Chapter Biological Oxidation Lecture 1 Biological oxidation Lecture

Chapter Ⅵ Biological Oxidation

Lecture 1 Biological oxidation Lecture 2 Oxidative phosphorylation

LECTURE 1 BIOLOGICAL OXIDATION

Biological oxidation, the process which nutrient substance, such as saccharides, lipids, and proteins are oxidized into water and carbon dioxide, and simultaneously produce energy.
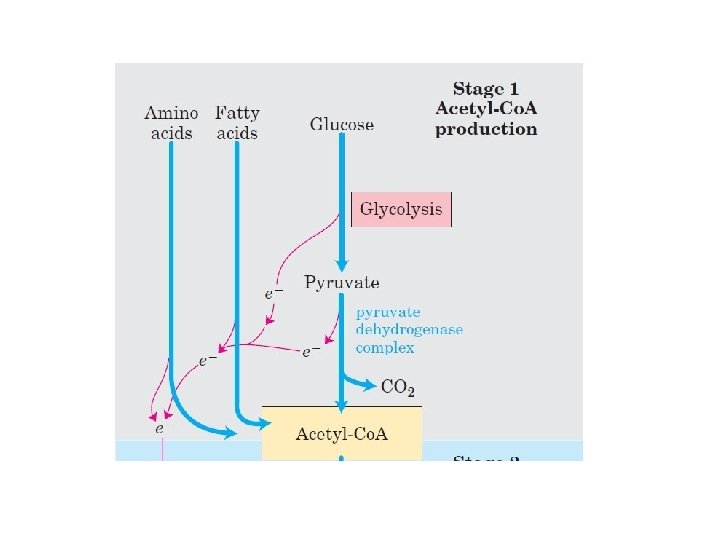
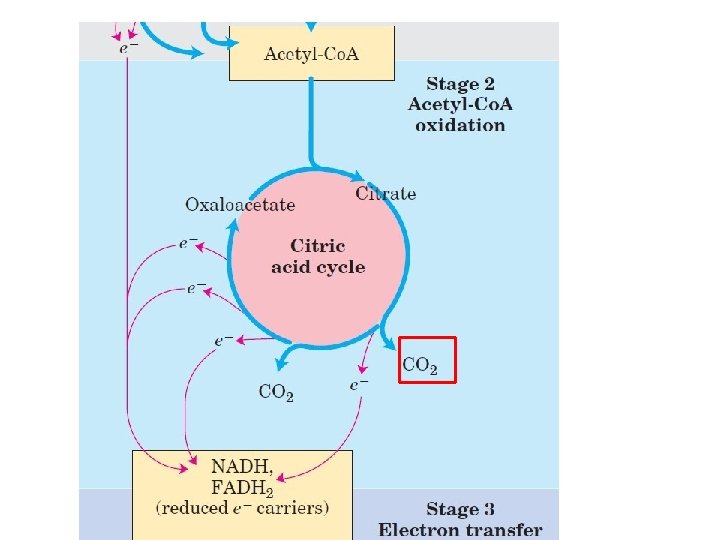
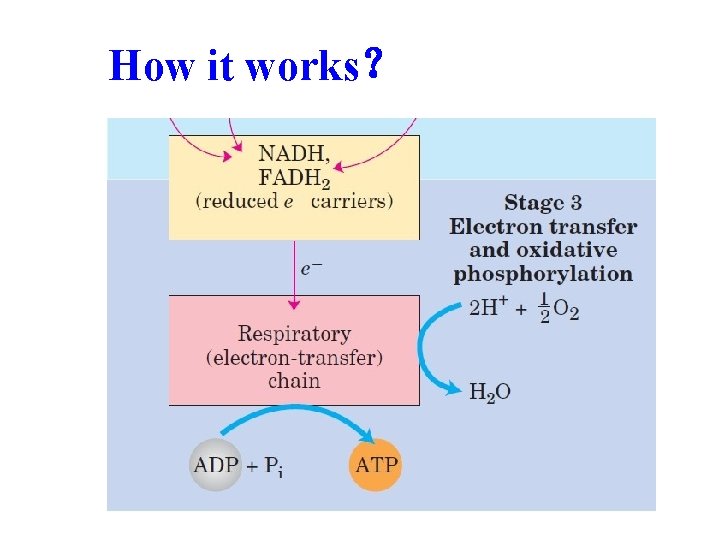
How it works?

