Chapter 9 Trace Elements Please bring to the
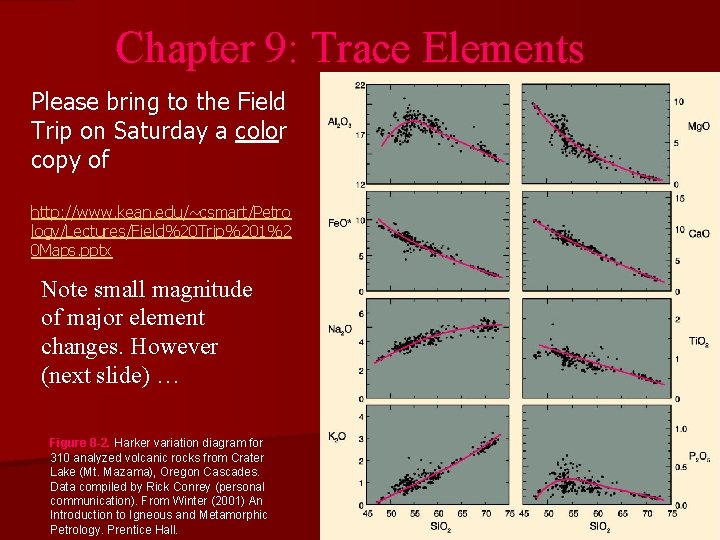
Chapter 9: Trace Elements Please bring to the Field Trip on Saturday a color copy of http: //www. kean. edu/~csmart/Petro logy/Lectures/Field%20 Trip%201%2 0 Maps. pptx Note small magnitude of major element changes. However (next slide) … Figure 8 -2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick Conrey (personal communication). From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
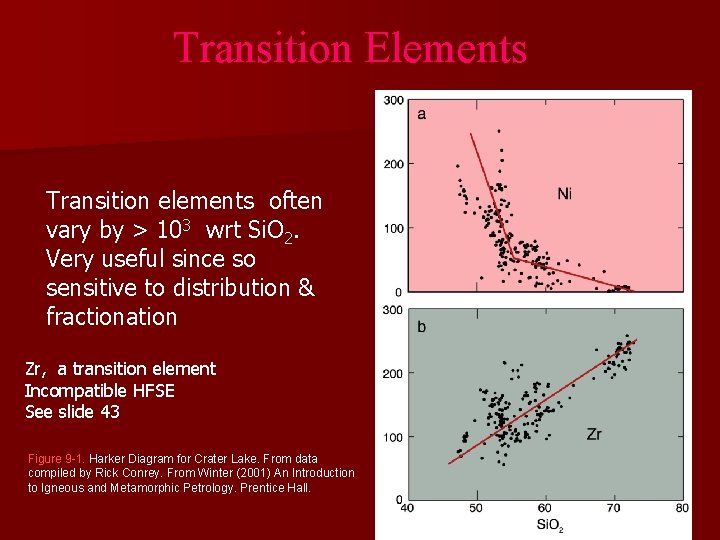
Transition Elements Transition elements often vary by > 103 wrt Si. O 2. Very useful since so sensitive to distribution & fractionation Zr, a transition element Incompatible HFSE See slide 43 Figure 9 -1. Harker Diagram for Crater Lake. From data compiled by Rick Conrey. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

Goldschmidt’s rules (simplistic, but useful) 1. 2 ions with the same valence and radius should exchange easily and enter a solid solution in amounts equal to their overall proportions How does Rb+ behave? As K+, concentrated in K-spars, micas, and evolved melts Ni++ behaves as Mg++, concentrates in Olivine TAKE OUT YOUR COORDINATION NUMBERS AND IONIC RADII CHART
![Rb+ [6] 1. 57 Å follows K+ [6] 1. 46 Å & conc. in Rb+ [6] 1. 57 Å follows K+ [6] 1. 46 Å & conc. in](http://slidetodoc.com/presentation_image/5202012d1e936af9d8843a5448ecebbe/image-4.jpg)
Rb+ [6] 1. 57 Å follows K+ [6] 1. 46 Å & conc. in K-Spar, mica, & late melt. Ni++ [6] 0. 77 Å follows Mg++ [6] 0. 8 Å & conc in Mg-Olivine “Forsterite”
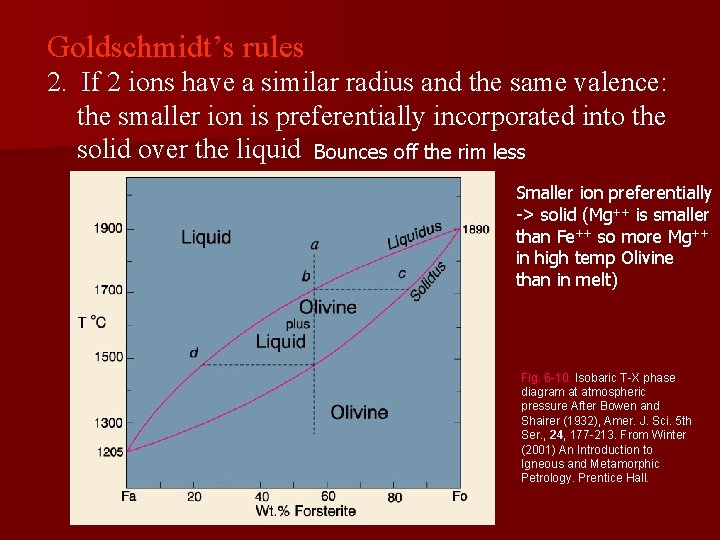
Goldschmidt’s rules 2. If 2 ions have a similar radius and the same valence: the smaller ion is preferentially incorporated into the solid over the liquid Bounces off the rim less Smaller ion preferentially -> solid (Mg++ is smaller than Fe++ so more Mg++ in high temp Olivine than in melt) Fig. 6 -10. Isobaric T-X phase diagram at atmospheric pressure After Bowen and Shairer (1932), Amer. J. Sci. 5 th Ser. , 24, 177 -213. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

1 Ångström = 1. 0 × 10 -10 meters 3. If 2 ions have a similar radius, but different valence: the ion with the higher charge is preferentially incorporated into the solid over the liquid Ti+4 Rutile Ti. O 2 versus Hematite Fe 2 O 3 Example [6] r=0. 69 Angstrom Other ions this size Fe +3 r=0. 68 A Mn +3 [6] r=. 70 A Ti+4 is always preferred in solids over liquids. Example Cr+3 [6] r=0. 76 Other ions this size Ni +2 [6] r=0. 77 A Fe +2 [6] r=. 77 Cr+3 is always preferred in solids over liquids.

