Chapter 9 Orbitals and Covalent Bond 1 Molecular

Chapter 9 Orbitals and Covalent Bond 1

Molecular Orbitals The overlap of atomic orbitals from separate atoms makes molecular orbitals n Each molecular orbital has room for two electrons n Two types of MO – Sigma ( ) between atoms – Pi ( ) above and below atoms n 2
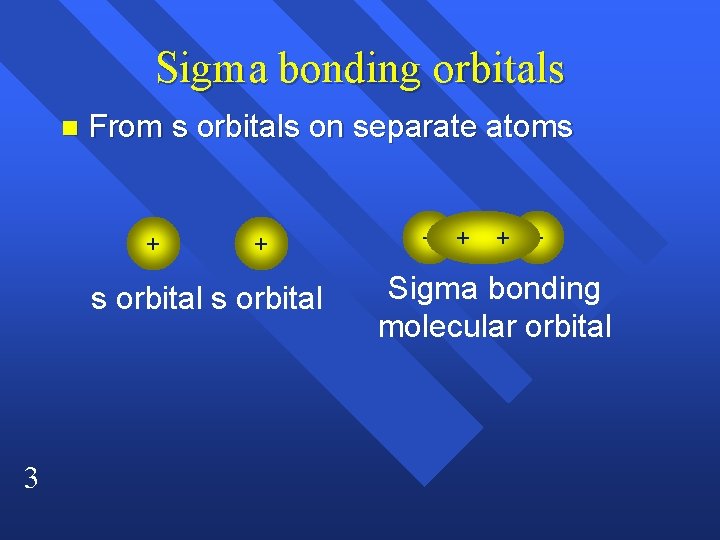
Sigma bonding orbitals n From s orbitals on separate atoms + + s orbital 3 + + Sigma bonding molecular orbital
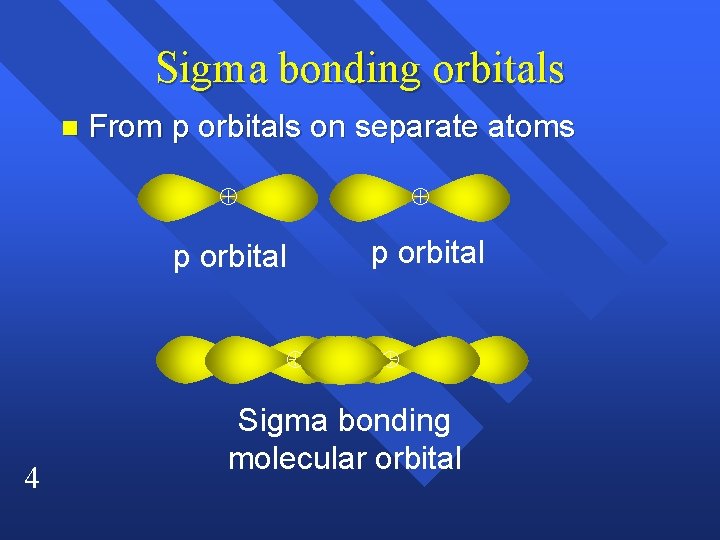
Sigma bonding orbitals n From p orbitals on separate atoms p orbital 4 p orbital Sigma bonding molecular orbital
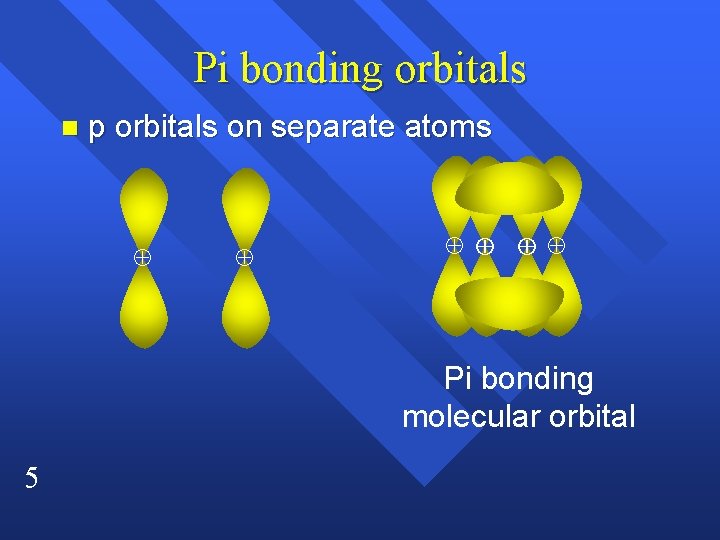
Pi bonding orbitals p orbitals on separate atoms n Pi bonding molecular orbital 5

Sigma and pi bonds All single bonds are sigma bonds n A double bond is one sigma and one pi bond n A triple bond is one sigma and two pi bonds. n 6

Atomic Orbitals Don’t Work to explain molecular geometry. n In methane, CH 4 , the shape is tetrahedral. n The valence electrons of carbon should be two in s, and two in p. n the p orbitals would have to be at right angles. n The atomic orbitals change when making a molecule n 7

