore throat); painful and difficult swallowing; purulent exudate over the tonsils – If untreated, it may extend to otitis, sinusitis, mastoiditis, and meningitis 44

Scarlet fever • complication of streptococcal pharyngitis that occurs when the infecting strain is lysogenized by a temperate bacteriophage that mediates production of a pyrogenic exotoxin. • Characterized by a diffuse erythematous rash initially appears on the upper chest and then spreads to the extremities Pyoderma (impetigo) is a confined, purulent (pyo-) infection of the skin (derma) that primarily affects exposed areas (i. e. , face, arms, legs). • The strains of streptococci that cause skin infections differ from those that cause pharyngitis 45

Erysipelas: Acute infection of skin with lymph node enlargement and systemic sign (chills, fever and leukocytosis) red skin; commonly on leg Necrotizing fasciitis: (“flesh-eating bacteria”): Infection deep in subcutaneous tissues that spreads along fascial planes, destroying muscle and fat; Initially cellulitis followed by bullae (fluid filled blisters; bulla is singular), gangrene, systemic toxicity, multiorgan failure and mortality in more than 50% of patients 46

. • Sepsis (streptococcal toxic shock syndrome or toxic shock-like syndrome, TSLS): Ø the organism is introduced into the subcutaneous tissue through a break in the skin cellulitis necrotizing faciitis systemic toxicity, multiple organ failure, and death (mortality > 40%). 47

3. Poststreptococcal diseases (occurs 1 -4 weeks after acute S. pyogenes infection)- Immunogenic disorder Rheumatic fever: most commonly preceded by infection of the respiratory tract. Inflammation of heart (pancarditis), joints, blood vessels, and subcutaneous tissue. Results from cross reactivity of anti-M protein Ab and the human heart tissue. Acute glomerulonephritis: Ø preceded by infection of the skin (more commonly) or the respiratory tract. Ø Initiated by Ag-Ab complexes on the glomerular basement membrane. 48

Summary • S. pyogenes causes three types of diseases: (1) pyogenic diseases such as pharyngitis and cellulitis, (2) toxigenic diseases such as scarlet fever and toxic shock syndrome, and (3) immunologic diseases such as rheumatic fever and acute glomerulonephritis 49

Epidemiology S. pyogens colonize the oropharynx of healthy individuals Transmission is mainly droplet and enter through broken skin Risky individuals Children(5 to 15 y) Pharyngitis Children(2 -5 y) poor personal hygiene Skin Children with sore throat develop RF infection Rarely tissue infection can lead to bactermia STSS 50
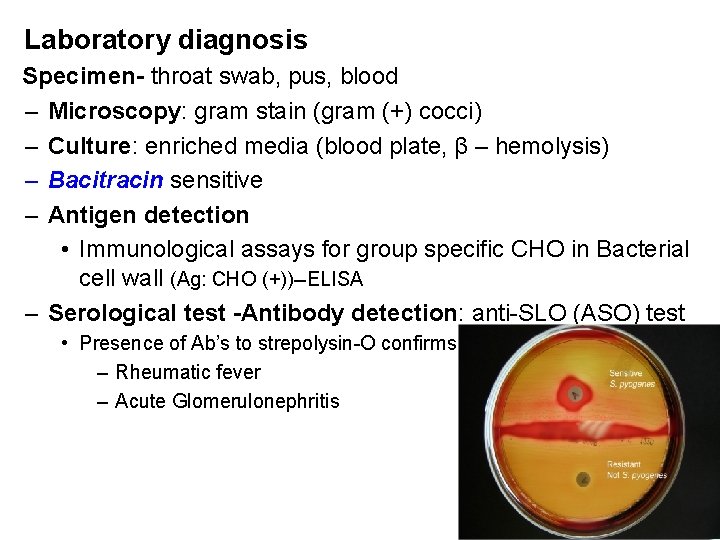
Laboratory diagnosis Specimen- throat swab, pus, blood – Microscopy: gram stain (gram (+) cocci) – Culture: enriched media (blood plate, β – hemolysis) – Bacitracin sensitive – Antigen detection • Immunological assays for group specific CHO in Bacterial cell wall (Ag: CHO (+))--ELISA – Serological test -Antibody detection: anti-SLO (ASO) test • Presence of Ab’s to strepolysin-O confirms – Rheumatic fever – Acute Glomerulonephritis 51
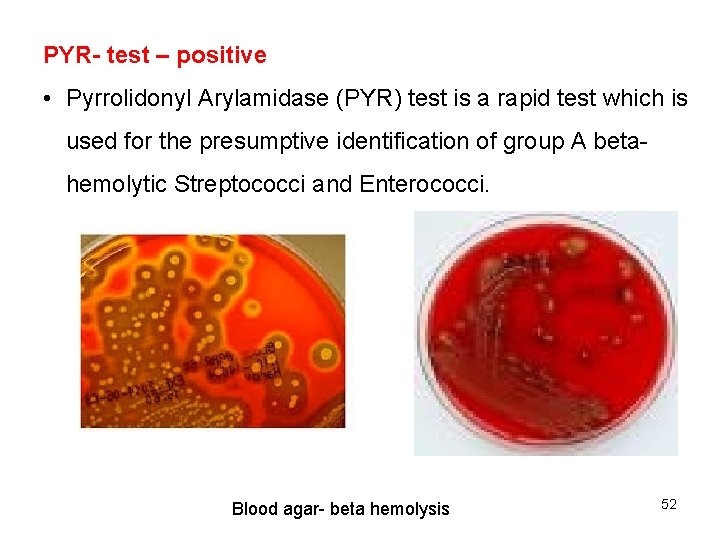
PYR- test – positive • Pyrrolidonyl Arylamidase (PYR) test is a rapid test which is used for the presumptive identification of group A betahemolytic Streptococci and Enterococci. Blood agar- beta hemolysis 52