Anorthite higher T than Albite Forsterite higher T than Fayalite Chemical Fractionation l l l The uneven distribution of an ion between two competing (equilibrium) phases Example: the ratio of Ca++/Na+ is always greater in plagioclase crystals than in the coexisting melt. Ca++ higher valance than Na+ , goes into Plag xtals. first. If melting Plag, Na+ goes to melt first. Example: the ratio of Mg++/Fe++ is always greater in Olivine than in the coexisting melt.

If the reaction between solid and liquid phases is a phase change of some component i i (liquid) i (solid) A distribution constant KD is Recall that we used the lever principle to estimate this ratio from our phase diagrams Where Xi is the mole fraction of component i in some phase. KD the ratio of solubility of component i in these two phases.

Different units As long as the concentrations of the components are dilute, call KD the partition coefficient Where C is the concentration of a trace element in the phase indicated, solid and liquid, in ppm or wt% Table 9. 1 (three slides below this) gives partition coefficients for commonly used trace elements in minerals precipitating from basaltic or andesitic melts.

Trace element activity varies in direct relation to their concentration in the system. n Thus if for Nickel XNi in the system doubles the XNi in all phases will double – This does not mean that XNi in all phases is the same, since trace elements do fractionate. Rather the XNi within each phase will vary in proportion to the system concentration n For example: suppose C(Ni) = 20 ppm in a system C(Ni) in olivine may be 100 ppm C(Ni) in plagioclase may be 1 ppm C(Ni) in liquid may be 10 ppm Double C(Ni) in system to 40 ppm: Ol -> 200 ppm, Plag -> 2 ppm and liquid -> 20 ppm

n Incompatible melt elements concentrate in the KD « 1 much less than 1 Lesson: incompatibles don’t stick on crystal faces unless the Temps are low. Melt a xtal, incompatibles go to the melt. n Compatible solid elements concentrate in the KD» 1 much more than 1
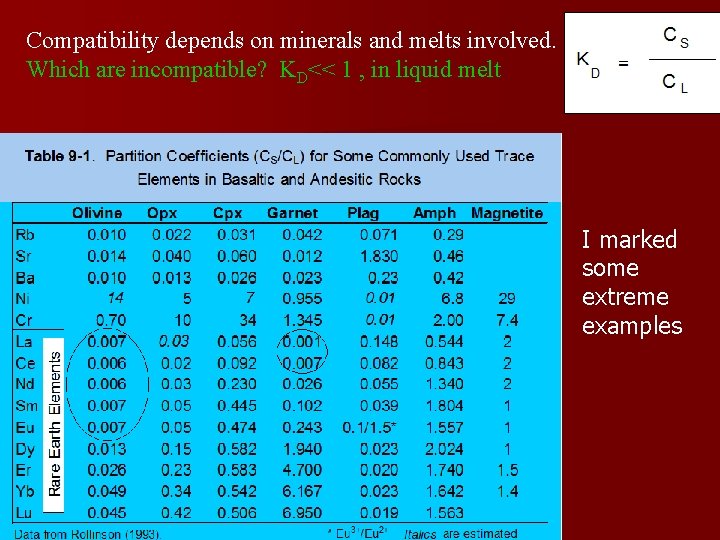
Compatibility depends on minerals and melts involved. Which are incompatible? KD<< 1 , in liquid melt I marked some extreme examples

n For a rock, determine the bulk distribution coefficient D for an element by calculating the contribution for each mineral eq. 9 -4: D i = W A Di A A WA = weight % of mineral A in the rock Di = partition coefficient of element i in A mineral A