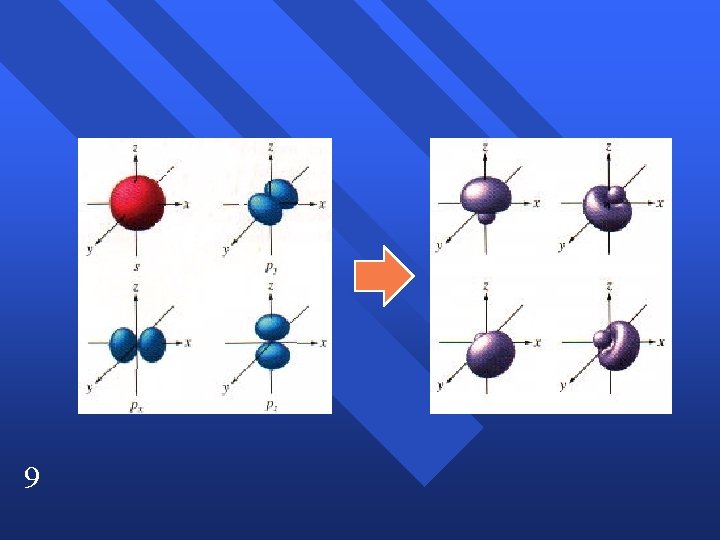
Hybridization We blend the s and p orbitals of the valence electrons and end up with the tetrahedral geometry. n We combine one s orbital and 3 p orbitals. n n 8 sp 3 hybridization has tetrahedral geometry.
9

10
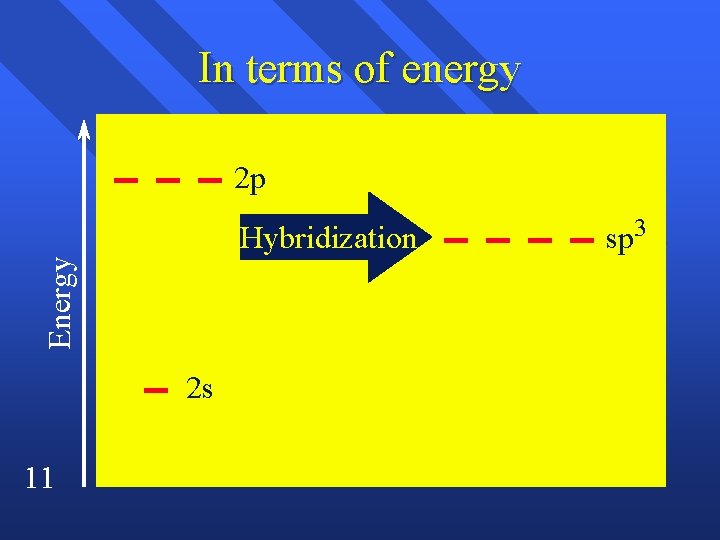
In terms of energy 2 p Energy Hybridization 2 s 11 sp 3

How we get to hybridization We know the geometry from experiment. n We know the orbitals of the atom n hybridizing atomic orbitals can explain the geometry. n So if the geometry requires a tetrahedral shape, it is sp 3 hybridized n This includes bent and trigonal pyramidal molecules because one of the sp 3 lobes holds the lone pair. n 12

sp 2 hybridization C 2 H 4 n Double bond acts as one pair. n trigonal planar n Have to end up with three blended orbitals. n Use one s and two p orbitals to make sp 2 orbitals. n Leaves one p orbital perpendicular. n 13
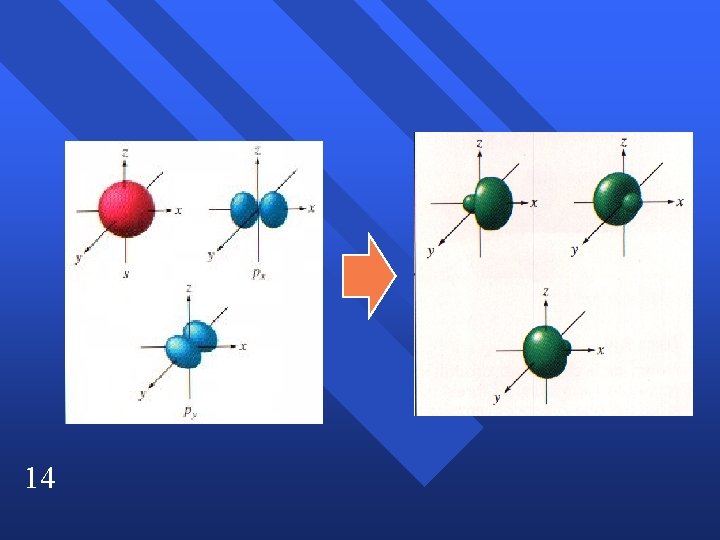
14
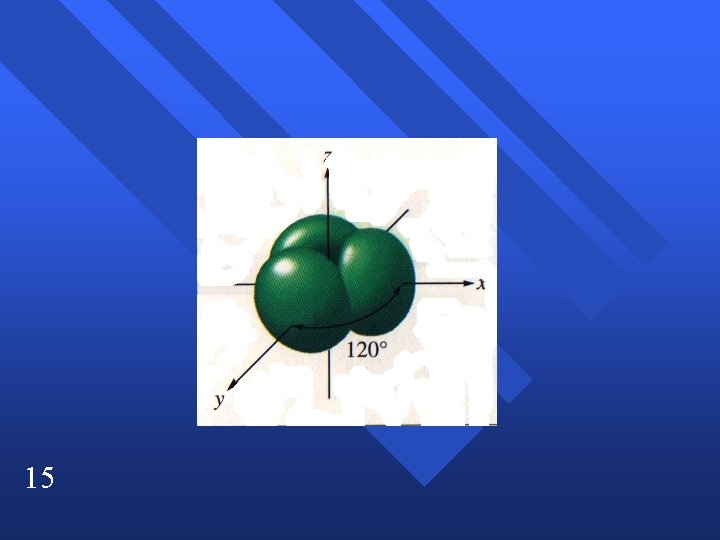
15