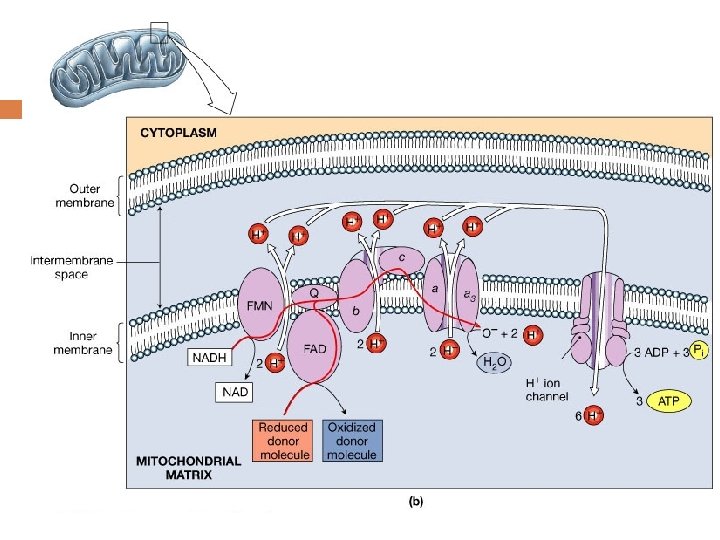
1. Overview of Membrane-associated Electron Transport and ATP Synthesis As electrons pass along the electron transport chain from NADH to O 2 the energy they release is used to transfer protons from inside the mitochondrion to the intermembrane space between the double membranes. This proton gradient is used to drive ATP synthesis in a reaction catalyzed by ATP synthase
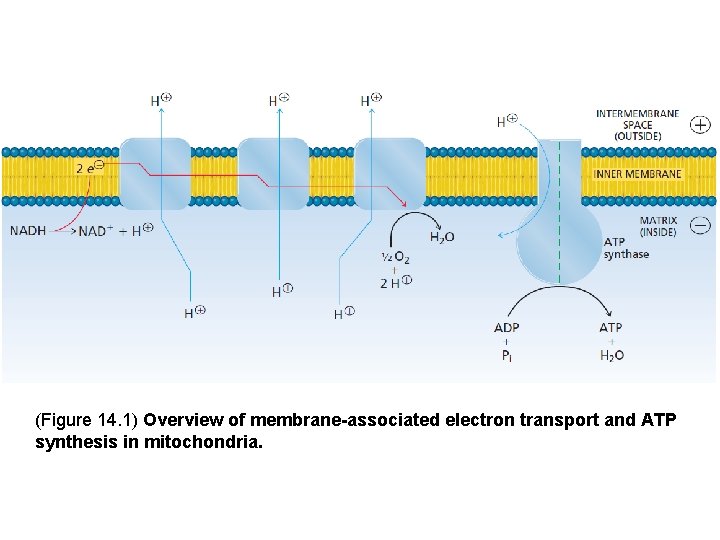
(Figure 14. 1) Overview of membrane-associated electron transport and ATP synthesis in mitochondria.
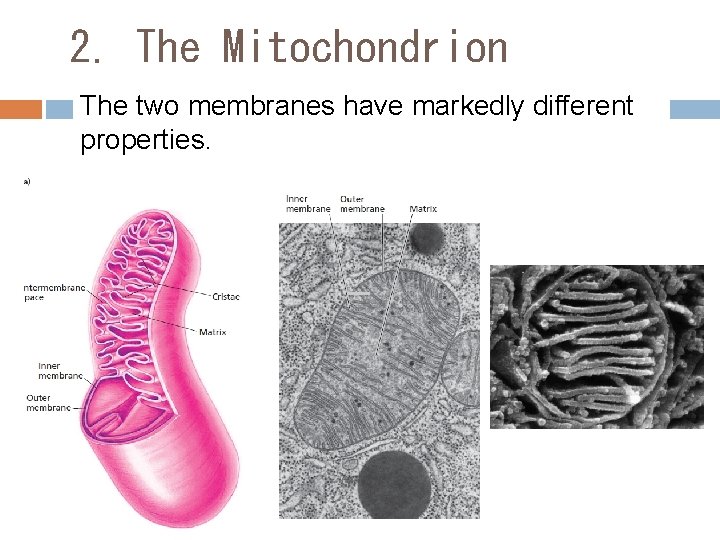
2. The Mitochondrion The two membranes have markedly different properties.

Alligator jaw muscles. You’re probably safe after this alligator has already snapped at you several times and missed. (If you trust your biochemistry textbook. )

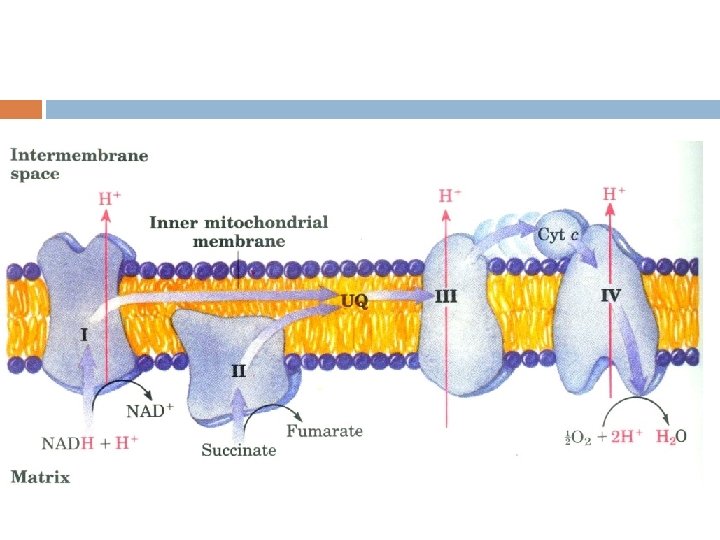
3. The Electron Transport Chain (ETC) ——Respiratory chain A series of membrane-associated electron carriers that pass electrons from reduced coenzymes (NADH and FADH 2) to molecular oxygen so as to recover free energy for the synthesis of ATP.