Garnet Lherzolite Mantle Xenolith (Garnet Lherzolite) Kimberley, SOUTH AFRICA Photographed by Tony Peterson
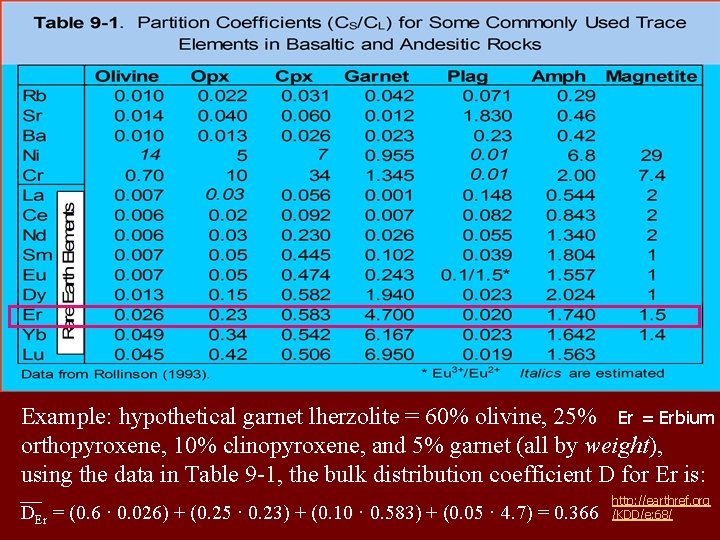
Example: hypothetical garnet lherzolite = 60% olivine, 25% Er = Erbium orthopyroxene, 10% clinopyroxene, and 5% garnet (all by weight), using the data in Table 9 -1, the bulk distribution coefficient D for Er is: DEr = (0. 6 · 0. 026) + (0. 25 · 0. 23) + (0. 10 · 0. 583) + (0. 05 · 4. 7) = 0. 366 http: //earthref. org /KDD/e: 68/
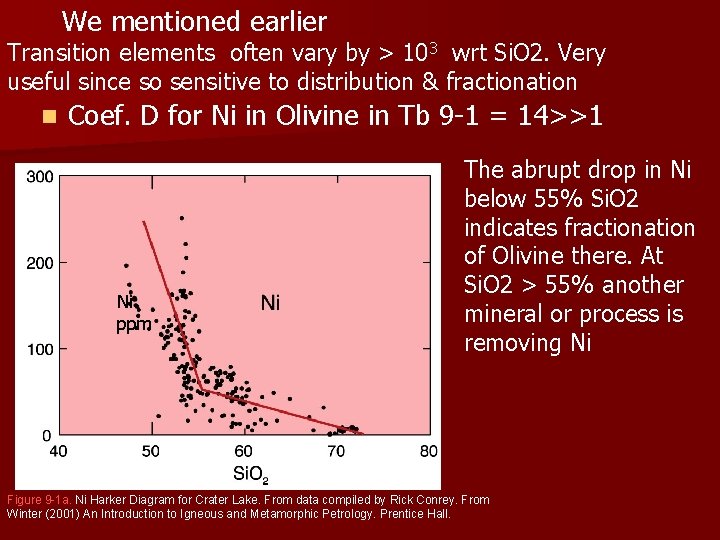
We mentioned earlier Transition elements often vary by > 103 wrt Si. O 2. Very useful since so sensitive to distribution & fractionation n Coef. D for Ni in Olivine in Tb 9 -1 = 14>>1 Ni ppm The abrupt drop in Ni below 55% Si. O 2 indicates fractionation of Olivine there. At Si. O 2 > 55% another mineral or process is removing Ni Figure 9 -1 a. Ni Harker Diagram for Crater Lake. From data compiled by Rick Conrey. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
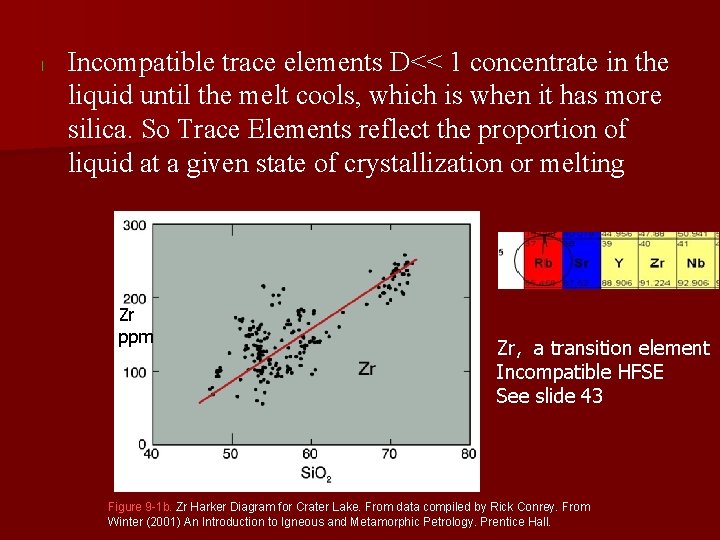
l Incompatible trace elements D<< 1 concentrate in the liquid until the melt cools, which is when it has more silica. So Trace Elements reflect the proportion of liquid at a given state of crystallization or melting Zr ppm Zr, a transition element Incompatible HFSE See slide 43 Figure 9 -1 b. Zr Harker Diagram for Crater Lake. From data compiled by Rick Conrey. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

Trace element concentrations are in the Henry’s Law* region of concentration, so their activity varies in direct relation to their concentration in the system Thus if XNi in the system doubles the XNi in all phases will double Because of this, the ratios of trace elements are often superior to the concentration of a single element in identifying the role of a specific mineral * "At a constant temperature, the amount of a given gas that dissolves in a given type and volume of liquid is directly proportional to the partial pressure of that gas in equilibrium with that liquid. "

n K/Rb often used to measure the importance of amphibole in a source rock – K & Rb behave very similarly, so K/Rb should be ~ constant – If amphibole important, almost all K and Rb reside in it – Amphibole has a D of about 1. 0 for K and 0. 3 for Rb

n Sr and Ba (also incompatible elements) § Sr is excluded from most common minerals except plagioclase § Ba similarly excluded except in alkali feldspar (Sanidine, Orthoclase, Microcline) § ratio Ba/Sr increases w plagioclase, decreases when orthoclase xtals present
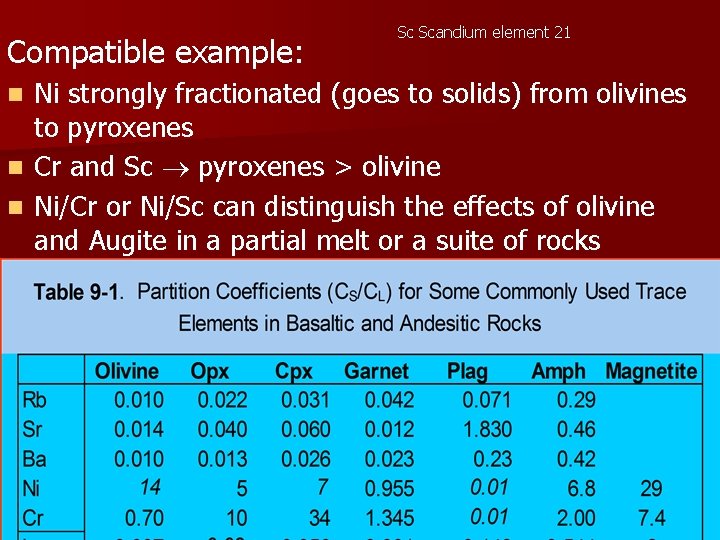
Compatible example: Sc Scandium element 21 Ni strongly fractionated (goes to solids) from olivines to pyroxenes n Cr and Sc pyroxenes > olivine n Ni/Cr or Ni/Sc can distinguish the effects of olivine and Augite in a partial melt or a suite of rocks produced by fractional crystallization n

Models of Magma Evolution l Batch Melting F The melt remains in equilibrium with the solid until at some point it floats upward, separating from the solids