• Treatment – Penicillin, erythromycin, cephalosporin Prevention ü Early detection and treatment ü Patient with history of RF should take prophylaxis treatment before doing certain procedures (dental extraction) to induce bacteremia endocarditis 53

Streptococcus pneunomiae (Pneumococcus) – Causative agent of Typical Pneumoniae • 80% of all bacterial pneumonias due to S. Pneunomiae – Normal flora of URT – General Characteristics • • • Gram (+) cocci occur singly or paired (diplococci) Lanceolate shaped Culture – blood agar or chocolate agar α – Hemolysis (partial hemolysis displayed; green color results) Catalase -negative Facultative anaerobe – grows best at elevated CO 2 54

Virulence Factors • Capsule – resists phagocytosis - Used for serotyping (classification) (91 types) • Pneumolysin – forms pores in host cell membrane by combining w/ cholesterol (in phospholipid bilayer) • Autolysin–PG hydrolase present in the CW; causes cell lysis – Responsible for release of intracellular Pneumolysin. Produce hydrogen peroxide which results tissue damage. Possess phosphoryl choline which helps the bacteria to attach on tissue cells 55

56

Contributing factors • Loss of natural resistance may be due to: 1. Viral respiratory infections (influenza or measles) 2. Alcohol or drug intoxication (interfere cilia function) 4. Malnutrition, general debility, spleen dysfunction or speenoectomy, nephrosis or complement deficiency ( have effect on bacterial clearance). 5. Young children and the elderly 57