The Electron Transport Chain (ETC) ETC is consisted of 5 oligomeric proteins and 2 mobile electron carriers. The 5 complexes are: 1. ComplexⅠ: NADH-Q Reductase 2. ComplexⅡ: Succinate –Q Reductase 3. ComplexⅢ: Cytochrome c Reductase 4. ComplexⅣ: Cytochrome c oxidase 5. ATP Synthase The two mobile electron carriers are: Co. Q and Cytochrome c
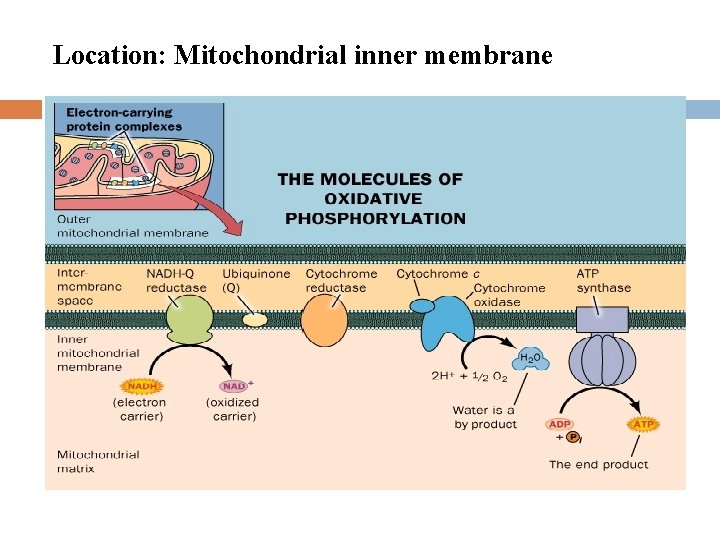
Location: Mitochondrial inner membrane
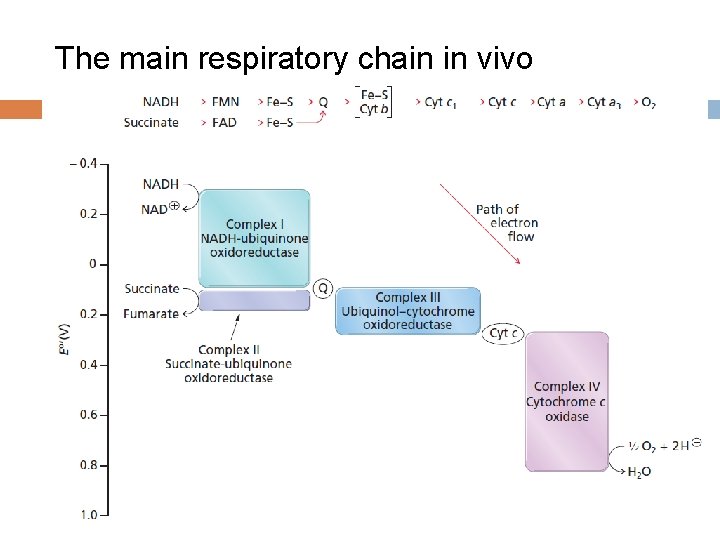
The main respiratory chain in vivo
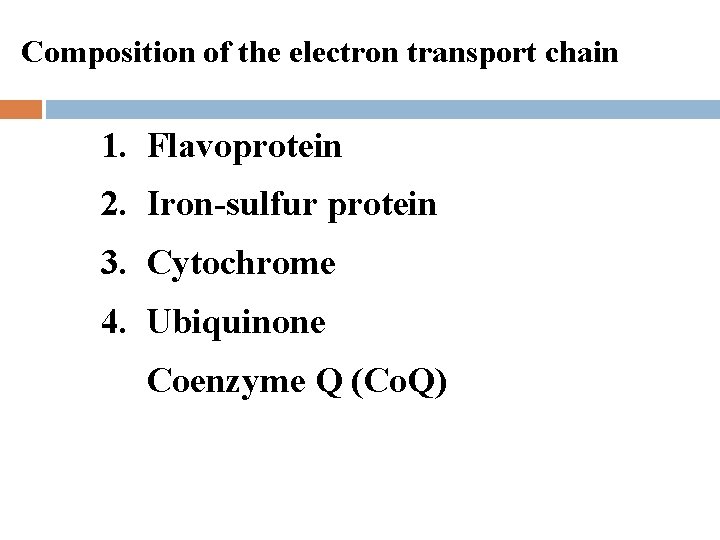
Composition of the electron transport chain 1. Flavoprotein 2. Iron-sulfur protein 3. Cytochrome 4. Ubiquinone Coenzyme Q (Co. Q)