Models of Magma Evolution n Batch Melting (Shaw) 1 eq. 9 -5 C L = C O Di(1 - F)+ F CL = trace element concentration in the liquid CO = trace element concentration in the original rock before melting began F = wt fraction of melt produced = melt/(melt + rock) Di = bulk distribution coefficient for element i
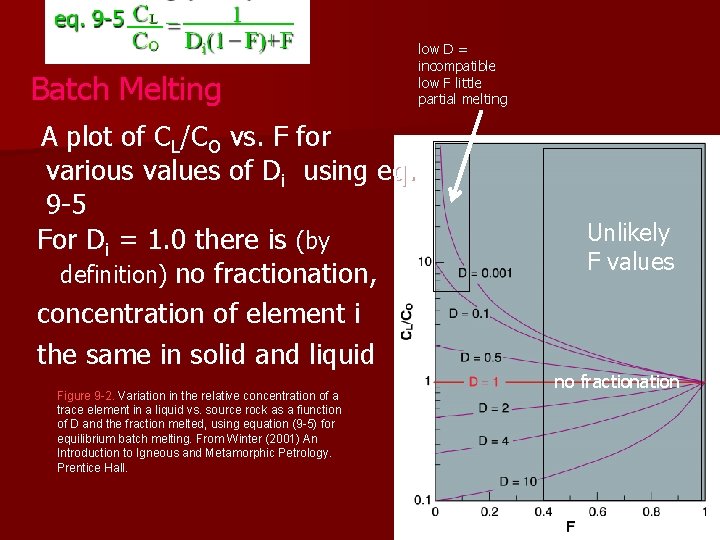
Batch Melting A plot of CL/CO vs. F for various values of Di using eq. 9 -5 For Di = 1. 0 there is (by definition) no fractionation, concentration of element i the same in solid and liquid Figure 9 -2. Variation in the relative concentration of a trace element in a liquid vs. source rock as a fiunction of D and the fraction melted, using equation (9 -5) for equilibrium batch melting. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall. low D = incompatible low F little partial melting Unlikely F values no fractionation
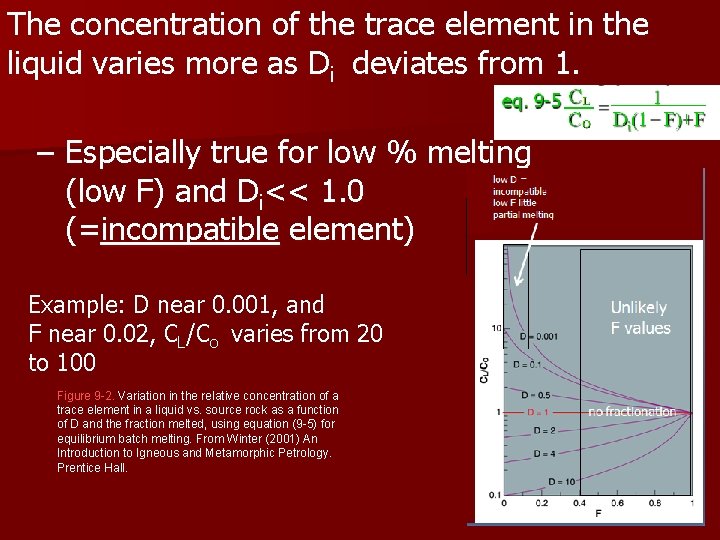
The concentration of the trace element in the liquid varies more as Di deviates from 1. – Especially true for low % melting (low F) and Di<< 1. 0 (=incompatible element) Example: D near 0. 001, and F near 0. 02, CL/Co varies from 20 to 100 Figure 9 -2. Variation in the relative concentration of a trace element in a liquid vs. source rock as a function of D and the fraction melted, using equation (9 -5) for equilibrium batch melting. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
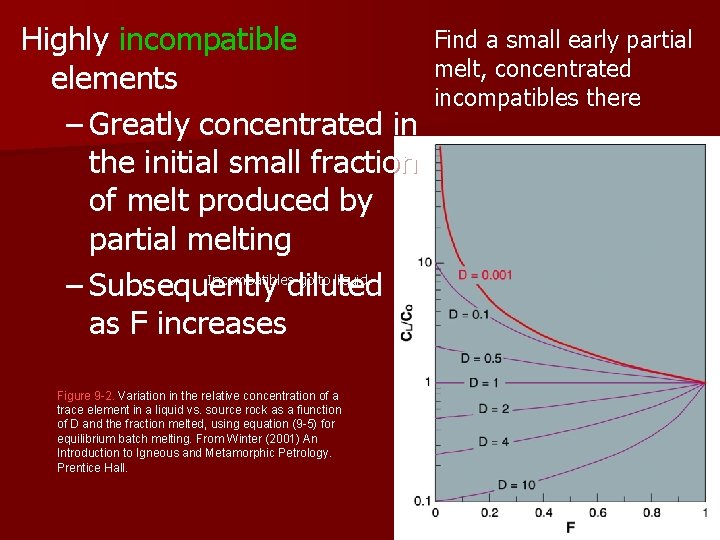
Highly incompatible elements – Greatly concentrated in the initial small fraction of melt produced by partial melting Incompatibles go to liquid – Subsequently diluted as F increases Figure 9 -2. Variation in the relative concentration of a trace element in a liquid vs. source rock as a fiunction of D and the fraction melted, using equation (9 -5) for equilibrium batch melting. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall. Find a small early partial melt, concentrated incompatibles there
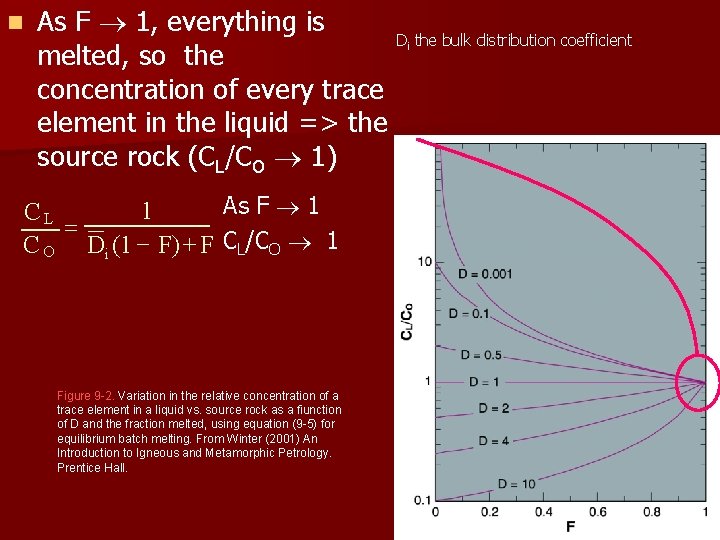
n As F 1, everything is D the bulk distribution coefficient melted, so the concentration of every trace element in the liquid => the source rock (CL/CO 1) i As F 1 1 CL = C O Di (1 - F) + F CL/CO 1 Figure 9 -2. Variation in the relative concentration of a trace element in a liquid vs. source rock as a fiunction of D and the fraction melted, using equation (9 -5) for equilibrium batch melting. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
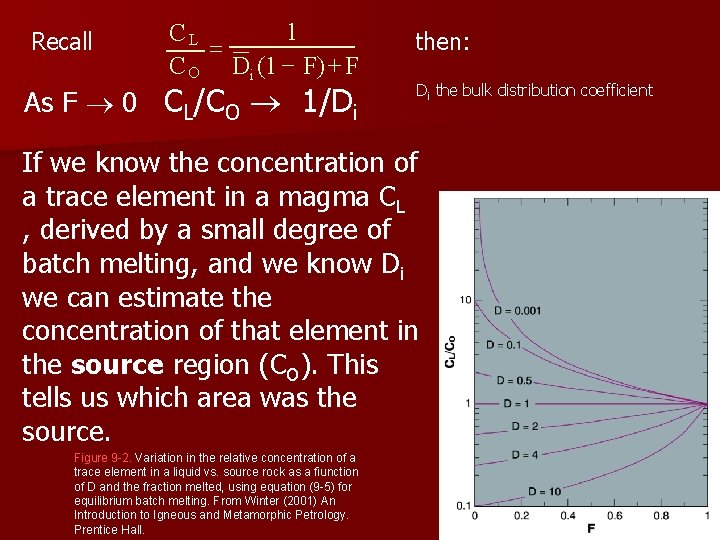
Recall 1 CL = C O Di (1 - F) + F As F 0 CL/CO 1/Di then: Di the bulk distribution coefficient If we know the concentration of a trace element in a magma CL , derived by a small degree of batch melting, and we know Di we can estimate the concentration of that element in the source region (CO). This tells us which area was the source. Figure 9 -2. Variation in the relative concentration of a trace element in a liquid vs. source rock as a fiunction of D and the fraction melted, using equation (9 -5) for equilibrium batch melting. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