Energy In terms of energy 2 s 16 2 p 2 p Hybridization sp 2
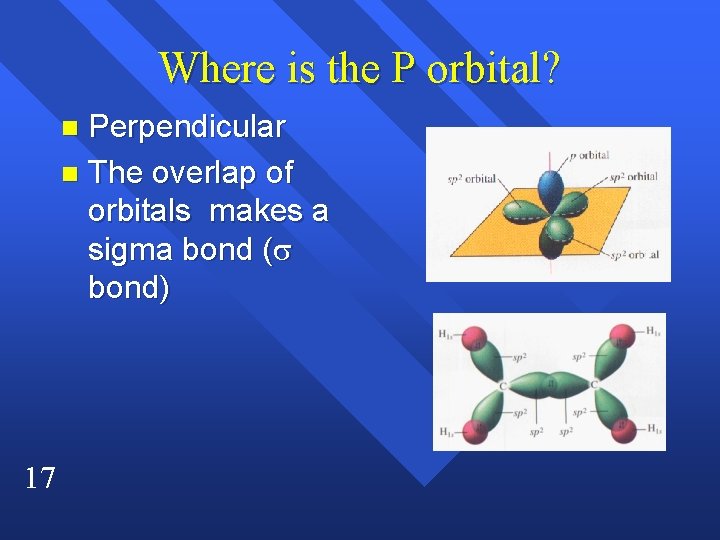
Where is the P orbital? Perpendicular n The overlap of orbitals makes a sigma bond ( bond) n 17

Two types of Bonds Sigma bonds from overlap of orbitals. n Between the atoms. n Pi bond ( bond) above and below atoms n Between adjacent p orbitals. n The two bonds of a double bond. n 18
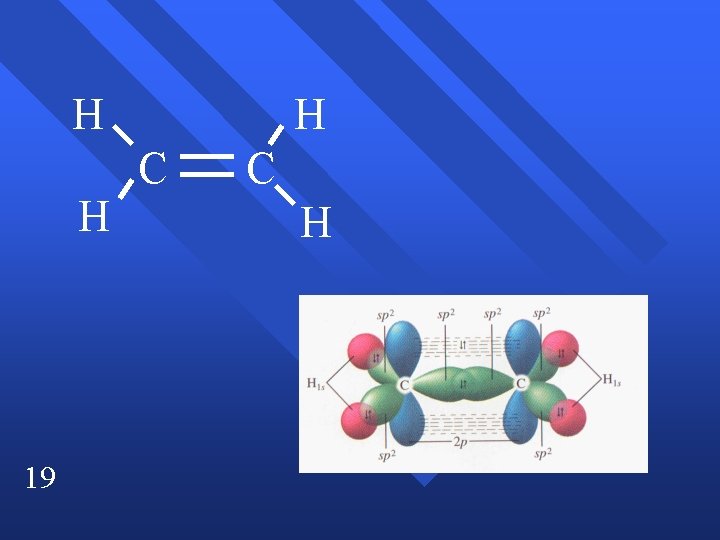
H H 19 H C C H

sp 2 hybridization When three things come off atom. n trigonal planar n 120º n One bond, + lp =3 n 20
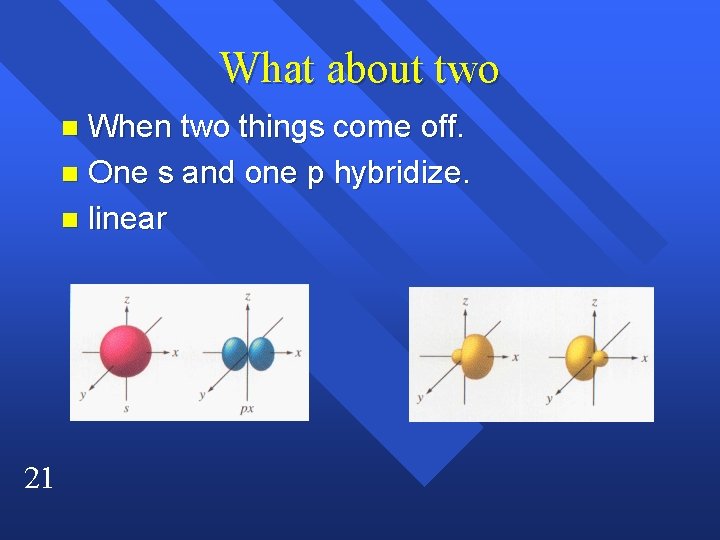
What about two When two things come off. n One s and one p hybridize. n linear n 21