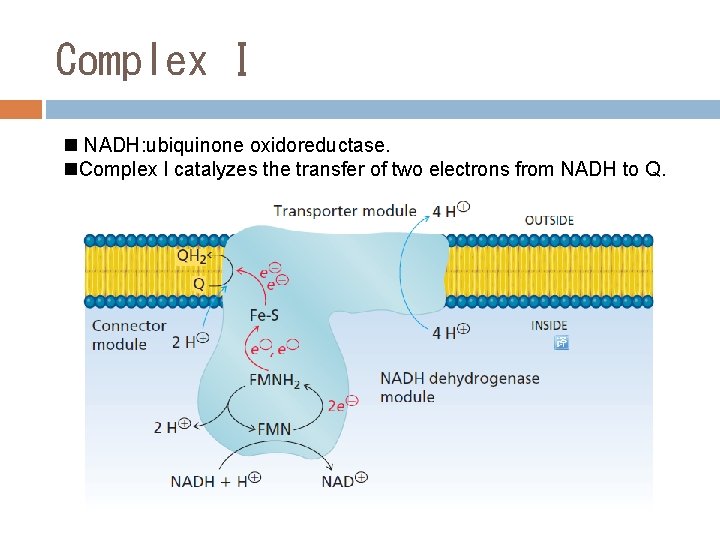
Complex I n NADH: ubiquinone oxidoreductase. n. Complex I catalyzes the transfer of two electrons from NADH to Q.
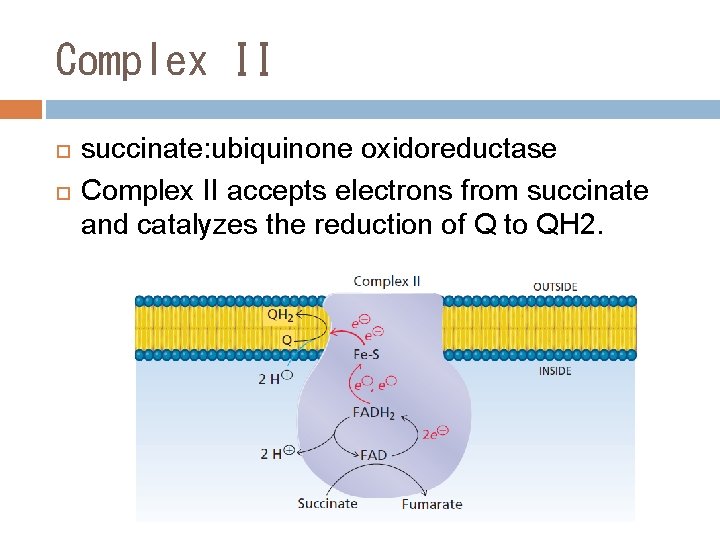
Complex II succinate: ubiquinone oxidoreductase Complex II accepts electrons from succinate and catalyzes the reduction of Q to QH 2.
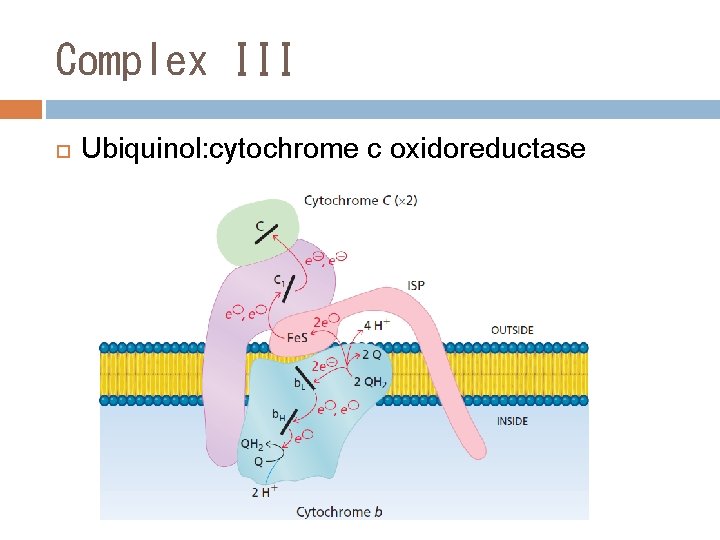
Complex III Ubiquinol: cytochrome c oxidoreductase
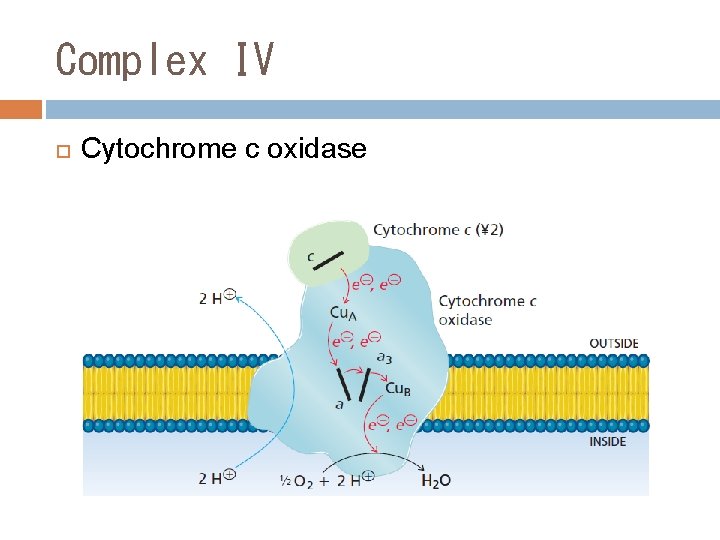
Complex IV Cytochrome c oxidase
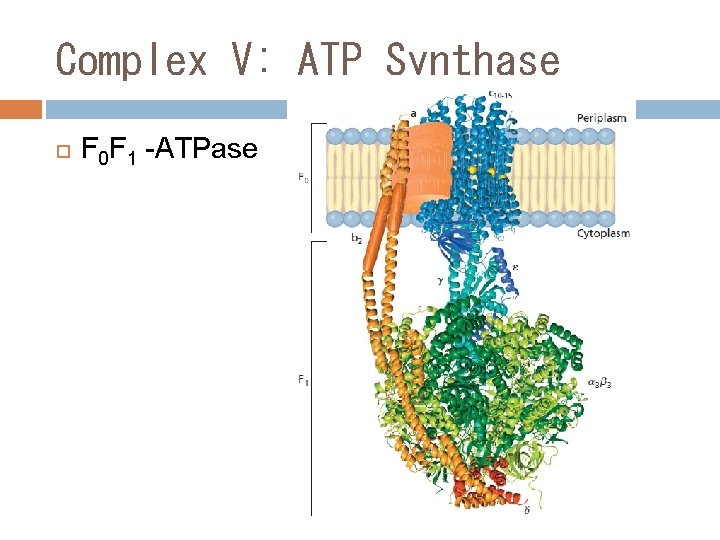
Complex V: ATP Synthase F 0 F 1 -ATPase

