Chapter 9 Nuclear Radiation 1 9 1 Natural









- Slides: 10

Chapter 9 Nuclear Radiation 1 9. 1 Natural Radioactivity Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Stable and Radioactive Isotopes 2 A radioactive isotope • has an unstable nucleus • emits radiation to become more stable, changing the composition of the nucleus • is identified by the mass number of the isotope Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Nuclear Radiation 3 Nuclear radiation is the radiation emitted by an unstable atom and takes the form of alpha particles, neutrons, beta particles, positrons, or gamma rays. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Types of Radiation 4 Alpha ( ) particle is two protons and two neutrons. Beta ( ) particle is a high-energy electron. Positron ( +) is a positive electron. Gamma rays are the high-energy radiation released from a nucleus when it decays. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 5 Give the name and symbol for the following types of radiation. A. alpha radiation B. radiation with a mass number of 0 and atomic number of +1 C. radiation that represents high-energy radiation released from the nucleus Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 6 Give the name and symbol for the following types of radiation. A. alpha radiation B. radiation with a mass number of 0 and atomic number of +1 C. radiation that represents high-energy radiation released from the nucleus Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

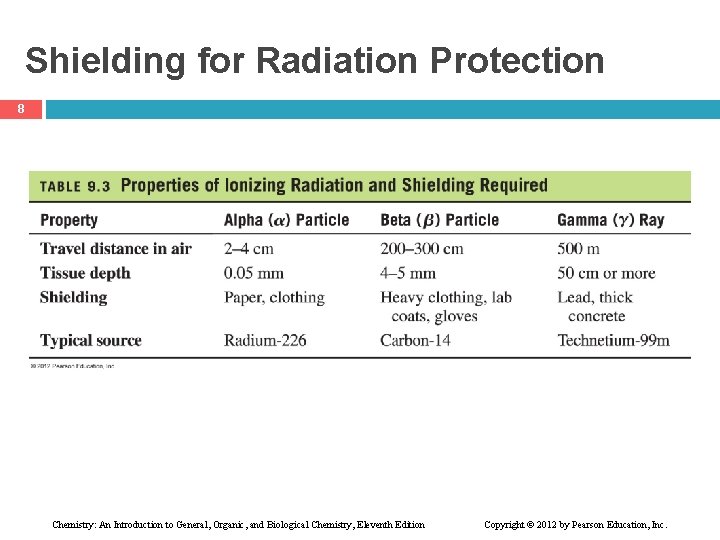
Radiation Protection 7 Radiation protection requires • paper and clothing for alpha particles • a lab coat or gloves for beta particles • a lead shield or a thick concrete wall for gamma rays • limiting the amount of time spent near a radioactive source • increasing distance from the source Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Shielding for Radiation Protection 8 Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Learning Check 9 What type of radiation will be blocked by A. lead shield B. paper, clothing Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.

Solution 10 What type of radiation will be blocked by A. lead shield B. paper, clothing gamma radiation alpha radiation Chemistry: An Introduction to General, Organic, and Biological Chemistry, Eleventh Edition Copyright © 2012 by Pearson Education, Inc.