Di the bulk distribution coefficient l For very incompatible elements as Di 0 equation 9 -5 1 CL = C O Di (1 - F) + F reduces to: CL 1 eq. 9 -7 = CO F If we know the concentration of a very incompatible element in both a magma and the source rock, we can determine the fraction of partial melt produced

Worked Example of Batch Melting: Rb and Sr Basalt with the “mode” = Volume% [cm 3]: Table 9 -2. Conversion from mode to 15 cm 3 x 3. 6 g/ cm 3 = 54 g weight percent Mineral Mode Density Wt prop ol 15 3. 6 cpx 33 3. 4 112. 2 0. 37 plag 51 2. 7 137. 7 0. 45 303. 9 1. 00 Sum 54 Wt% 54 g/303. 9 g ~ 0. 18 so Wol ~ 0. 18 1. Convert to weight % minerals (Wol Wcpx etc. ) Done, see rightmost column above.

A Basalt (ol, cpx, plag) with the mode: Worked Example of Batch Melting: Rb and Sr Table 9 -2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3. 6 54 0. 18 cpx 33 3. 4 112. 2 0. 37 plag 51 2. 7 137. 7 0. 45 Sum 303. 9 1. 00 1. Convert to weight % minerals (Wol Wcpx Wplag) (previous slide) 2. Use equation eq. 9 -4: D i = W A Di and the table of D (Table 9. 1) values for Rb and Sr in each mineral to calculate the bulk distribution coefficients: DRb = 0. 01 x 0. 18 + 0. 031 x 0. 37 + 0. 071 x 0. 45 = 0. 045 DRb = 0. 045 (Rb incompatible) and DSr = 0. 848 (close to 1)

3. Use the batch melting equation to calculate CL/CO for various values of F For F =0. 05 and for Rb: CL/Co = 1/ ((0. 045(1 -0. 05)) +0. 05) = 10. 78 Similarly for F = 0. 05 and Sr CL/Co = 1. 18 Then the ratio Rb/Sr = 10. 78/1. 18 = 9. 12 Continue with remaining F From Winter (2010) 2 nd edition An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
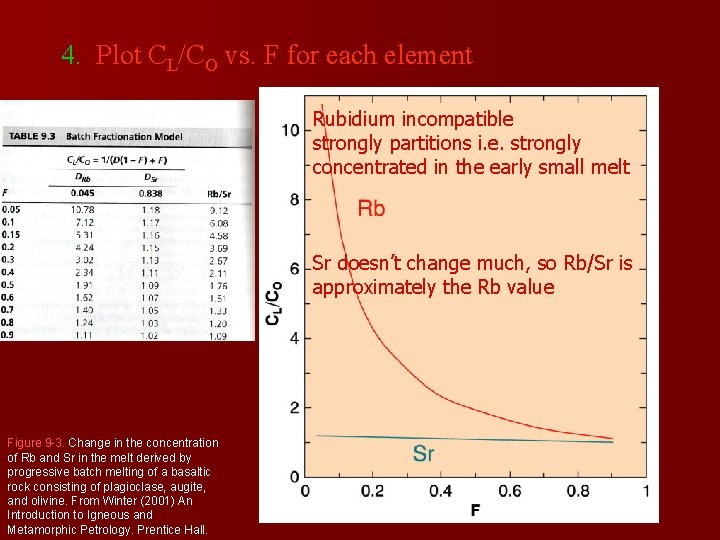
4. Plot CL/CO vs. F for each element Rubidium incompatible strongly partitions i. e. strongly concentrated in the early small melt Sr doesn’t change much, so Rb/Sr is approximately the Rb value Figure 9 -3. Change in the concentration of Rb and Sr in the melt derived by progressive batch melting of a basaltic rock consisting of plagioclase, augite, and olivine. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

Discussion n Any ratio of Incompatible to Compatible elements will be sensitive to the degree of partial melting in initial stages n Our experience with Ternary systems tells us it is unrealistic to expect a ratio of the minerals in the solid residues to remain constant. n What should we do?