sp hybridization End up with two lobes 180º apart. n p orbitals are at right angles n Makes room for two bonds and two sigma bonds. n A triple bond or two double bonds. n 22
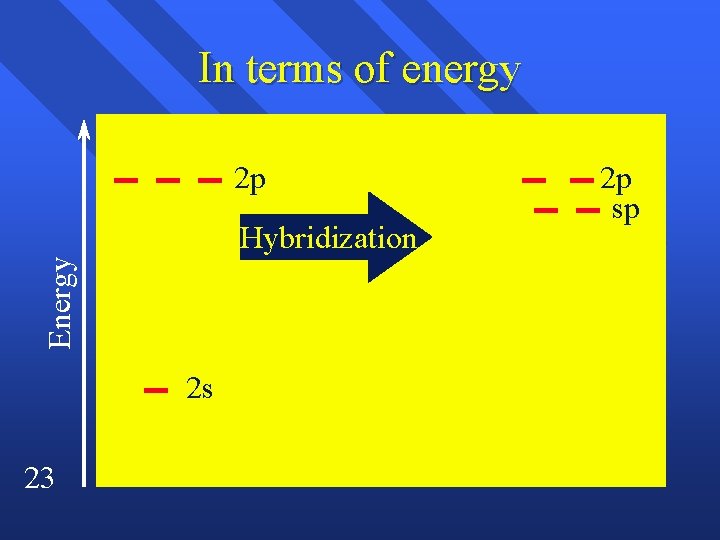
In terms of energy 2 p Energy Hybridization 2 s 23 2 p sp
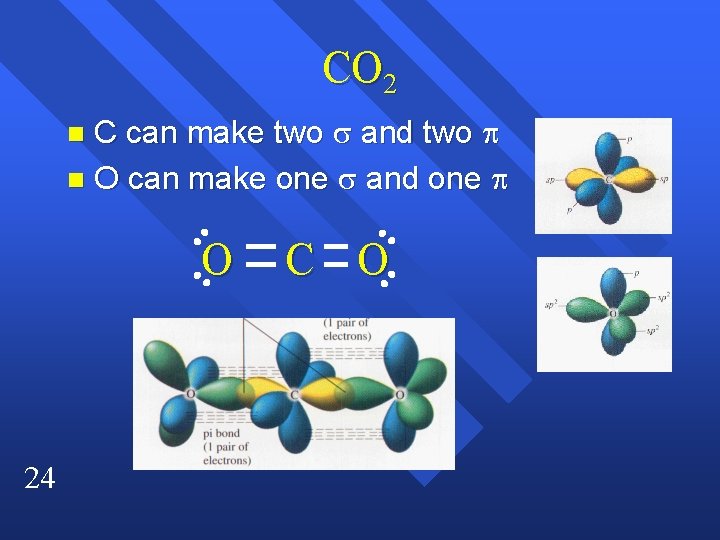
CO 2 C can make two and two n O can make one and one n O 24 C O
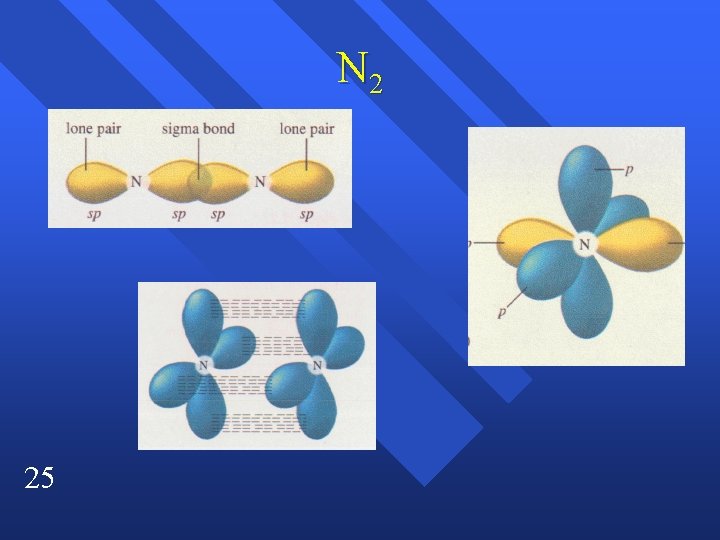
N 2 25
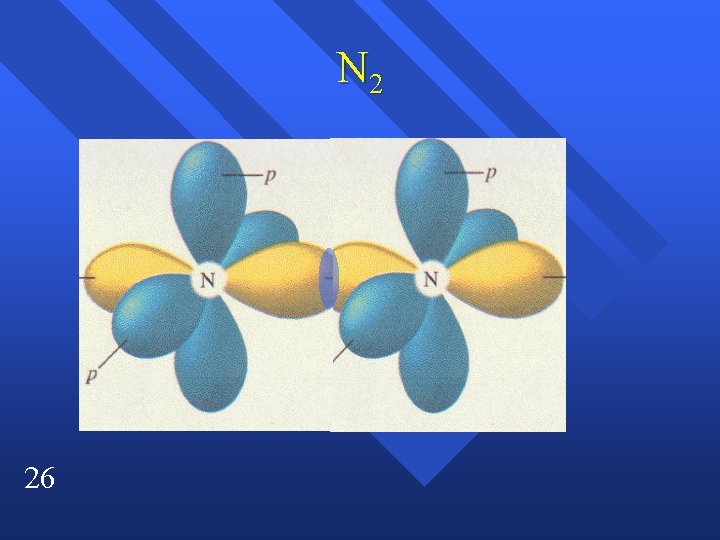
N 2 26

