Chapter Eleven UltravioletVisible Spectrophotometry Spectrophotometry is one of

Chapter Eleven Ultraviolet-Visible Spectrophotometry • Spectrophotometry is one of the most widely used methods of analysis in clinical chemistry, hygiene analysis, medicine analysis. • It is used to measure the absorption capabilities of certain compounds with reference to wavelengths of light on the ultraviolet and visible light ranges. 2008 -11 -19 1

• High sensitive, can used microanalysis 10 -3~ 10 -6 mol/L e. g. There is 10 -8 g Fe in 10 ml solution, if titrate it using KMn. O 4 standard solution (c=2. 0× 10 -4 mol/L), only consumed 0. 02 ml KMn. O 4 standard solution. 2008 -11 -19 2

11 -1 Properties of Light 11 -2 Absorption of Light * 11 -3 The Spectrophotometer 11 -4 Typical Analysis Procedures 2008 -11 -19 3

11 -1 Properties of Light • Light is electromagnetic wave • The dual nature of particles and waves *Wavelength λ unit: nm *Frequencyν (Hz) unit of frequency is s-1 Hertz: One oscillations per second is called 1 Hz 106 s-1 is therefore said to be 106 Hz, or one megahertz (MHz). *Wavenumber 2008 -11 -19 unit: nm-1, cm-1 4

The relationship between wavelength and frequency c is the speed of 1 ight in vacuum. c=2. 99792458 × 108 m s-1 2008 -11 -19 5

• The light with certain wavelength or frequency have definite energy • Photons • E=hv = Planck's constant h=6. 626176× 10 -34 J·s One mole of photons is called one einstein. 2008 -11 -19 6

• Complement Color: The observed color is said to be the complement of the absorbed color. • Any substance that purple red absorbs visible light orange will appear colored blue greenish blue 2008 -11 -19 white light Bluish green yellow green 7

Table 11 -1 Colors of visible light and complementary hue Wavelength Color Absorbed/nm absorbed observed 380 -420 Violet Green-yellow 420 -440 Violet-blue Yellow 440 -470 Blue Orange 470 -500 Blue-green Red 500 -520 Green Purple 520 -550 Yellow-green Violet 550 -580 Yellow Violet-blue 580 -620 Orange Blue 620 -680 Red Blue-green 680 -780 Purple Green 2008 -11 -19 8
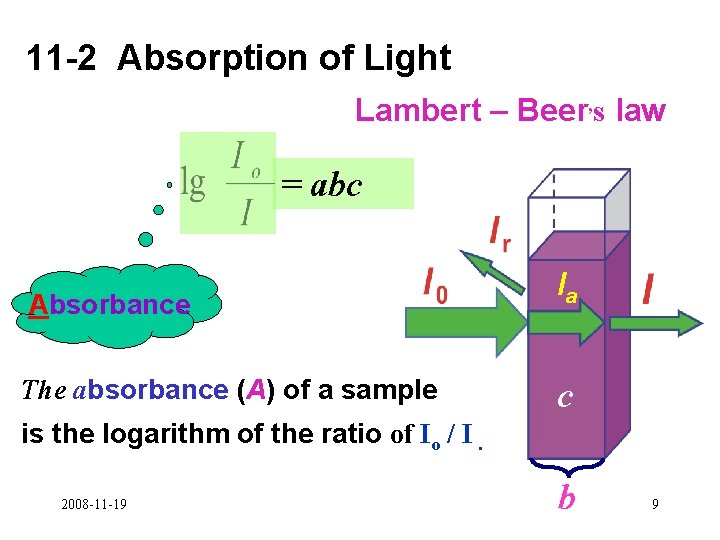
11 -2 Absorption of Light Lambert – Beer, s law = abc Absorbance Ia The absorbance (A) of a sample c is the logarithm of the ratio of Io / I. 2008 -11 -19 b 9