Pathogenesis Diseases • S. pneumoniae causes lobar pneumonia – Sudden onset with fever, chills and sharp chest pain. Bloody or rusty sputum. • Complications caused by spreading of pneumococci to other organs: sinusitis, middle ear infection, meningitis, endocarditis, septic arthritis, bacteremia. • In tropical and developing countries, S. pneumoniae is a major pathogen, responsible for acute life-threatening pneumonia and bacteraemia in those co-infected with HIV. 58
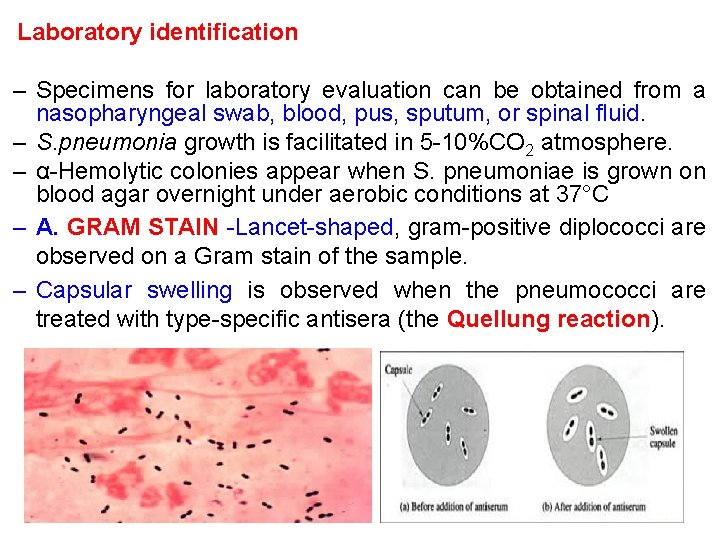
Laboratory identification – Specimens for laboratory evaluation can be obtained from a nasopharyngeal swab, blood, pus, sputum, or spinal fluid. – S. pneumonia growth is facilitated in 5 -10%CO 2 atmosphere. – α-Hemolytic colonies appear when S. pneumoniae is grown on blood agar overnight under aerobic conditions at 37°C – A. GRAM STAIN -Lancet-shaped, gram-positive diplococci are observed on a Gram stain of the sample. – Capsular swelling is observed when the pneumococci are treated with type-specific antisera (the Quellung reaction).
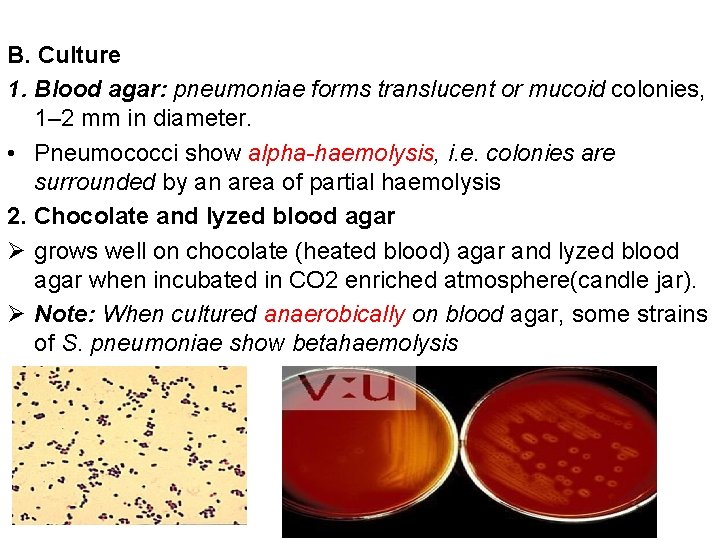
B. Culture 1. Blood agar: pneumoniae forms translucent or mucoid colonies, 1– 2 mm in diameter. • Pneumococci show alpha-haemolysis, i. e. colonies are surrounded by an area of partial haemolysis 2. Chocolate and lyzed blood agar Ø grows well on chocolate (heated blood) agar and lyzed blood agar when incubated in CO 2 enriched atmosphere(candle jar). Ø Note: When cultured anaerobically on blood agar, some strains of S. pneumoniae show betahaemolysis

C. Biochemical tests ● Catalase negative ● Sensitive to optochin (zone of inhibition should at least 10 mm) ● Bile soluble- positive Bile solubility test Ø Bile or bile salts (surface-active agents) activate an autolytic AMIDASE which cleaves the bond between alanine & muramic acid in the peptidoglycan resulting in lysis of microorganism – Amidase is present in pneumococcus but not in viridans streptococci

D. Direct detection of pneumococcal antigen in body fluid • Rapid latex and coagglutination tests are available to detect capsular pneumococcal antigen in CSF, pleural fluid, serum and urine. Treatment, • Penicillin is the drug of choice for penicillin sensitive strains • Cephalosprin, erythromycin and chloramphenicol (meningitis) are used for penicillin allergic or for penicillin resistant strain • Prevention • Vaccination of high-risk population (too old, too young, and people losing natural resistance) with vaccines • Immunization with 23 -valent vaccine

Streptococcus agalactiae (Group B beta-Hemolytic Strept. ) – S. agalactiae is found in the Ø vaginocervical tract of female carriers, and Ø urethral mucous membranes of male carriers, as well as in the GI tract. v. Transmission occurs from an infected mother to her infant at birth, and v venereally (propagated by sexual contact) among adults. – Group B streptococci are a leading cause of meningitis and septicemia in neonates, with a high mortality rate. – Group B streptococci are β-hemolytic with larger colonies and less hemolysis than group A. – About 5% of strains are nonhaemolytic

LABORATORY FEATURES • Specimens: Include • cerebrospinal fluid, ear swab and blood for culture from neonates. • High vaginal swab is required from women with suspected sepsis. Morphology Gram stain • Group B streptococci are Gram positive cocci, occurring characteristically in short chains but also in pairs and singly. • The organisms are non-motile. Most strains are capsulated.