Breaking the octet PCl 5 n The model predicts that we must use the d orbitals. n dsp 3 hybridization n There is some controversy about how involved the d orbitals are. n 27

dsp 3 Trigonal bipyrimidal n can only bond. n can’t bond. n basic shape for five things. n 28
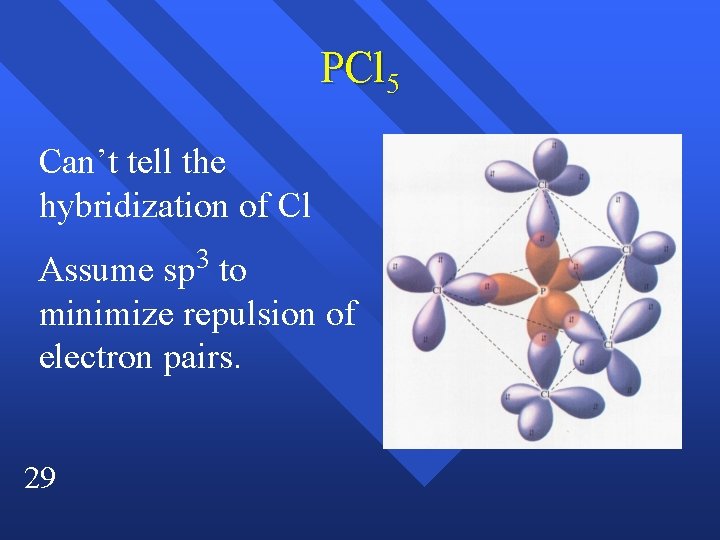
PCl 5 Can’t tell the hybridization of Cl Assume sp 3 to minimize repulsion of electron pairs. 29

d 2 sp 3 gets us to six things around n Octahedral n Only σ bond n 30

Molecular Orbital Model Localized Model we have learned explains much about bonding. n It doesn’t deal well with the ideal of resonance, unpaired electrons, and bond energy. n The MO model is a parallel of the atomic orbital, using quantum mechanics. n Each MO can hold two electrons with opposite spins 31 n Square of wave function tells probability n

What do you get? Solve the equations for H 2 n HA H B n get two orbitals n 32 n MO 2 = 1 s. A - 1 s. B n MO 1 = 1 s. A + 1 s. B

The Molecular Orbital Model • The molecular orbitals are centered on a line through the nuclei – MO 1 the greatest probability is between the nuclei – MO 2 it is on either side of the nuclei – this shape is called a sigma molecular orbital 33

The Molecular Orbital Model • In the molecule only the molecular • • 34 orbitals exist, the atomic orbitals are gone MO 1 is lower in energy than the 1 s orbitals they came from. – This favors molecule formation – Called an bonding orbital MO 2 is higher in energy – This goes against bonding – antibonding orbital
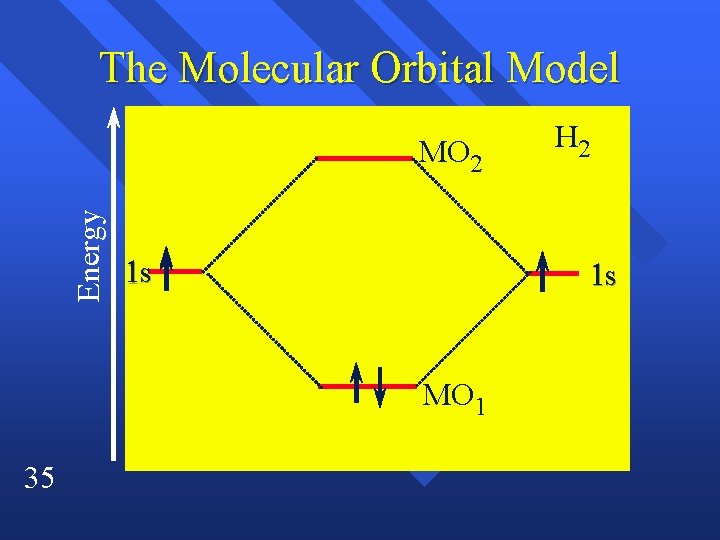
The Molecular Orbital Model Energy MO 2 1 s 1 s MO 1 35 H 2

The Molecular Orbital Model • We use labels to indicate shapes, and • 36 whether the MO’s are bonding or antibonding. – MO 1 = 1 s – MO 2 = 1 s* (* indicates antibonding) Can write them the same way as atomic orbitals – H 2 = 1 s 2

The Molecular Orbital Model • Each MO can hold two electrons, but • 37 they must have opposite spins Orbitals are conserved. • The number of molecular orbitals must equal the number atomic orbitals that are used to make them.
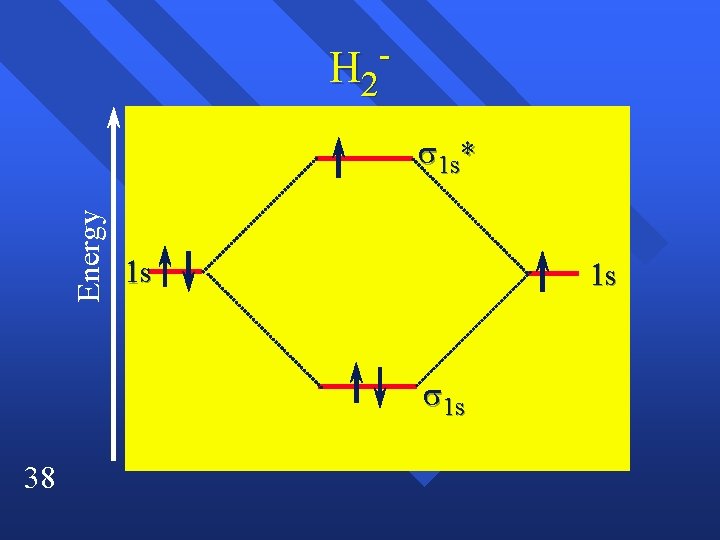
H 2 Energy 1 s* 1 s 1 s 38

Bond Order n 39 The difference between the number of bonding electrons and the number of antibonding electrons divided by two