= abc Lambert–Beer, s law A- absorbance a - absorptivity (L·g-1·cm-1 ) b - path length (cm) c - mass concentration (g/L) c:mass concentration(g/L)→ concentration (mol·L-1) a:absorptivity (L/g·cm ) → ε (L·mol-1·cm-1 ) ε:the molar absorptivity 2008 -11 -19 10

Transmittance (T) is the ratio of I/Io. 2008 -11 -19 11

• T: range 0 ~ 1 • l 00·T : (the percent transmittance) varies between 0 and 100%. • When no light is absorbed T=1=100% , P=P 0 and A=0 I= I 0 and A=0 2008 -11 -19 12

• Lambert – Beer, s law, commonly called Beer's law: Absorbance is directly proportional to the concentration of light-absorbing species in the sample and the pathlength of the solution. Lambert–Beer, s law is strictly valid for purely monochromatic radiation; that is, for radiation consisting of only one wavelength. Monochromatic light 2008 -11 -19 13
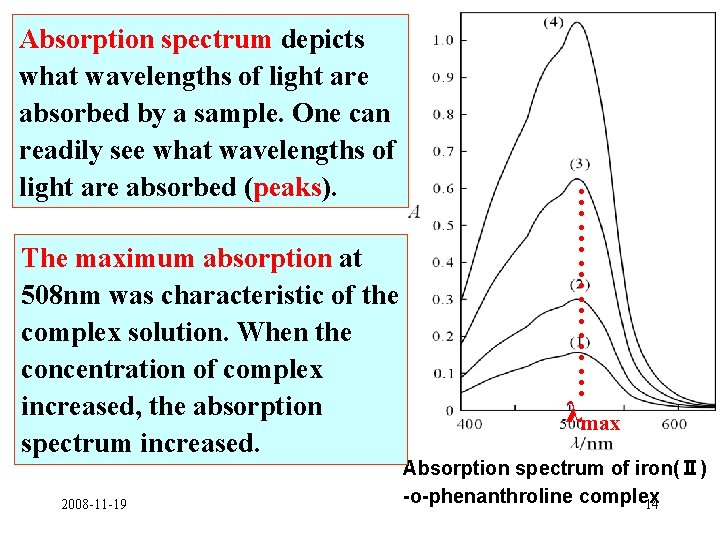
The maximum absorption at 508 nm was characteristic of the complex solution. When the concentration of complex increased, the absorption spectrum increased. 2008 -11 -19 ……………… Absorption spectrum depicts what wavelengths of light are absorbed by a sample. One can readily see what wavelengths of light are absorbed (peaks). λmax Absorption spectrum of iron(Ⅱ) -o-phenanthroline complex 14
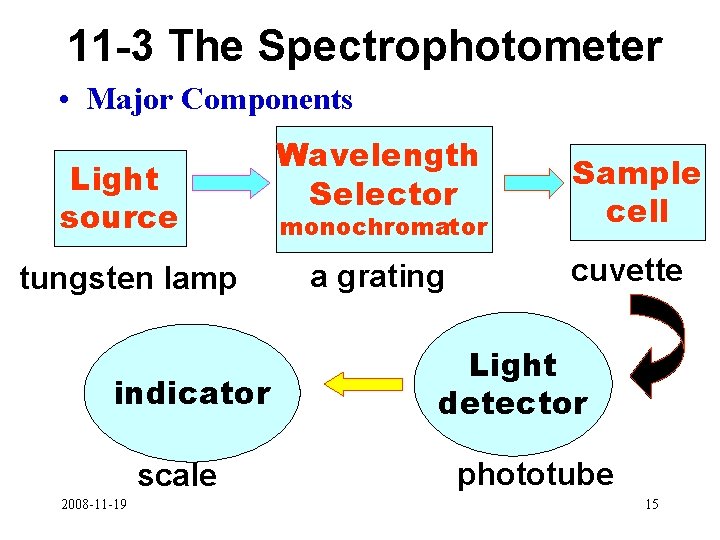
11 -3 The Spectrophotometer • Major Components Light source tungsten lamp indicator scale 2008 -11 -19 Wavelength Selector monochromator a grating Sample cell cuvette Light detector phototube 15