4. Inhibitors of electron transfer Common inhibitor: Rotenone, Amytal, Antimycin A CN-,H 2 S,N 3,CO.
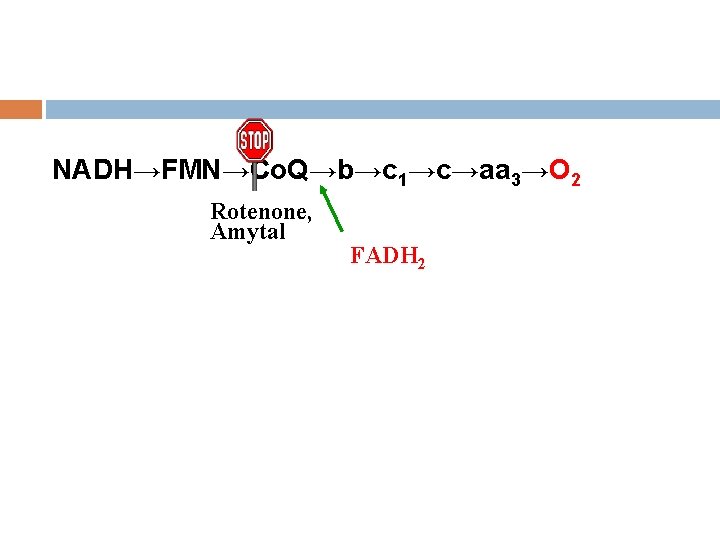
NADH→FMN→Co. Q→b→c 1→c→aa 3→O 2 Rotenone, Amytal FADH 2
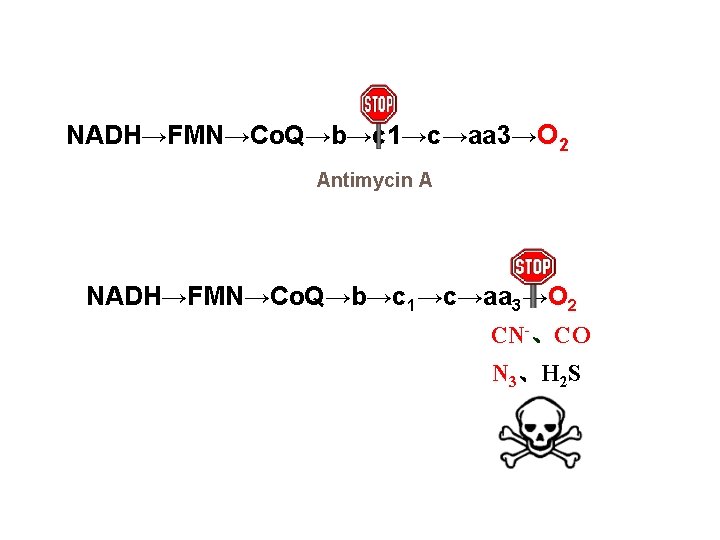
NADH→FMN→Co. Q→b→c 1→c→aa 3→O 2 Antimycin A NADH→FMN→Co. Q→b→c 1→c→aa 3→O 2 CN-、CO N 3、H 2 S