Incremental Batch Melting n Calculate batch melting for successive batches (same equation) n Must recalculate Di as solids change and minerals are selectively melted n Figure 9 -2 and 9 -3 (earlier slides) show that the model we just used is most sensitive to Di at low values of F, so it is most important to use small increments in that area. Once F > 0. 4, batch melts vary less and are less likely anyway.

Crystallization Extremes Either: Crystals remain in equilibrium with each melt increment OR The other extreme: separation of each crystal as it formed = perfectly continuous fractional crystallization in a magma chamber

n Rayleigh fractional crystallization The second extreme: separation of each crystal as it formed = perfectly continuous fractional crystallization in a magma chamber Crystals accumulate on floor of magma chamber, isolating formed crystals – Concentration of some element in the residual liquid, CL is modeled by the Rayleigh equation: eq. 9 -8 CL/CO = F (Di -1) Co conc. of element in original melt F fraction of melt remaining after xtals removed

Other models are used to analyze l Mixing of magmas l Wall-rock assimilation l Zone refining l Combinations of processes End of Part 1
The Rare Earth Elements (REE) Begins Part 2

REEs n Group IIIA, +3 oxidation state, ionic radius decreases with increasing atomic number (the “Lanthanide contraction”) so right side smaller ions, fit more readily, more compatible. bigger smaller

Density Groups of REEs LREE = light rare earth elements (Sc, La, Ce, Pr, Nd, Pm, Sm, Eu, and Gd; also known as the cerium group) n HREE = heavy rare earth elements (Y, Tb, Dy, Ho, Er, Tm, Yb, and Lu; also known as the yttrium group) n n The densities of the LREEs (as pure elements) range from 2. 989 (scandium) to 7. 9 g/cc (gadolinium), while those of the HREEs are from 8. 2 to 9. 8, except for yttrium (4. 47) and ytterbium (between 6. 9 and 7) The latter are groups with the HREE due to similar geological behavior.

Oxygen Fugacity is the partial pressure of a gas phase which takes into account that the gas is able to chemically react with other components in the system. A measure of departure from Ideal Gas behavior. n Oxygen fugacity is the measure of the availability of oxygen (within a given system) to partake in chemical reactions. It represents the chemical potential of oxygen. It is also a way to quantify the redox state of a given system. n This is very important in terms of determining the stable mineral present in a given geological system. More oxidizing conditions, in igneous or hydrothermal systems for example, favor the crystallization (and stability) of the more oxidized mineral phases. More available oxygen in the system = more oxidizing n System precipitates components with more oxygen (i. e. Hematite Fe 2 O 3 (ratio 3/2 = 1. 5) would crystallize, rather than Magnetite Fe 3 O 4 (ratio 4/3 = 1. 333).

n n Incompatible elements An element unsuitable in size and/or charge to the cation sites of the rock forming minerals. The partition coefficient for them between rock-forming minerals and melt is much smaller than 1. During the partial melting of the Earth's mantle and crust, elements that have difficulty in entering cation sites of the basaltic minerals (Olivine, Clinopyroxenes, Carich Plagioclases) are concentrated in the liquid phase of the magma. They are destined for later minerals and glasses. For example, Potassium K enters the K-spars: Sanidine, Orthoclase, Microcline very late in fractionation. n Two groups of incompatible elements are known by acronyms. One group includes elements having large ionic radius, (called LILE, or large-ion lithophile elements. This LILE group includes potassium, rubidium, cesium, strontium, and barium. n The other group includes elements of large ionic valences (or high charges), such as zirconium Zr+4 , niobium, hafnium, rare earth elements (REE), thorium, uranium and tantalum (called HFSE, or high field strength elements). These can occur in pegmatites, for example the alkaline pegmatites we will see at Cranberry Lake.

Europium n Note: at low Oxygen fugacity, Europium can have a +2 valence, and Eu+2 can be more abundant than Eu+3 Eu+2 is a Large Ion Lithophile (LIL) Eu+3 is a high field strength (HFS) element. LILs include K, Rb, Cs, Ba, Pb+2 , Sr and Eu+2 LIL’s are low field strength, e. g. Potassium’s ion is K +, and are generally more mobile if a fluid is adjacent.