Only outer orbitals bond The 1 s orbital is much smaller than the 2 s orbital n When only the 2 s orbitals are involved in bonding n Don’t use the 1 s or 1 s* for Li 2 n Li 2 = ( 2 s)2 n In order to participate in bonds the orbitals must overlap in space. 40 n

Bonding in Homonuclear Diatomic Molecules n n n 41 n Need to use Homonuclear so that we know the relative energies. Li 2( 2 s)2 ( 2 s*)1 Be 2 ( 2 s)2 ( 2 s*)2 What about the p orbitals? How do they form orbitals? Remember that orbitals must be conserved.
B 2 42
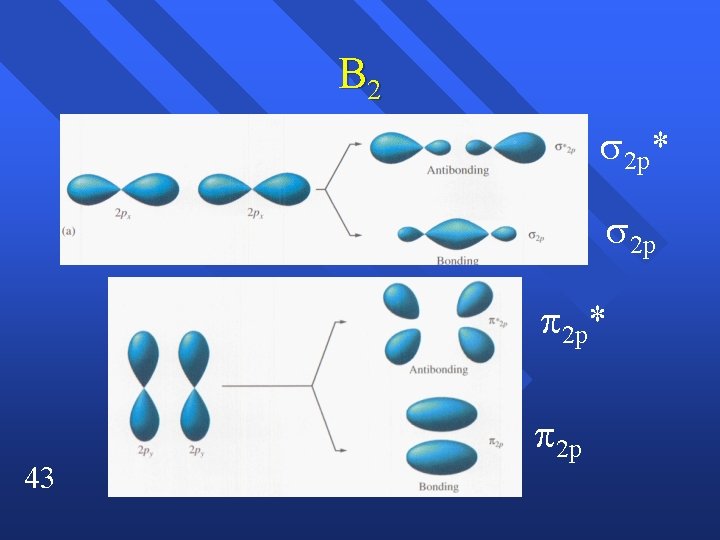
B 2 2 p* 2 p 2 p* 43 2 p
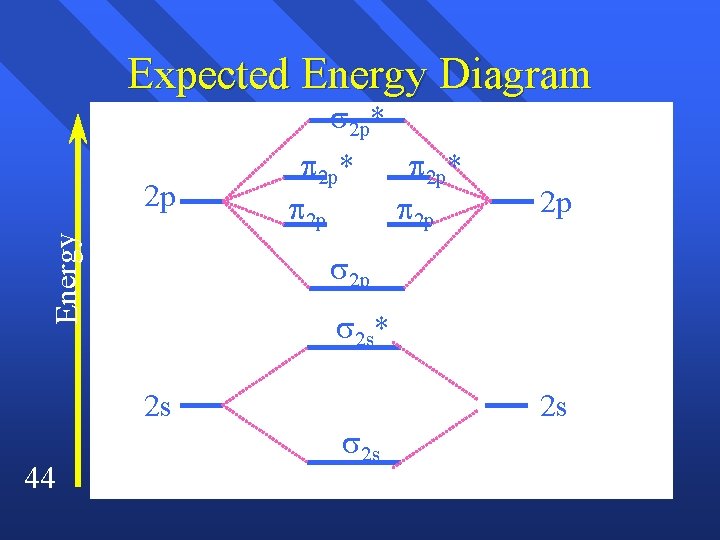
Expected Energy Diagram 2 p* Energy 2 p 2 p* 2 p 2 p 2 s* 2 s 44 2 p* 2 p 2 s 2 s
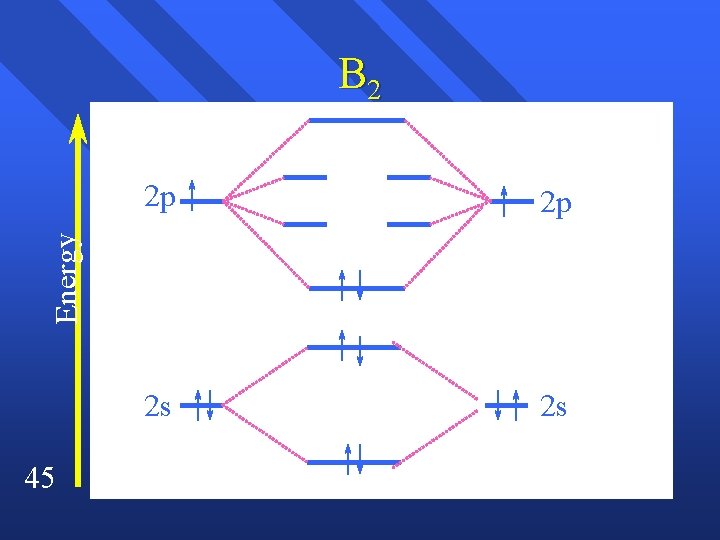
B 2 2 p 2 s 2 s Energy 2 p 45

B 2 ( 2 s)2( 2 s*)2 ( 2 p)2 n Bond order = (4 -2) / 2 n Should be stable. n This assumes there is no interaction between the s and p orbitals. n Hard to believe since they overlap n proof comes from magnetism. n 46

Magnetism has to do with electrons. n Remember that spin is how an electron reacts to a magnetic field n Paramagnetism attracted by a magnet. – associated with unpaired electrons. n Diamagnetism repelled by a magnet. – associated with paired electrons. n B 2 is paramagnetic. n 47