11 -4 Typical Analytical Procedures Methods and Applications of Spectrophotometry Ⅰ. Direct comparison method A 1=εbc 1 A 2=εbc 2 2008 -11 -19 Direct comparison method 16
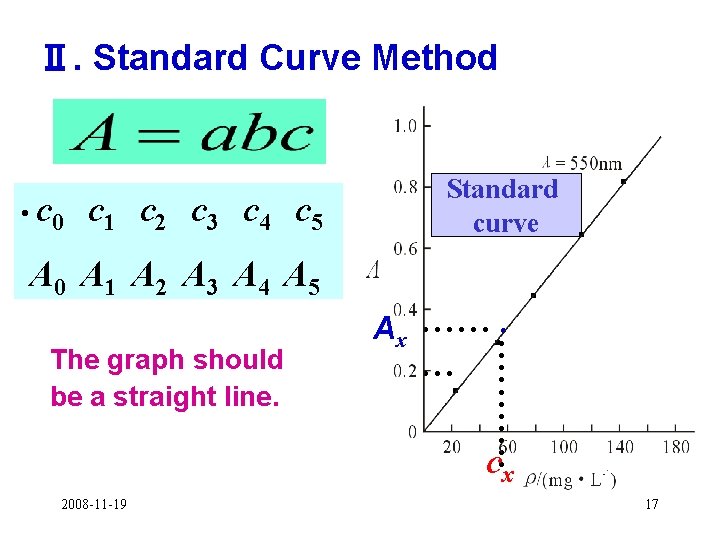
Ⅱ. Standard Curve Method • c 0 c 1 c 2 c 3 c 4 c 5 Standard curve A 0 A 1 A 2 A 3 A 4 A 5 ···· · · ······ The graph should be a straight line. Ax ······ · cx 2008 -11 -19 17

• Analytical Procedures 1) Selection of monochromatic light (Choice of Wavelength) 2) Prepare a reagent blank containing all reagents, but with analyte replaced by distilled water. 3) Prepare a series of standards solution and measure their absorbance 4) Establish a calibration curve 5) the unknowns be prepared using the same procedure as for standards solution 6) Calculate the content of unknown 2008 -11 -19 18

Choice of λmax Analytical Prepare solutions Measure their absorbance Procedures Establish a calibration curve Calculate the content of unknown 2008 -11 -19 19

Example: A sample in a 1. 0 cm cell is determined with a spectrometer to transmit 80% light at a certain wavelength. If the absorptivity of this substance at this wavelength is 2. 0, 1) what is the concentration of the substance(g/L). 2) what is the absorbance of the substance 2008 -11 -19 20

Solution: The percent transmittance is 80%, and so T=0. 80,a=2. 0,b=1. 0 A=abc = - log. T = -log 0. 80 = 0. 0969 2008 -11 -19 21

Problems: 1. The transmittance of a solution is found to be 2. 35. 0%. What is the transmittance if the solution 3. is diluted in half? 2. The transmittance of a solution is found to be 85. 0% when measured in a cell whose path length is 1. 00 cm. What is the transmittance if the path length is increased to 4. 00 cm ? 2008 -11 -19 22

3. A colored substance M has an absorption maximum at 520 nm. A solution containing 2. 00 mg M per liter has an absorbance of 0. 840 using a 2. 00 cm cell. The formula weight of M is 150. (a) Calculate the molar absorptivity of M at 520 nm. (b) How many milligrams of M are contained in 25. 00 m. L of a solution giving an absorbance of 0. 250 at 520 nm when measured with a 1. 00 cm cell ? 4. The molar absorptivity of benzoic acid (M=122. 1 g·mol 1) in methanol at 275 nm is about 1950 L·mol-1·cm-1. If it is desired to use an absorbance not exceeding 1. 25, what is the maximum allowable concentration in g·L-1 that can be used in a 2. 00 cm cell. 2008 -11 -19 23
- Slides: 23