n The term LILE is restricted to lithophile trace elements having a large ionic radius to charge ratio; that have ionic radii greater than those of Ca 2+ and Na 1+, some of the largest cations common to rock forming minerals. By this definition, LILEs are K, Rb, Cs, Sr, Ba, Pb and Eu+2
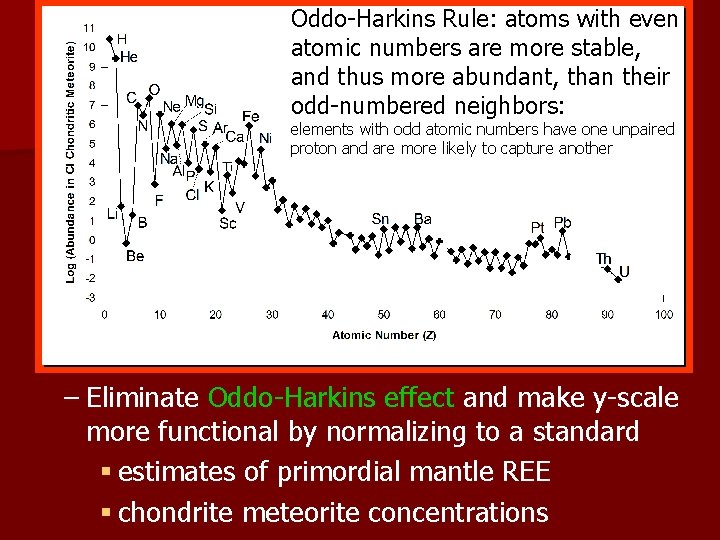
Oddo-Harkins Rule: atoms with even atomic numbers are more stable, and thus more abundant, than their odd-numbered neighbors: elements with odd atomic numbers have one unpaired proton and are more likely to capture another – Eliminate Oddo-Harkins effect and make y-scale more functional by normalizing to a standard § estimates of primordial mantle REE § chondrite meteorite concentrations
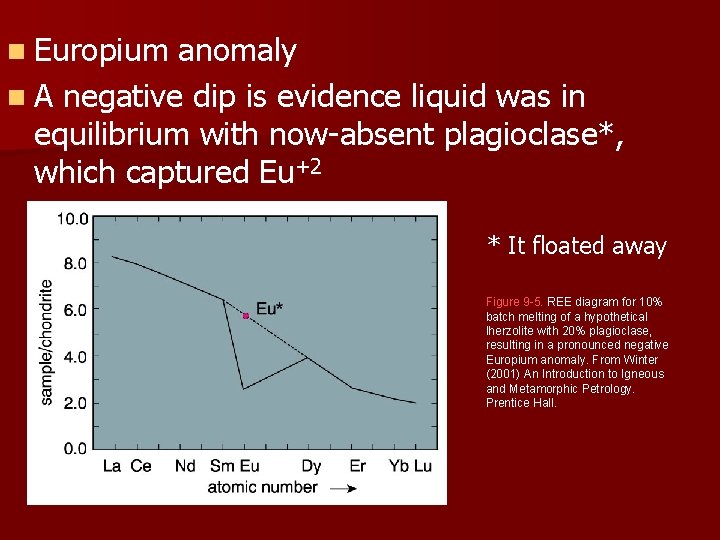
n Europium anomaly n A negative dip is evidence liquid was in equilibrium with now-absent plagioclase*, which captured Eu+2 * It floated away Figure 9 -5. REE diagram for 10% batch melting of a hypothetical lherzolite with 20% plagioclase, resulting in a pronounced negative Europium anomaly. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
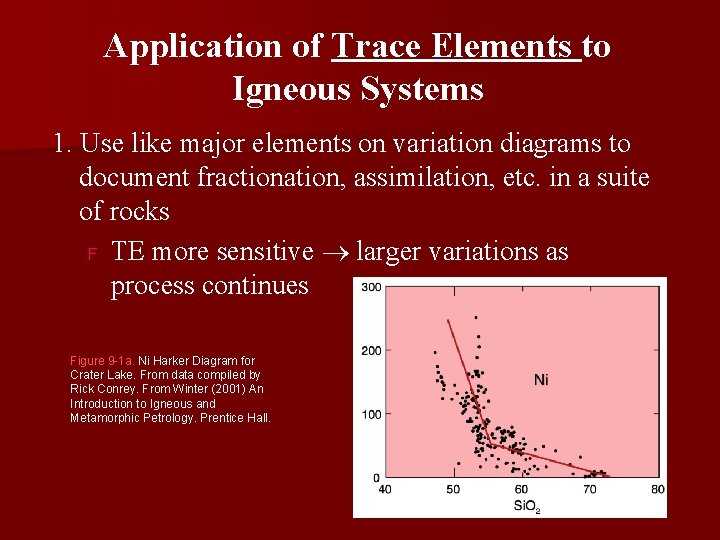
Application of Trace Elements to Igneous Systems 1. Use like major elements on variation diagrams to document fractionation, assimilation, etc. in a suite of rocks F TE more sensitive larger variations as process continues Figure 9 -1 a. Ni Harker Diagram for Crater Lake. From data compiled by Rick Conrey. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.

Application of Trace Elements to Igneous Systems 2. Identification of the source rock or a particular mineral involved in either partial melting or fractional crystallization processes Example: can use REE to distinguish between high pressure and low pressure sources of a mantlederived magma In the deep continental crust, and at depths over about 100 km in the mantle, garnet and clinopyroxene are important phases, which remain as residual solids during the generation of up to 15 -20% partial melting

Garnet concentrates the HREE and fractionates among them Thus if Garnet is in equilibrium with the partial melt (a residual phase in the source left behind) expect a concentration of Tb, Er, Yb, and Lu in the Garnet Shallow (< 40 km) partial melting of the mantle will have plagioclase in the residuum and a Eu anomaly will result