Culture • Blood agar: Most strains of S. agalactiae produce grey mucoid colonies about 2 mm in diameter, surrounded by a small zone of beta haemolysis. • Mac. Conkey agar: Most strains grow on this medium. • Neomycin blood agar: A useful selective medium for isolating S. agalactiae from urogenital specimens. • Orange pigment: Produced by S. agalactiae when cultured on serum starch agar anaerobically.

Biochemical Tests 1. CAMP (Christie, Atkins, Munch, Peterson) test to identify presumptively S. agalactiae • This test requires the use of a beta-lysin producing strain of S. aureus (NCTC 1803 or ATCC 25923) to detect the CAMP factor, i. e. extracellular diffusible protein produced by S. agalactiae. This protein interacts with the staphylococcal betalysin on sheep (or ox) blood agar producing enhanced haemolysis.

• The test is performed by streaking a known beta-lytic Staphylococcus strain across a 10% blood agar plate and then inoculating the test organism at right angles to it. The test organism must not touch the staphylococcal inoculum. • An Enterococcus species is also inoculated as a negative control. • The test organism is presumed to be S. agalactiae, if after overnight incubation at 35– 37 ºC, there is an arrow-head shaped area of haemolysis where the staphylococcal organism meets the test organism (see Fig. ).
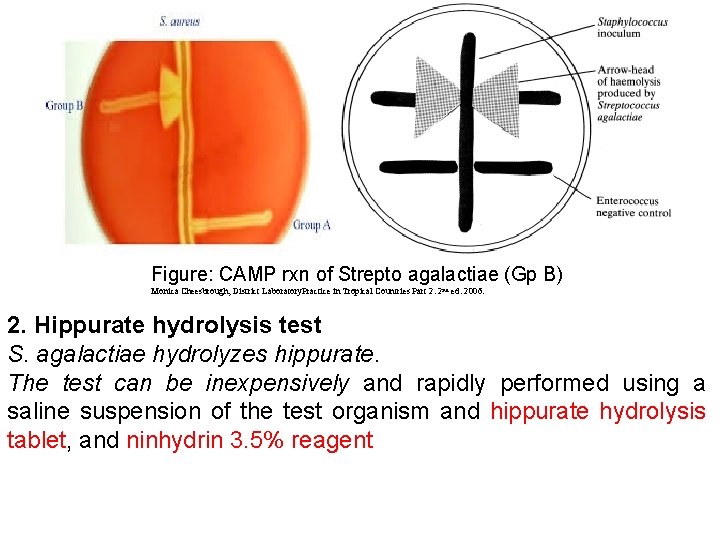
Figure: CAMP rxn of Strepto agalactiae (Gp B) Monica Cheesbrough, District Laboratory. Practice in Tropical Countries Part 2. 2 nd ed. 2006. 2. Hippurate hydrolysis test S. agalactiae hydrolyzes hippurate. The test can be inexpensively and rapidly performed using a saline suspension of the test organism and hippurate hydrolysis tablet, and ninhydrin 3. 5% reagent

Direct detection of Group B streptococcal antigen in CSF • When Group B streptococcal meningitis is suspected, a rapid diagnosis can be made by detecting Group B streptococcal antigen directly in CSF, serum or urine using a latex or coagglutination slide test. Treatment v S. agalactiae has the same susceptibility profile as S. pyogenes v Most isolates remain sensitive to penicillin G and ampicillin, which are still the antibiotics of choice.

Enterococcus species • Enterococci are aerobic organisms capable of growing over a wide temperature range, 10– 45 ºC. • They can grow in high salt con. (6. 5%Na. Cl) • Catalase negative • Can survive harsh environment (disinfec. , high Temp. , PH) • Pathogenicity Ø E. faecalis (formerly classified Streptococcus. faecalis) is the main pathogen in the genus Enterococcus, Ø causing about 95% of enterococcal infections including infections of the urinary tract, biliary tract, ulcers (e. g. bed sores), wounds and occasionally endocarditis or meningitis. Ø It is a normal commensal of the vagina and intestinal tract. Ø A minority of infections are caused by E. faecium.

LABORATORY FEATURES Morphology • Gram positive cocci, occurring in pairs or short chains. They are non-capsulate and the majority are non-motile. Culture • Blood agar: mainly nonhaemolytic but some strains show alpha or beta-haemolysis. • Mac. Conkey: E. faecalis ferments lactose, producing small dark -red magenta colonies on Mac. Conkey agar • Enterococcus species are also able to grow in the presence of 6. 5% sodium chloride and 40% bile. • When grown on media containing aesculin, enterococci hydrolyze the aesculin, producing black colonies.