Magnetism The energies of of the 2 p and the 2 p are reversed by p and s interacting n The 2 s and the 2 s* are no longer equally spaced. n Here’s what it looks like. n 48
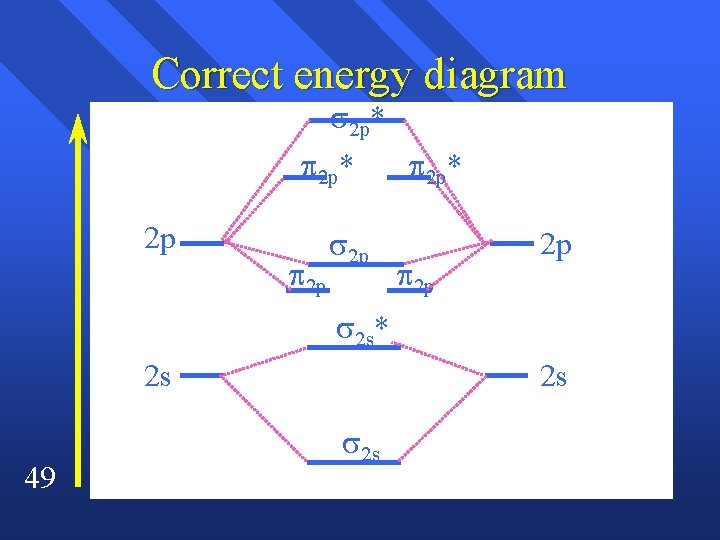
Correct energy diagram 2 p* 2 p 2 p 2 p* 2 p 2 p 2 s* 2 s 49 2 s 2 s
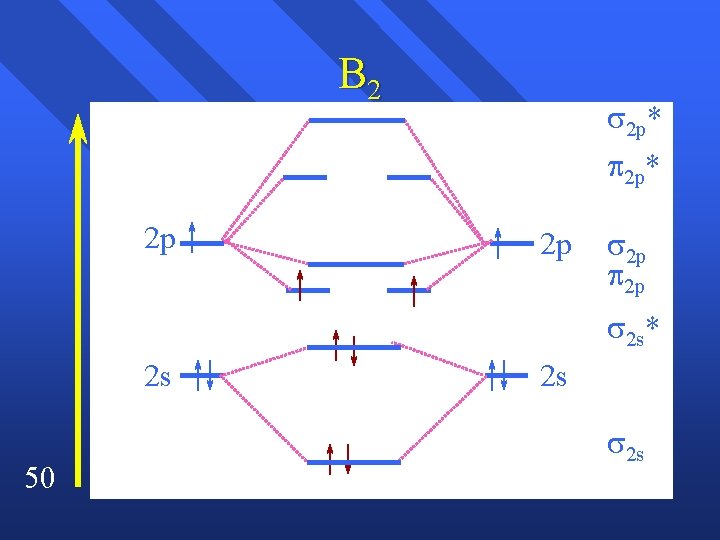
B 2 2 p* 2 p 2 p 2 s* 2 s 50 2 s 2 s

Patterns As bond order increases, bond energy increases. n As bond order increases, bond length decreases. n Supports basis of MO model. n There is not a direct correlation of bond order to bond energy. n O 2 is known to be paramagnetic. n Movie. n 51

Magnetism Ferromagnetic strongly attracted n Paramagnetic weakly attracted – Liquid Oxygen n Diamagnetic weakly repelled – Graphite – Water Frog n 52

Examples C 2 n N 2 n O 2 n F 2 n P 2 n 53

Heteronuclear Diatomic Species Simple type has them in the same energy level, so can use the orbitals we already know. n Slight energy differences. n NO n 54
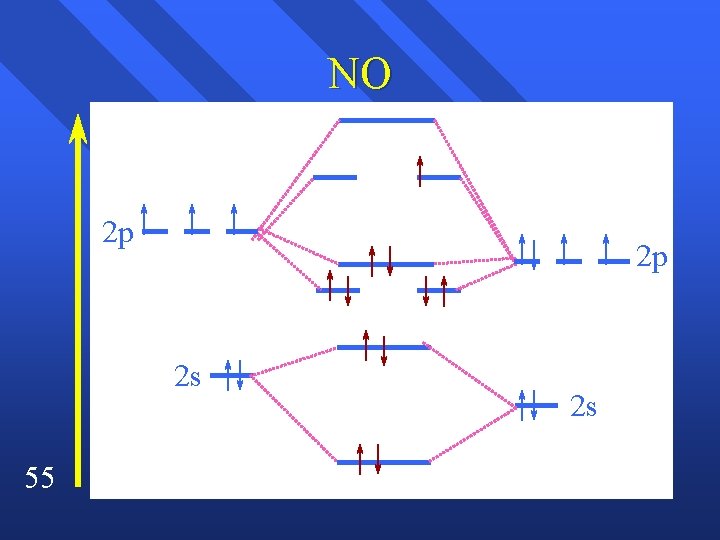
NO 2 p 2 p 2 s 55 2 s