5. ATP Synthesis in Bio-oxidation High-energy compound ——Undergoes hydrolysis with a high negative standard free energy change. Commom compound, △G°=-8~ -20 KJ/mol High-energy compound △G°=-30~ -60 KJ/mol

l Types of high-energy compounds Phosphorus-oxygen bond (-O~P) Nitrogen and phosphorus bond (-N~P) Thioester bond Met-bond
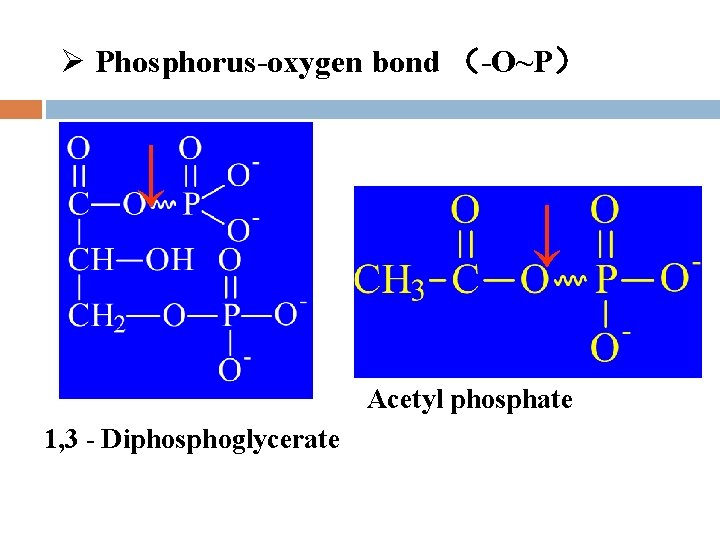
Ø Phosphorus-oxygen bond (-O~P) ↓ ↓ Acetyl phosphate 1, 3 - Diphosphoglycerate
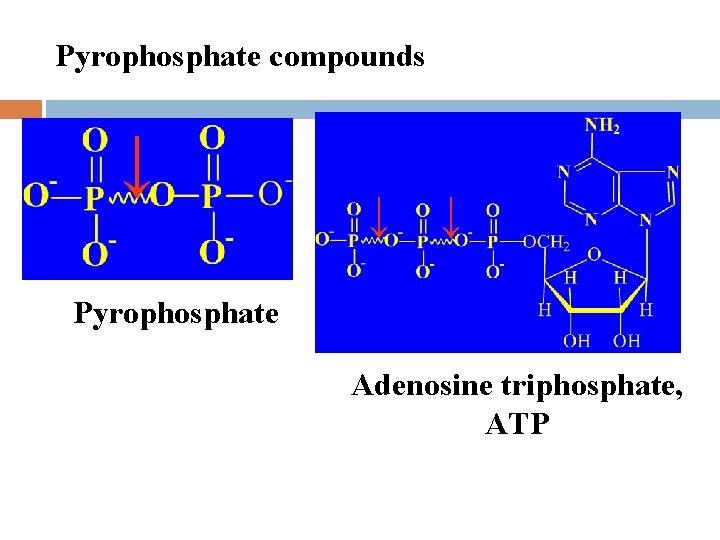
Pyrophosphate compounds ↓ ↓ ↓ Pyrophosphate Adenosine triphosphate, ATP
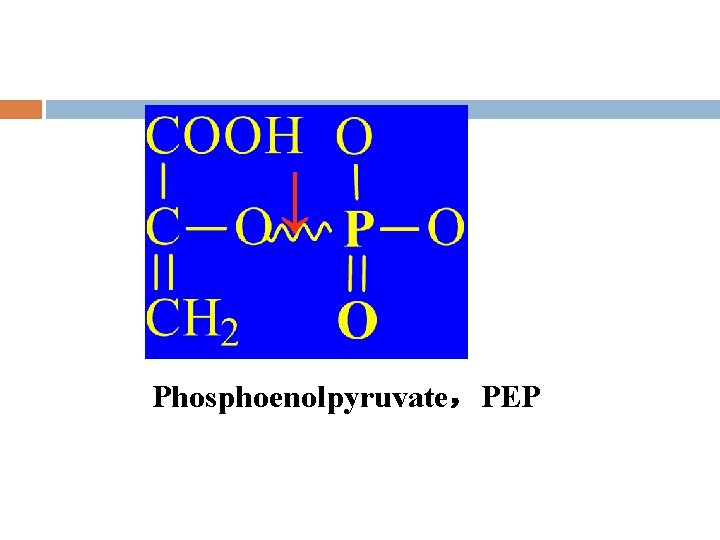
↓ Phosphoenolpyruvate,PEP
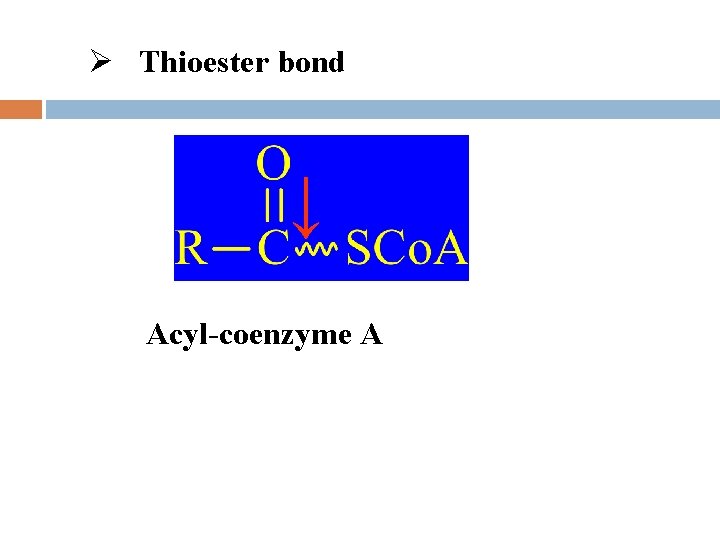
Ø Thioester bond ↓ Acyl-coenzyme A