Biochemical tests ● Ferment lactose (also mannitol and other sugars). ● Hydrolyze aesculin ● Reduce litmus milk • PYR test positive • Note: Like streptococci, enterococci are catalase negative. Treatment • Most enterococci are susceptible to ampicillin and resistant to cephalosporins. • Resistance is shown against penicillin. Vancomycin and ampicillin resistance appear to be emerging, associated particularly with hospital infections.


Viridans streptococci • Although often alpha-haemolytic on blood agar, the viridans group of streptococci can also be nonhaemolytic and occasionally beta-haemolytic. • They form part of the normal microbial flora of the upper respiratory tract (particularly oropharynx) and gastrointestinal tract. • They may therefore be found with S. pneumoniae in sputum (as commensals). • A few species are pathogenic (e. g. S. mutans, S. sanguis, S. mitior) causing endocarditis, bacteraemia, and dental caries.
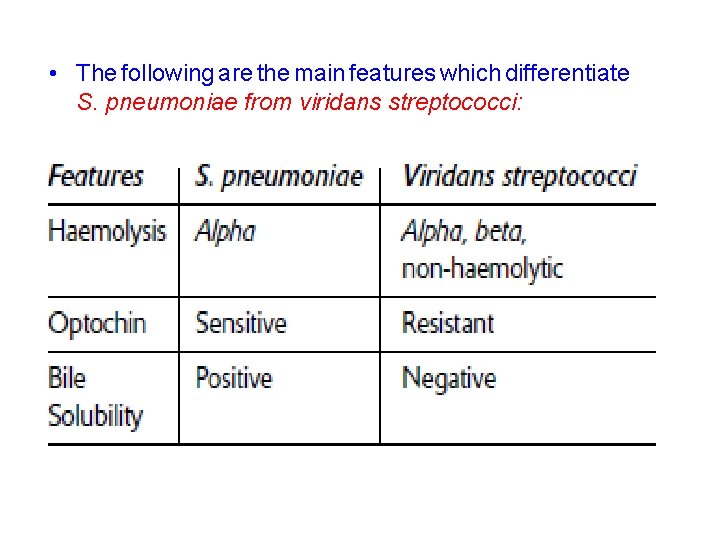
• The following are the main features which differentiate S. pneumoniae from viridans streptococci:

Table: Usual Hemolytic, Biochemical, and Cultural Reactions of Common Streptococci and Enterococcia (Sherris, Medical microbiology, an introduction to infectious disease. 4 th ed. 2004. )
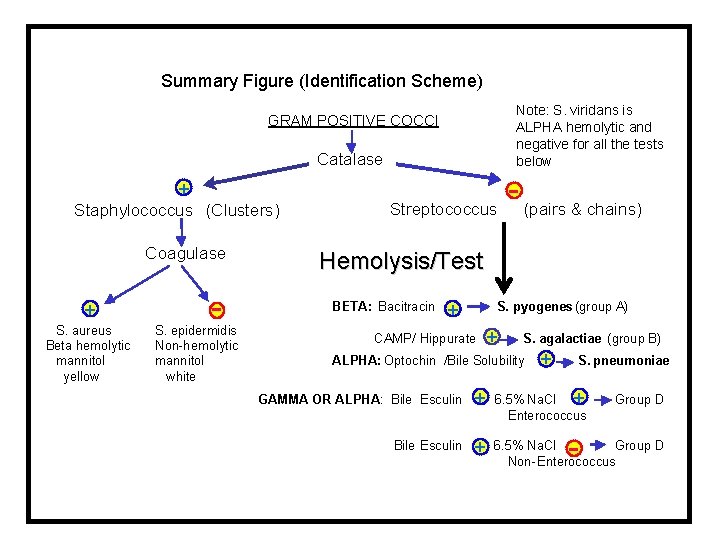
Summary Figure (Identification Scheme) Note: S. viridans is ALPHA hemolytic and negative for all the tests below GRAM POSITIVE COCCI Catalase + Staphylococcus (Clusters) Coagulase + S. aureus Beta hemolytic mannitol yellow - S. epidermidis Non-hemolytic mannitol white Streptococcus - (pairs & chains) Hemolysis/Test BETA: Bacitracin + S. pyogenes (group A) CAMP/ Hippurate + S. agalactiae (group B) ALPHA: Optochin /Bile Solubility + S. pneumoniae GAMMA OR ALPHA: Bile Esculin + 6. 5% Na. Cl Enterococcus + Bile Esculin + 6. 5% Na. Cl Group D Non-Enterococcus Group D - 77
- Slides: 77