Table 9 -6 A brief summary of some particularly useful trace elements in igneous petrology Element Use as a petrogenetic indicator Ni, Co, Cr Highly compatible elements. Ni (and Co) are concentrated in olivine, and Cr in spinel and clinopyroxene. High concentrations indicate a mantle source. V, Ti Both show strong fractionation into Fe-Ti oxides (ilmenite or titanomagnetite). If they behave differently, Ti probably fractionates into an accessory phase, such as sphene or rutile. Zr, Hf Very incompatible elements that do not substitute into major silicate phases (although they may replace Ti in sphene or rutile). Ba, Rb Incompatible element that substitutes for K in K-feldspar, micas, or hornblende. Rb substitutes less readily in hornblende than K-spar and micas, such that the K/Ba ratio may distinguish these phases. Sr Substitutes for Ca in plagioclase (but not in pyroxene), and, to a lesser extent, for K in Kfeldspar. Behaves as a compatible element at low pressure where plagioclase forms early, but as an incompatible at higher pressure where plagioclase is no longer stable. REE Garnet accommodates the HREE more than the LREE, and orthopyroxene and hornblende do so to a lesser degree. Sphene and plagioclase accommodates more LREE. Eu 2+ is strongly partitioned into plagioclase. Y Commonly incompatible (like HREE). Strongly partitioned into garnet and amphibole. Sphene and apatite also concentrate Y, so the presence of these as accessories could have a significant effect. Table 9 -6. After Green (1980). Tectonophys. , 63, 367385. From Winter (2001) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall.
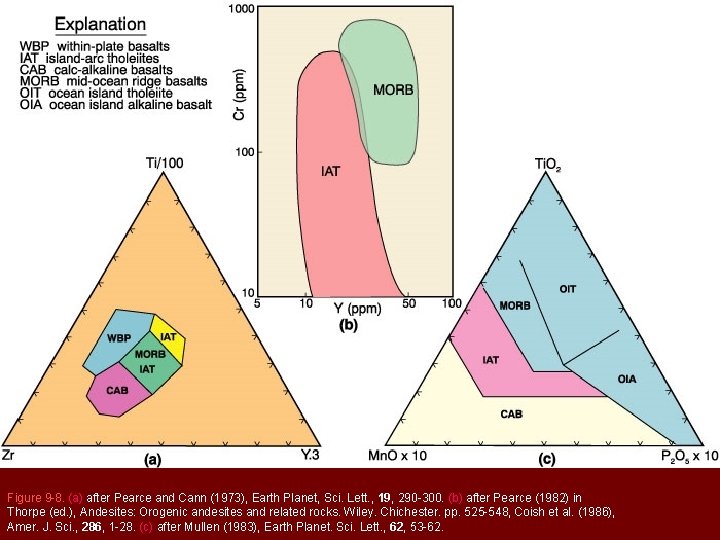
Figure 9 -8. (a) after Pearce and Cann (1973), Earth Planet, Sci. Lett. , 19, 290 -300. (b) after Pearce (1982) in Thorpe (ed. ), Andesites: Orogenic andesites and related rocks. Wiley. Chichester. pp. 525 -548, Coish et al. (1986), Amer. J. Sci. , 286, 1 -28. (c) after Mullen (1983), Earth Planet. Sci. Lett. , 62, 53 -62.
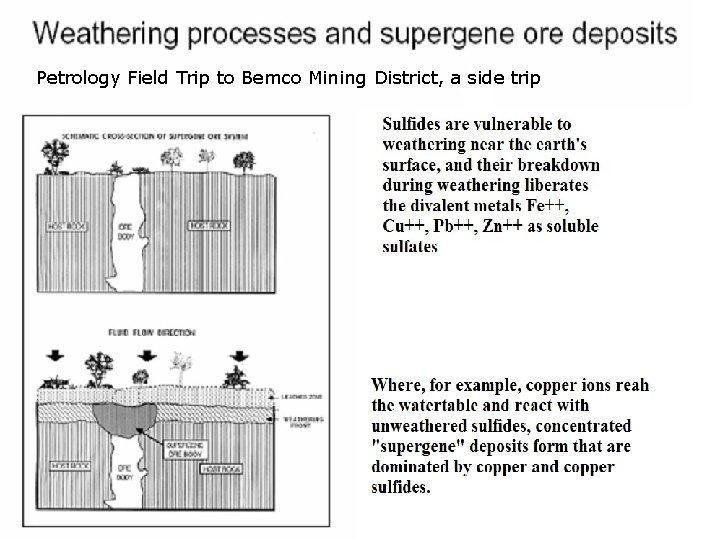
Petrology Field Trip to Bemco Mining District, a side trip

Ores from weathered Sulfide deposits • Mineral deposits containing sulfide minerals, e. g. copper sulfides that are subjected to weathering, can go into solution and trickle down to the reducing conditions below the water table, where native metals or rich concentrations of ores are precipitated. Black Smokers, hydrothermal circulations Gossan Intensely oxidized, weathered or decomposed rock, usually the upper and exposed part of an ore deposit or mineral vein. In the classic gossan or iron cap all that remains is iron oxides and quartz often in the form of boxworks, quartz lined cavities retaining the shape of the dissolved ore minerals .

Solubility in water The Solubility Rules 1. Salts containing Group I elements are soluble (Li+, Na+, K+, Cs+, Rb+). Exceptions to this rule are rare. Salts containing the ammonium ion (NH 4+) are also soluble. 2. Salts containing nitrate ion (NO 3 -) are generally soluble. 3. Salts containing Cl -, Br -, I - are generally soluble. Important exceptions to this rule are halide salts of Ag+, Pb 2+, and (Hg 2)2+. Thus, Ag. Cl, Pb. Br 2, and Hg 2 Cl 2 are all insoluble. 4. Most silver salts are insoluble. Ag. NO 3 and Ag(C 2 H 3 O 2) are common soluble salts of silver; virtually anything else is insoluble. 5. Most sulfate salts are soluble, for example Fe. SO 4 is soluble. Important exceptions to this rule include Ba. SO , Pb. SO , Ag SO and Sr. SO . 4 4 2 4 4 6. Most hydroxide salts are only slightly soluble. Hydroxide salts of Group I elements are soluble. Hydroxide salts of Group II elements (Ca, Sr, and Ba) are slightly soluble. Hydroxide salts of transition metals and Al 3+ are insoluble. Thus, Fe(OH)3, Al(OH)3, Co(OH)2 are not soluble. 7. Most sulfides of transition metals are highly insoluble. Thus, Cu. S, Fe. S 2, Zn. S, Ag 2 S are all insoluble. Arsenic, antimony, bismuth, and lead sulfides are also insoluble. 8. Carbonates are frequently insoluble. Group II carbonates (Ca, Sr, and Ba) are insoluble. Some other insoluble carbonates include Fe. CO 3 and Pb. CO 3. 9. Chromates are frequently insoluble. Examples: Pb. Cr. O 4, Ba. Cr. O 4 10. Phosphates are frequently insoluble. Examples: Ca 3(PO 4)2, Ag 3 PO 4 11. Fluorides are frequently insoluble. Examples: Ba. F 2, Mg. F 2 Pb. F 2.

Putting insoluble sulfides into solution • Oxidizing Zone above the water table • Sulfide minerals, for example ferrous and copper sulfides, are subject to weathering. • Sulfide minerals are oxidized near the surface and produce sulfuric acid. For example: • Fe. S 2 (s) + 7 O + H 2 O →Fe. SO 4 (aq) + H 2 SO 4

Reaction and Trickling Down • Iron sulfate reacts with sulfides, they go into solution as sulfates, acid rainwater then carries, for example copper, as copper sulfate, down to the water table. • Cu. S(s) + Fe 2(SO 4)3 (aq) → 2 Fe. SO 4 (aq) + S(s) + Cu. SO 4 (aq) • The net result is that dissolved copper sulfide trickles down from the oxidizing upper portion of the deposit to that portion at and just below the water table.

Reducing Zone below the water table • Below the water table, where additional sulfide minerals remain solid and unoxidized (e. g. Pyrite Fe. S 2), any iron sulfide grains present will react with the copper sulfate solution, putting iron into solution and precipitating copper. • Fe. S 2 (s) + Cu. SO 4 (aq) → Fe. SO 4 (aq) + Cu(s) + 2 S(s)

Hydrothermal Deposit, Bemco Mine
- Slides: 59