ATP( Adenosine Triphosphate) works as “Energy Currency” in biosystems
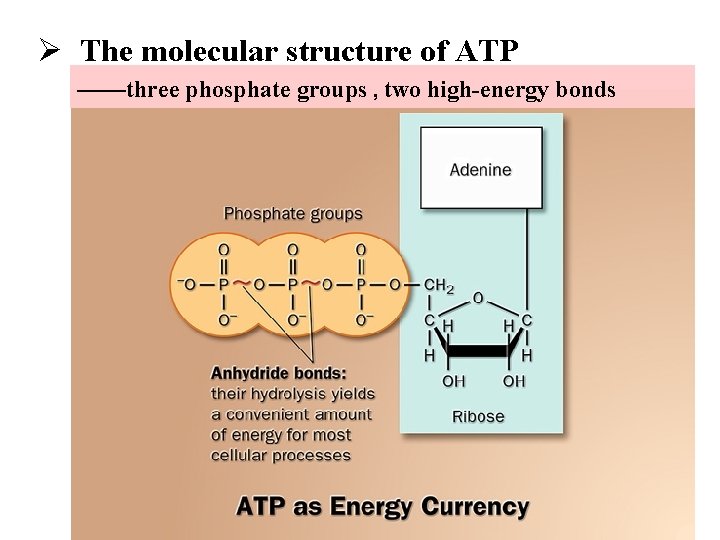
Ø The molecular structure of ATP ——three phosphate groups , two high-energy bonds

Ø Roles in energy exchange : 1. ATP is an immediate energy carrier. 2. ATP acts as a “ transfer station”during the transfer of phosphate group in enzyme-catalzed reactions

Synthesis of ATP ? Ø Respiratory —— oxidative phosphorylation Ø Photosynthesis —— Photophosphorylation

LECTURE 2 OXIDATIVE PHOSPHORYLATION

1. Oxidative phosphorylation The phosphorylation of ADP to ATP that accompanies the oxidation of a metabolite through the operation of the respiratory chain.
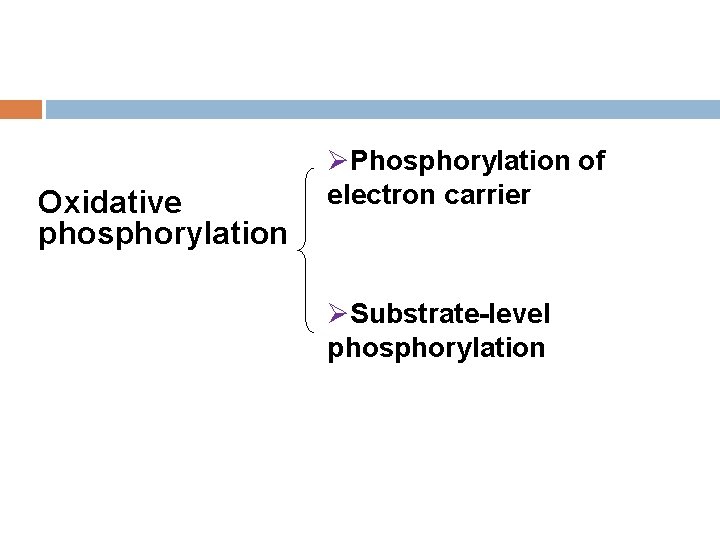
Oxidative phosphorylation ØPhosphorylation of electron carrier ØSubstrate-level phosphorylation

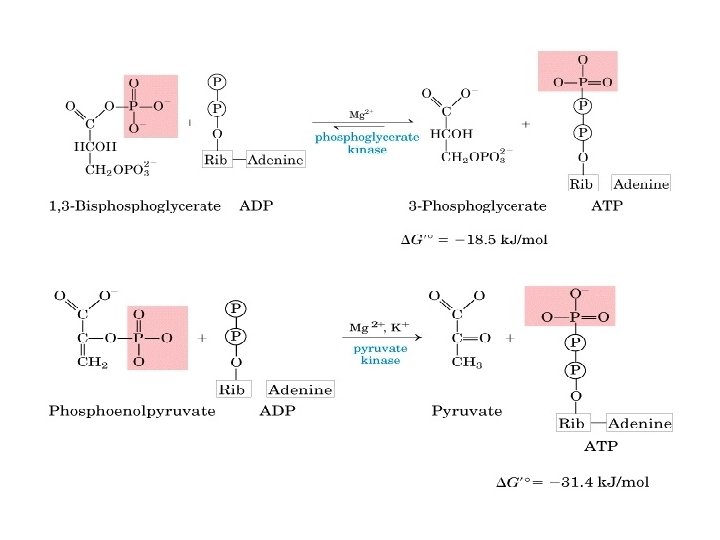
Ø Substrate-level phosphorylation —ATP is generated by transfer of the high-energy phosphate intermediate on a reactive intermediate to ADP via a kinase • S → M~P+ADP → MH+ATP

Ø Phosphorylation of electron carrier —Oxidative phosphorylation occurs when NADH and FADH 2 are oxidized by electron transport through the respiratory chain.