You try NO+ n CNn What if they come from completely different orbitals and energy? n HF n Simplify first by assuming that F only uses one if its 2 p orbitals. n F holds onto its electrons, so they have low energy n 56
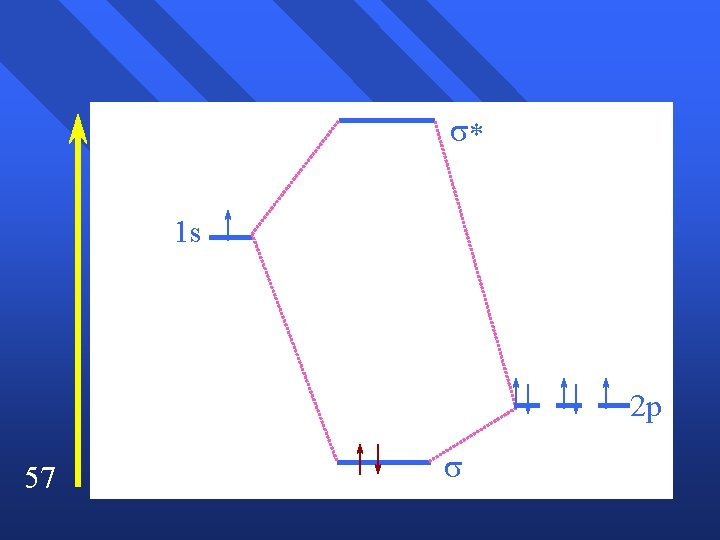
* 1 s 2 p 57

Consequences Paramagnetic n Since 2 p is lower in energy, favored by electrons. n Electrons spend time closer to fluorine. n Compatible with polarity and electronegativity. n 58

Names sp orbitals are called the Localized electron model n and Molecular orbital model n Localized is good for geometry, doesn’t deal well with resonance. n seeing bonds as localized works well n It is the bonds in the resonance structures that can move. n 59
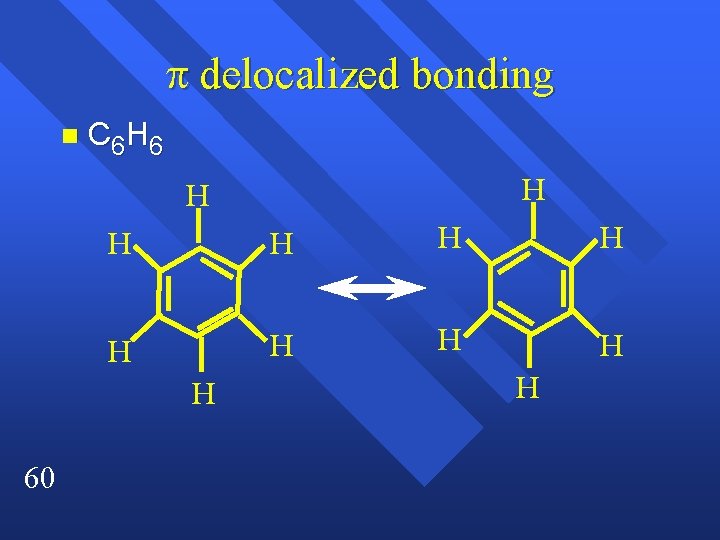
delocalized bonding n C 6 H 6 H H H 60 H
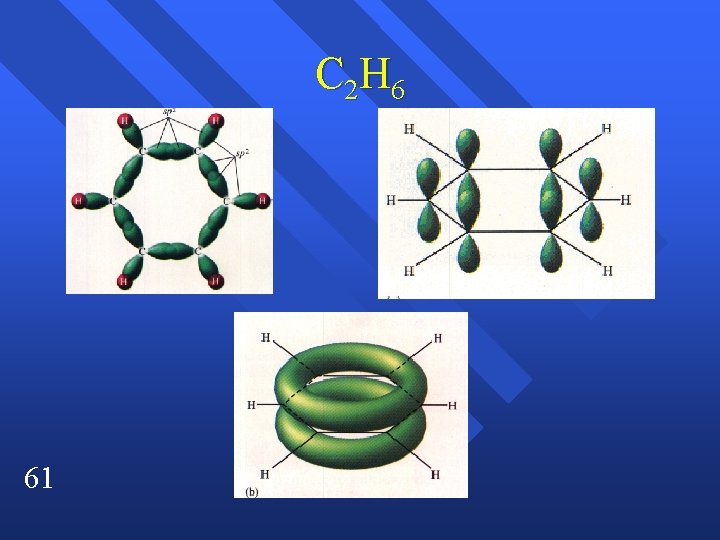
C 2 H 6 61
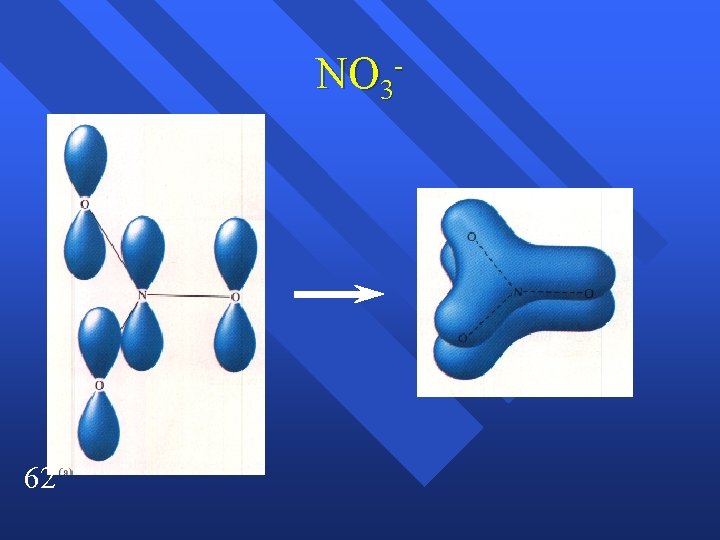
NO 3 - 62
- Slides: 62