As discussed earlier, electrons entering from NADH produce 2. 5 ATPs per pair of electrons, from FADH 2 produce 1. 5 ATPs per pair of electrons.

The coupling mechanism of oxidation phosphorylation l Chemical coupling hypothesis l Conformational coupling hypothesis l Chemiosmotic hypothesis
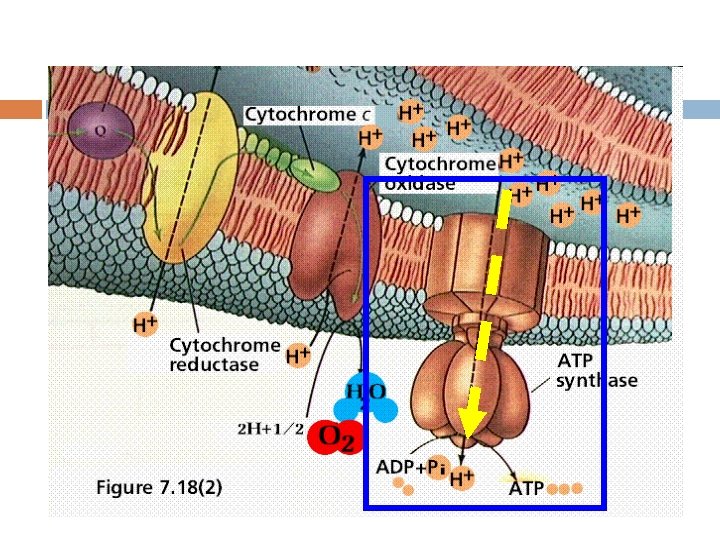

Uncoupling the role and uncoupler Uncoupling —— Affects ATP synthesis, but in a manner that does not involve direct binding to any of the proteins of the electron transport chain or the F 1 -F 0–ATPase. 2, 4 - dinitrophenol (DNP)

— Brown adipose tissue

Oxidative phosphorylation of Cytosolic NADH Ø Glycerol-3 -phospate shuttle Ø Malate-aspartate shuttle
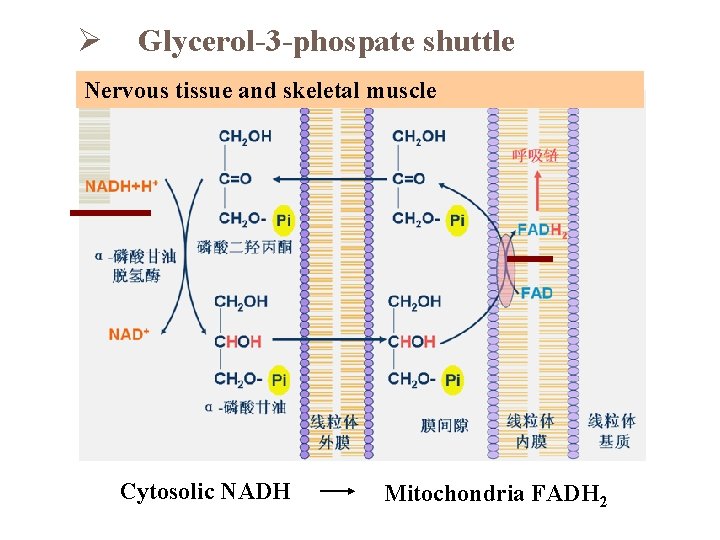
Ø Glycerol-3 -phospate shuttle Nervous tissue and skeletal muscle Cytosolic NADH Mitochondria FADH 2
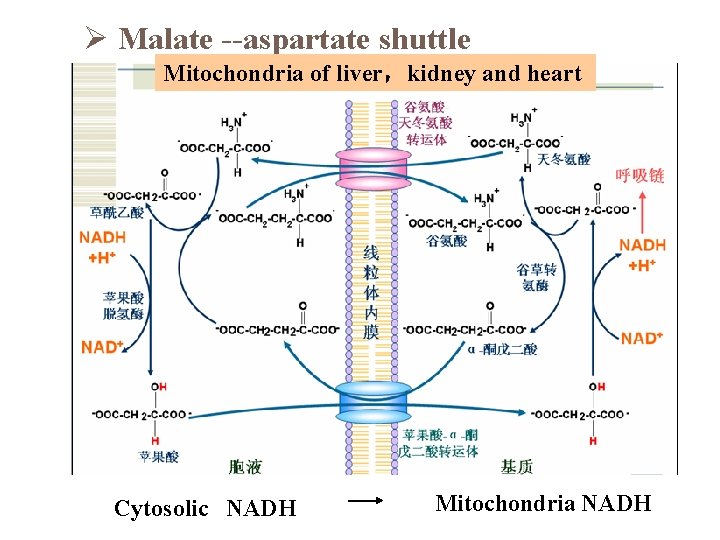
Ø Malate --aspartate shuttle Mitochondria of liver,kidney and heart Cytosolic NADH Mitochondria NADH
- Slides: 50